Abstract
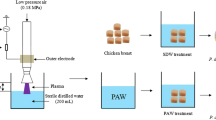
Ozone has been studied to control microorganisms in food, as well as to control biofilm. In this context, the goals of this work were to determine the effect of ozonated water in the removal of Pseudomonas paracarnis biofilm and the effect of ozone gas and ozonated water on inactivating P. paracarnis in deboned chicken breast meat and its effect on product color. AISI 304 coupons were used as a surface for biofilm formation. The coupons were immerged into minimal medium for Pseudomonas inoculated with the P. paracarnis overnight culture (1% w/v) followed by incubation at 25 °C for 7 days. To obtain ozonized water, two different systems were used: system with microbubble generator (MB) and system with porous stone diffuser (PSD). The inlet ozone concentration was 19 mg/L and flow rate of 1 L/min. The coupons were subjected to ozonized water for 10 and 20 min. The chicken breast meat was exposed to gaseous ozone and ozonized water for 40 min. After the ozonation process, chicken meat samples were stored at 8 °C, for 5 days. More expressive removals of biofilm were obtained when using ozonized water obtained in the system with microbubble generator (MB for 20 min—reduction of 2.3 log cycles) and system with porous stone diffuser (PSD for 10 min—reduction of 2.7 log cycles; PSD for 20 min—reduction of 2.6 log cycles). The treatment of chicken meat with ozone gas resulted in lower counting of Pseudomonas, when compared with the control treatments and with ozonized water, both immediately after ozonation (day 1) and after 5 days of storage. The luminosity in the chicken meat samples treated with ozonized water was higher than that verified in the control treatments and with ozone gas, immediately after ozonation (day 1). A similar trend was observed in hue angle and color difference, in which the highest values were obtained for treatment with ozonized water. Based on the results obtained in this study, it was concluded that ozonated water can be used to remove P. paracarnis biofilm from stainless steel under static conditions and gaseous ozone is more efficient in the inactivation of P. paracarnis from chicken breast meat, when compared to ozonated water.



Similar content being viewed by others
References
Samapundo S, de Baenst I, Aerts M, Cnockaert M, Devlieghere F, Van Damme P (2019) Tracking the sources of psychrotrophic bacteria contaminating chicken cuts during processing. Food Microbiol 81:40–50. https://doi.org/10.1016/j.fm.2018.06.003
Ayranci UG, Ozunlu O, Ergezer H, Karaca H (2020) Effects of ozone treatment on microbiological quality and physicochemical properties of turkey breast meat. Ozone-Sci Eng 42(1):95–103. https://doi.org/10.1080/01919512.2019.1653168
Nel S, Lues JFR, Buys EM, Venter P (2004) Bacterial populations associated with meat from the deboning room of a high throughput red meat abattoir. Meat sci 66(3):667–674. https://doi.org/10.1016/S0309-1740(03)00187-6
Shale K, Jacoby A, Plaatjies Z (2006) The impact of extrinsic sources on selected indicator organisms in a typical deboning room. Int J Environ Health Res 16(4):263–272. https://doi.org/10.1080/09603120600734162
Williams GD, Keener KM (2008) Design considerations for the construction and operation of meat and poultry processing facilities. In: Proc Annu Int Meet Am Soc Agric Biol Eng 2008. Providence, pp 1–25. https://doi.org/10.13031/2013.24882
Botta C, Ferrocino I, Pessione A, Cocolin L, Rantsiou K (2020) Spatiotemporal distribution of the environmental microbiota in food processing plants as impacted by cleaning and sanitizing procedures: the case of slaughterhouses and gaseous ozone. Appl Environ Microbiol 86(23):e01861-e1920. https://doi.org/10.1128/AEM.01861-20
Chen SH, Fegan N, Kocharunchitt C, Bowman JP, Duffy LL (2020) Changes of the bacterial community diversity on chicken carcasses through an Australian poultry processing line. Food Microb 86:103350. https://doi.org/10.1016/j.fm.2019.103350
Wickramasinghe NN, Ravensdale J, Coorey R, Chandry SP, Dykes GA (2019) The predominance of psychrotrophic pseudomonads on aerobically stored chilled red meat. Compr Rev Food Sci Food Saf 18(5):1622–1635. https://doi.org/10.1111/1541-4337.12483
Rajmohan S, Dodd CER, Waites WM (2002) Enzymes from isolates of Pseudomonas fluorescens involved in food spoilage. J Appl Microbiol 93(2):205–213. https://doi.org/10.1046/j.1365-2672.2002.01674.x
Lick S, Kröckel L, Wibberg D, Winkler A, Blom J, Bantleon A et al (2020) Pseudomonas carnis sp. nov,. isolated from meat. Int J Syst Evol Microbiol 70(3):1528–1540. https://doi.org/10.1099/ijsem.0.003928
Lick S, Wibberg D, Winkler A, Blom J, Grimmler C, Goesmann A et al (2021) Pseudomonas paracarnis sp. nov,. isolated from refrigerated beef. Int J Syst Evol Microbiol 71(2):004652. https://doi.org/10.1099/ijsem.0.004652
Marino M, Maifreni M, Baggio A, Innocente N (2018) Inactivation of foodborne bacteria biofilms by aqueous and gaseous ozone. Front Microbiol 9:1–12. https://doi.org/10.3389/fmicb.2018.02024
Waters EM, Rowe SE, O’Gara JP, Conlon BP (2016) Convergence of Staphylococcus aureus persister and biofilm research: can biofilms be defined as communities of adherent persister cells? PLoS Pathog 12(12):e1006012. https://doi.org/10.1371/journal.ppat.1006012
Masák J, Čejková A, Schreiberová O, Řezanka T (2014) Pseudomonas biofilms: possibilities of their control. FEMS Microbiol Ecol 89(1):1–14. https://doi.org/10.1111/1574-6941.12344
Kim JG, Yousef AE, Dave S (1999) Application of ozone for enhancing the microbiological safety and quality of foods: a review. J Food Protect 62:1071–1087. https://doi.org/10.4315/0362-028x-62.9.1071
Cullen PJ, Tiwari BK, O’Donnell CP, Muthukumarappan K (2009) Modelling approaches to ozone processing of liquid foods. Trends Food Sci Technol 20(3–4):125–136. https://doi.org/10.1016/j.tifs.2009.01.049
Alencar ER, Faroni LRDA, Soares NDFF, da Silva WA, da Silva Carvalho MC (2012) Efficacy of ozone as a fungicidal and detoxifying agent of aflatoxins in peanuts. J Sci Food Agric 92(4):899–905. https://doi.org/10.1002/jsfa.4668
Epelle EI, Macfarlane A, Cusack M, Burns A, Thissera B, Mackay W, Rateb ME (2022) Bacterial and fungal disinfection via ozonation in air. J Microbiol Methods 194:106431. https://doi.org/10.1016/j.mimet.2022.106431
Silva MVDA, Faroni LRDA, de Alencar ER, de Sousa AH, Cecon PR, Nogueira JVF, Mason Filho V (2022) Ozone injection at low pressure: decomposition kinetics, control of Sitophilus zeamais, and popcorn kernel quality. Ozone-Sci Eng 44(1):66–78. https://doi.org/10.1080/01919512.2021.1937043
Parish ME, Beuchat LR, Suslow TV, Harris LJ, Garrett EH, Farber JN, Busta FF (2003) Methods to reduce/eliminate pathogens from fresh and fresh-cut produce. Compr Rev Food Sci Food Saf 2:161–173
King BS, Page EH, Mueller CA, Dollberg DD, Gomez KE, Warren AM (2006) Eye and respiratory symptoms in poultry processing workers exposed to chlorine by-products. Am J Ind Med 49(2):119–126. https://doi.org/10.1002/ajim.20259
Cardador MJ, Gallego M (2018) Determination of several common disinfection by-products in frozen foods. Food Addit Contam A 35(1):56–65. https://doi.org/10.1080/19440049.2017.1382731
Pandiselvam R, Subhashini S, BanuuPriya EP, Kothakota A, Ramesh SV, Shahir S (2019) Ozone based food preservation: a promising green technology for enhanced food safety. Ozone: Sci Eng 41(1):17–34. https://doi.org/10.1080/01919512.2021.1904204
Pandiselvam R, Thirupathi V (2015) Reaction kinetics of ozone gas in green gram (Vigna radiate). Ozone-Sci Eng 37(4):309–315. https://doi.org/10.1080/01919512.2014.984158
Pandiselvam R, Sunoj S, Manikantan MR, Kothakota A, Hebbar KB (2017) Application and kinetics of ozone in food preservation. Ozone-Sci Eng 39(2):115–126. https://doi.org/10.1080/01919512.2016.1268947
Korany AM, Hua Z, Green T, Hanrahan I, El-Shinawy SH, El-Kholy A, Hassan G, Zhu MJ (2018) Efficacy of ozonated water, chlorine, chlorine dioxide, quaternary ammonium compounds and peroxyacetic acid against Listeria monocytogenes biofilm on polystyrene surfaces. Front Microbiol 9:2296. https://doi.org/10.3389/fmicb.2018.02296
Harada AMM, Nascimento MS (2021) Effect of dry sanitizing methods on Bacillus cereus biofilm. Braz J Microbiol 52(2):919–926. https://doi.org/10.1007/2Fs42770-021-00451-0
Cárdenas FC, Andres S, Giannuzzi L, Zaritzky N (2011) Antimicrobial action and effects on beef quality attributes of a gaseous ozone treatment at refrigeration temperatures. Food Cont 22(8):1442–1447. https://doi.org/10.1016/j.foodcont.2011.03.006
Panebianco F, Rubiola S, Chiesa F, Civera T, Di Ciccio PA (2021) Effect of gaseous ozone on Listeria monocytogenes planktonic cells and biofilm: an in vitro study. Foods 10(7):1484. https://doi.org/10.3390/foods10071484
Kanaan MH (2018) Antibacterial effect of ozonated water against methicillin-resistant Staphylococcus aureus contaminating chicken meat in Wasit Province Iraq. Vet World 11(10):1445. https://doi.org/10.14202/vetworld.2018.1445-1453
Torlak E, Sert D, Ulca P (2013) Efficacy of gaseous ozone against Salmonella and microbial population on dried oregano. Int J Food Microbiol 165(3):276–280. https://doi.org/10.1016/j.ijfoodmicro.2013.05.030
Rezende AJ, de Alencar ER, Ferreira MDA, Ferreira WFDS (2021) Control of Listeria monocytogenes in refrigerated ozonated water. Ozone-Sci Eng 44(3):281–290. https://doi.org/10.1080/01919512.2021.1912586
Rodrigues RS, Machado SG, de Carvalho AF, Nero LA (2021) Pseudomonas sp. as the causative agent of anomalous blue discoloration in Brazilian fresh soft cheese (Minas Frescal). Int Dairy J 117:105020. https://doi.org/10.1016/j.idairyj.2021.105020
Chierici M, Picozzi C, La Spina MG, Orsi C, Vigentini I, Zambrini V, Foschino R (2016) Strain diversity of Pseudomonas fluorescens group with potential blue pigment phenotype isolated from dairy products. J Food Prot 79(8):1430–1435. https://doi.org/10.4315/0362-028X.JFP-15-589
Swanson KMJ, Petran RL, Hanlin JH (2015) Culture methods for enumeration of microorganisms Compendium of methods for the microbiological examination of foods. Am Pub Health Assoc 4:53–62. https://doi.org/10.2105/MBEF.0222
Kang C, Xiang Q, Zhao D, Wang W, Niu L, Bai Y (2019) Inactivation of Pseudomonas deceptionensis CM2 on chicken breasts using plasma-activated water. J Food Sci Tech 56:4938–4945. https://doi.org/10.1007/s13197-019-03964-7
Machado SG, da Silva FL, Bazzolli DM, Heyndrickx M, Costa PMDA, Vanetti MCD (2015) Pseudomonas spp. and Serratia liquefaciens as predominant spoilers in cold raw milk. J Food Sci 80(8):1842–1849. https://doi.org/10.1111/1750-3841.12957
Asawanonda P, Taylor CR (1999) Wood’s light in dermatology. Int J Dermatol 38(11):801–807. https://doi.org/10.1046/j.1365-4362.1999.00794.x
Francis FJ (1975) The original of tan-1a/b. J Food Sci 40:412
Little A (1975) A research note on a tangent. J Food Sci 40(2):410
McLellan MR, Lind LR, Kime RW (1995) Hue angle determinations and statistical analysis for muiltquadrant hinter, L, a, b data. J Food Qual 18(3):235–240. https://doi.org/10.1111/j.1745-4557.1995.tb00377.x
Gardoni D, Vailati A, Canziani R (2012) Decay of ozone in water: a review. Ozone: Sci Eng 34(4):233–242. https://doi.org/10.1080/01919512.2012.686354
Christensen PA, Yonar T, Zakaria K (2013) The electrochemical generation of ozone: a review. Ozone: Sci Eng 35(3):149–167. https://doi.org/10.1080/01919512.2013.761564
Galdeano MC, Wilhelm AE, Goulart IB, Tonon RV, Freitas-Silva O, Germani R, Chávez DWH (2018) Effect of water temperature and pH on the concentration and time of ozone saturation. Braz J Food Technol 21:e2017156. https://doi.org/10.1590/1981-6723.15617
Shao L, Dong Y, Chen X, Xu X, Wang H (2020) Modeling the elimination of mature biofilms formed by Staphylococcus aureus and Salmonella spp Using combined ultrasound and disinfectants. Ult Sonoch 69:105269. https://doi.org/10.1016/j.ultsonch.2020.105269
Panebianco F, Rubiola S, Di Ciccio PA (2022) The use of ozone as an eco-friendly strategy against microbial biofilm in dairy manufacturing plants: a review. Microorganisms 10(1):162. https://doi.org/10.3390/microorganisms10010162
Khadre MA, Yousef AE, Kim JG (2001) Microbiological aspects of ozone applications in food: a review. J Food Sci 66(9):1242–1252. https://doi.org/10.1111/j.1365-2621.2001.tb15196.x
Souza SMO, de Alencar ER, Ribeiro JL, de Aguiar Ferreira M (2019) Inactivation of Escherichia coli O157:H7 by ozone in different substrates. Braz J Microbiol 50(1):247–253. https://doi.org/10.1007/s42770-018-0025-2
Meyer JM (2000) Pyoverdines: pigments, siderophores and potential taxonomic markers of fluorescent Pseudomonas species. Arch Microbiol 174(3):135–142. https://doi.org/10.1007/s002030000188
Castell‐Perez, ME, Moreira, RG (2004) Decontamination systems. Preharvest and postharvest food safety: contemporary issues and future directions, 337–348. https://doi.org/10.1002/9780470752579.ch25
Cullen PJ, Tiwari BK, O’Donnell CP, Muthukumarappan K (2009) Modelling approaches to ozone processing of liquid foods. Trends in Food Sci Technol 20(3–4):125–136. https://doi.org/10.1016/j.tifs.2009.01.049
Lee SK, Chon JW, Yun YK, Lee JC, Jo C, Song KY, Kim DH, Bae D, Kim H, Moon JS, Seo KH (2022) Properties of broiler breast meat with pale color and a new approach for evaluating meat freshness in poultry processing plants. Poult Sci 101(3):101627. https://doi.org/10.1016/j.psj.2021.101627
AMSA. American Meat Science Association (2012) AMSA meat color measurement guidelines. AMSA, Champaign. https://meatscience.org/docs/default-source/publications-resources/hottopics/2012_12_meat_clr_guide.pdf
Funding
The authors acknowledge the support from the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq, Grant Number: 405894/2021–0 and 406719/2023-3), the Research Support Foundation of the State of Minas Gerais (Fundação de Amparo à Pesquisa do Estado de Minas Gerais, FAPEMIG, Grant Number: APQ-00088–21 and APQ-01701–22), the Coordination for the Improvement of Higher Education Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível de Superior, CAPES), Funding Code 001, FARA, and Tertiary Education Trust Fund (TetFund).
Author information
Authors and Affiliations
Contributions
EAO, ERA, LRDF, and SGM conceived and designed the experiment. EAO, ERA, SGM, LRDF, MVAS, AF, and NASC performed the experiments and analyzed the data. EAO, ERA, LRDF, and SGM wrote the manuscript. EAO, ERA, LRDF, and SGM revised the manuscript. ERA, LRDF, and SGM provided the funding for the study.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Elaine Cristina Pereira de Martinis
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Okolo, E.A., de Alencar, E.R., Machado, S.G. et al. Ozonation for Pseudomonas paracarnis control: biofilm removal and preservation of chicken meat during refrigerated storage. Braz J Microbiol 54, 3051–3060 (2023). https://doi.org/10.1007/s42770-023-01157-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-023-01157-1