Abstract
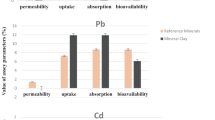
Clay minerals, such as layered double hydroxide (LDH) and montmorillonite (MMT), have attracted a great deal of attention for biological applications. Along with the rapid development of nanotechnology, public concern about the potential toxicity of nanoparticles is growing. In the present work, cytotoxicity of LDH and MMT was assessed in terms of inhibition of cell proliferation, generation of oxidative stress, and induction of inflammation response. Moreover, the biokinetics of LDH and MMT were evaluated; biokinetics provides information about in vivo absorption, distribution, and excretion kinetics. The results demonstrated that both LDH and MMT inhibited cell proliferation at relatively large concentrations and after long exposure time compared to other inorganic nanoparticles, although they generated reactive oxygen species (ROS). LDH induced pro-inflammatory cytokines in a size-dependent manner. Biokinetic study revealed that, after single-dose oral administration to mice, both LDH and MMT had extremely slow oral rates of absorption and did not accumulate in any specific organ. All the results suggest great potential of clay minerals for biological application at safe levels.










Similar content being viewed by others
References
Baek, M. & Choi, S. J. (2012). Effect of orally administered glutathione-montmorillonite hybrid systems on tissue distribution. Journal of Nanomaterials, 2012, 469372.
Baek, M., Kim, I. S., Yu, J., Chung, H. E., Choy, J. H., & Choi, S. J. (2011). Effect of different forms of anionic nanoclays on cytotoxicity. Journal of Nanoscience Nanotechnology, 11, 1803–1806.
Baek, M., Choy, J. H., & Choi, S. J. (2012a). Montmorillonite intercalated with glutathione for antioxidant delivery: synthesis, characterization, and bioavailability evaluation. International Journal of Pharmaceutics, 425, 29–34.
Baek, M., Chung, H. E., Yu, J., Lee, J. A., Kim, T. H., Oh, J. M., Lee, W. J., Paek, S. M., Lee, J. K., Jeong, J., Choy, J. H., & Choi, S. J. (2012b). Pharmacokinetics, tissue distribution, and excretion of zinc oxide nanoparticles. International Journal Nanomedicine, 7, 3081–3097.
Baek, M., Lee, J. A., & Choi, S. J. (2012c). Toxicological effects of a cationic clay, montmorillonite in vitro and in vivo. Molecular & Cellular Toxicology, 8, 95–101.
Chan, J. Y. W., Tsui, J. C. C., Law, P. T. W., So, W. K. W., Leung, D. Y. P., Sham, M. M. K., Tsui, S. K. W., & Chan, C. W. H. (2018). RNA-Seq revealed ATF3-regulated inflammation induced by silica. Toxicology, 393, 34–41.
Choi, S. J., Choi, G. E., Oh, J. M., Oh, Y. J., Park, M. C., & Choy, J. H. (2010). Anticancer drug encapsulated in inorganic lattice can overcome drug resistance. Journal of Materials Chemistry, 20, 9463–9469.
Choi, S. J. & Choy, J. H. (2011a). Layered double hydroxide nanoparticles as target-specific delivery carriers: uptake mechanism and toxicity. Nanomedicine, 6, 803–814.
Choi, S. J. & Choy, J. H. (2011b). Effect of physico-chemical parameters on the toxicity of inorganic nanoparticles. Journal of Materials Chemistry, 21, 5547–5554.
Choi, S. J. & Choy, J. H. (2014). Biokinetics of zinc oxide nanoparticles: toxicokinetics, biological fates, and protein interaction. International Journal of Nanomedicine, 9, 261–269.
Choi, S. J. Oh, J. M., & Choy, J. H. (2008a). Human-related application and nanotoxicology of inorganic particles: complementary aspects. Journal of Materials Chemistry, 18, 615–620.
Choi, S. J., Oh, J. M., & Choy, J. H. (2008b). Safety aspect of inorganic layered nanoparticles: size-dependency in vitro and in vivo. Journal of Nanoscience Nanotechnology, 8, 5297–5301.
Choi, S. J., Oh, J. M., & Choy, J. H. (2009). Toxicological effects of inorganic nanoparticles on human lung cancer A549 cells. Journal of Inorganic Biochemistry, 103, 463–471.
Choi, S. J., Paek, H. J., & Yu, J. (2015). Oxidative stress by layered double hydroxide nanoparticles via an SFK-JNK and p38-NF-kappaB signaling pathway mediates induction of interleukin-6 and interleukin-8 in human lung epithelial cells. International Journal of Nanomedicine, 10, 3217–3229.
Choy, J. H., Choi, S. J., Oh, J. M., & Park, T. (2007). Clay minerals and layered double hydroxides for novel biological applications. Applied Clay Science, 36, 122–132.
Chung, H. E., Park, D. H., Choy, J. H., & Choi, S. J. (2012). Intracellular trafficking pathway of layered double hydroxide nanoparticles in human cells: Size-dependent cellular delivery. Applied Clay Science, 65–66, 24–30.
Jo, M. R., Yu, J., Kim, H. J., Song, J. H., Kim, K. M., Oh, J. M., & Choi, S. J. (2016). Titanium dioxide nanoparticle-biomolecule interactions influence oral absorption. Nanomaterials (Basel), 6.
Kim, M. K., Lee, J. A., Jo, M. R., & Choi, S. J. (2016). Bioavailability of silica, titanium dioxide, and zinc oxide nanoparticles in rats. Journal of Nanoscience and Nanotechnology, 16, 6580–6586.
Ng, C. T., Yong, L. Q., Hande, M. P., Ong, C. N., Yu, L. E., Bay, B. H., & Baeg, G. H. (2017). Zinc oxide nanoparticles exhibit cytotoxicity and genotoxicity through oxidative stress responses in human lung fibroblasts and Drosophila melanogaster. International Journal of Nanomedicine, 12, 1621–1637.
Oh, J. M., Choi, S. J., Kim, S. T., & Choy, J. H. (2006). Cellular uptake mechanism of an inorganic nanovehicle and its drug conjugates: Enhanced efficacy due to clathrin-mediated endocytosis. Bioconjugugate Chemistry, 17, 1411–1417.
Oh, J. M., Choi, S. J., Lee, G. E., Kim, J. E., & Choy, J. H. (2009). Inorganic metal hydroxide nanoparticles for targeted cellular uptake through clathrin-mediated endocytosis. Chemistry – An Asian Journal, 4, 67–73.
Paek, H. J., Chung, H. E., Lee, J. A., Kim, M. K., Lee, Y. J., Kim, M. S., Kim, S. H., Maeng, E. H., Lee, J. K., Jeong, J., & Choi, S. J. (2014). Quantitative determination of silica nanoparticles in biological matrices and their pharmacokinetics and toxicokinetics in rats. Science of Advanced Materials, 6, 1605–1610.
Paek, H. J., Lee, Y. J., Chung, H. E., Yoo, N. H., Lee, J. A., Kim, M. K., Lee, J. K., Jeong, J., & Choi, S. J. (2013). Modulation of the pharmacokinetics of zinc oxide nanoparticles and their fates in vivo. Nanoscale, 5, 11416–11427.
Pongrac, I. M., Pavicic, I., Milic, M., Brkic Ahmed, L., Babic, M., Horak, D., Vinkovic Vrcek, I., & Gajovic, S. (2016). Oxidative stress response in neural stem cells exposed to different superparamagnetic iron oxide nanoparticles. International Journal of Nanomedicine, 11, 1701–1715.
Poulsen, S. S., Jackson, P., Kling, K., Knudsen, K. B., Skaug, V., Kyjovska, Z. O., Thomsen, B. L., Clausen, P. A., Atluri, R., Berthing, T., Bengtson, S., Wolff, H., Jensen, K. A., Wallin, H., & Vogel, U. (2016). Multi-walled carbon nanotube physicochemical properties predict pulmonary inflammation and genotoxicity. Nanotoxicology, 10, 1263–1275.
Shvedova, A. A., Pietroiusti, A., Fadeel, B., & Kagan, V. E. (2012). Mechanisms of carbon nanotube-induced toxicity: focus on oxidative stress. Toxicology and Applied Pharmacology, 261, 121–133.
Yu, J., Chung, H. E., & Choi, S. J. (2013). Acute oral toxicity and kinetic behaviors of inorganic layered nanoparticles. Journal of Nanomaterials. https://doi.org/10.1155/2013/628381.
Zuo, D., Duan, Z., Jia, Y., Chu, T., He, Q., Yuan, J., Dai, W., Li, Z., Xing, L., & Wu, Y. (2016). Amphipathic silica nanoparticles induce cytotoxicity through oxidative stress mediated and p53 dependent apoptosis pathway in human liver cell line HL-7702 and rat liver cell line BRL-3A. Colloids and Surfaces B: Biointerfaces, 145, 232–240.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2018R1A2B6001238).
Author information
Authors and Affiliations
Corresponding author
Additional information
(Received 25 April 2018; revised 7 August 2018; AE: J.-H. Choy)
Rights and permissions
About this article
Cite this article
Choi, SJ. Cytotoxicity and Biokinetic Evaluation of Clay Minerals. Clays Clay Miner. 67, 91–98 (2019). https://doi.org/10.1007/s42860-019-0007-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42860-019-0007-y