Abstract
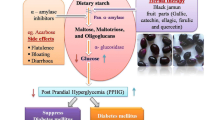
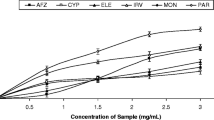
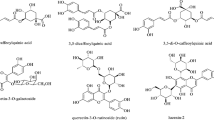
The use of medicinal therapies towards controlling hyperglycemia is a major step in managing diabetes and its complications. Of the various and numerous ways of lowering high blood sugar levels, one is by inhibiting key enzymes involved in carbohydrate metabolism. The purpose of this research is to compare the antioxidant potential, as well as enzyme inhibition activities, of aqueous extracts of Acacia nilotica seeds and pods. The antioxidant potential of the seed and pod were assessed using standard antioxidant protocols, including total phenolic and flavonoids content, DPPH scavenging ability, nitric oxide scavenging ability, and ferric reducing antioxidant power assay. The extracts’ inhibitory potential against α-amylase and α-glucosidase was also determined. Our findings showed that the aqueous extract of seed contains higher amounts of total phenolics and flavonoids than the pod. In all antioxidant parameters, the seed showed higher antioxidant activity than the pod. The extract of seed showed a higher (p < 0.05) inhibitory effect on α-glucosidase (IC50 = 0.08 µg/mL) than the pod (IC50 = 4.28 µg/mL), whereas the extract of pod showed higher inhibitory activity (IC50 = 5.49 µg/mL) than seed (IC50 = 6.99 µg/mL). A. nilotica Fruit extracts exhibited auspicious antioxidant as well as antidiabetic properties. Nevertheless, continuous research is required in order to separate and recognize the components that are actively responsible for the antioxidant and enzyme inhibitory effects.








Similar content being viewed by others
Data Availability
Data are available on reasonable request.
References
Khan MAB (2020) J.Epidemiology of type 2 diabetes - global burden of Disease and Forecasted Trends. J Epidemiol Glob Health 10:107–111. https://doi.org/10.2991/jegh.k.191028.001. MustafaH.
Ojo OA, Ojo AB, Ajiboye BO, Oyinloye BE, Imiere O, Adeyonu O (2017) Ameliorative potentials of Blighia sapida K.D. Koenig bark against pancreatic-cell dysfunction in alloxan-induced diabetic rats. J Complement Integr Med 14:20160145
Ojo OA, Afon AA, Ojo AB, Ajiboye BO, Oyinloye BE, Kappo AP (2018) Inhibitory Effects of Solvent-Partitioned Fractions of Two Nigerian Herbs (Spondias mombin Linn. and Mangifera indica L.) on α-Amylase and α-Glucosidase. Antioxidants 7(6), 73, https://doi.org/10.3390/antiox7060073.
Feunaing RT, Tamfu AN, Gbaweng AJY, Mekontso Magnibou L, Ntchapda F, Henoumont C, Dinica RM (2023) In Vitro evaluation of α-amylase and α-glucosidase inhibition of 2, 3-Epoxyprocyanidin C1 and other constituents from Pterocarpus erinaceus Poir. Molecules 28(1):126
Tamfu, A. N., Roland, N., Mfifen, A. M., Kucukaydin, S., Gaye, M., Botezatu, A. V.,… Dinica, R. M. (2022). Phenolic composition, antioxidant and enzyme inhibitory activities of Parkia biglobosa (Jacq.) Benth., Tithonia diversifolia (Hemsl) A. Gray, and Crossopteryx febrifuga (Afzel.) Benth. Arabian Journal of Chemistry, 15(4), 103675
Ojo OA, Amanze J, Oni AP, Grant S, Iyobhebhe M, Elebiyo TC, Rotimi D, Asogwa NT, Oyinloye BE, Ajiboye BE, Ojo AB (2022a) Antidiabetic activity of avocado seeds (Persea americana Mill.) In diabetic rats via activation of PI3K/AKT signaling pathway. Sci Rep 12:2919
Ojo OA, Oni AP, Grant S, Amanze J, Ojo AB, Taiwo OA, Maimako RF, Evbuomwan IO, Iyobhebhe M, Nwonuma CO, Osemwegie O, Agboola AO, Akintayo CO, Asogwa NT, Aljarba NH, Alkahtani S, Mostafa-Hedeab G, Batiha GE-S, Adeyemi OS (2022b) Antidiabetic activity of Elephant grass (Cenchrus purpureus (Schumach.) Morrone) via activation of PI3K/AkT signaling pathway, oxidative stress inhibition, and apoptosis in Wistar rats. Front Pharmacol 13:845196. https://doi.org/10.3389/fphar.2022.845196
Amadou I (2020) An overview on the importance of Acacia nilotica (L.) Willd. Ex Del.: a review. Asian J Res Agric For 5(3):12–18. https://doi.org/10.9734/ajraf/2020/v5i330085. ;SaléM.A
Maldini M, Montoro P, Al H, Mahalel UA, Oleszek W, Stochmal A, Piacente S (2011) Strong antioxidant phenolics from a nilotica: profiling by ESI-MS and qualitative, quantitative determination by LC-ESI-MS. J Pharm Biomed Anal 56:228–239
Pattnaik S, Mani S, Rajkumari J, Sudharshan SJ, Madhu D, Busi S (2017) Determination of antioxidant potential of Acacia nilotica leaf extract in oxidative stress response system of Saccharomyces cerevisiae. J Sci Food Agric 15:5247–5253
Muddathir AM, Mohieldin E, Mitsunaga T (2020) In vitro activities of Acacia nilotica (L.) Delile bark fractions against oral Bacteria,glucosyltransferase and as antioxidant. BMC Complement Med Ther 20:360
Hussein G, Miyashiro H, Nakamura N, Hattori M, Kawahata T, Otake T, Kakiuchi N, Shimotohno K (1999) Inhibitory effects of sudanese medicinal plant extracts on HIV-1 replication and HIV-1 protease. Phytother Res 13:31–36
Dafallah AA, Al-Mustafa Z (1996) Investigation of the anti-inflammatory activity of Acacia nilotica and Hibiscus sabdariffa. Am J Chin Medi 24:263–269
Ahmad M, Zaman F, Sharif T, Ch MZ (2008) Antidiabetic and hypolipidemic effects of aqueous methanolic extract of Acacia nilotica pods in alloxan-induced diabetic rabbits. Scandinavian J Lab Anim Sci 35(1):29–34
Omara EA, Nada SA, Farrag ARH, Sharaf WM, El-Toumy SA (2012) Therapeutic effect of Acacia nilotica pods extract on streptozotocin induced diabetic nephropathy in rat. Phytomedicine 19(12):1059–1067
Vadivel V, Biesalski HK (2012) Total phenolic content, in vitro antioxidant activity and type II diabetes relevant enzyme inhibition properties of methanolic extract of traditionally processed underutilized food legume, Acacia nilotica (L.) Willd ex. Delile. Int Food Res J 19(2):593–601
Rather LJ, Mohammad F (2015) Acacia nilotica (L.): a review of its traditional uses, phytochemistry, and pharmacology. Sustainable Chem Pharm 2:12–30
Kumar G, Singh NK, Srivastava M (2022) Comparative phytochemical investigation and antioxidant activity in different parts of Acacia nilotica seed. Indian J Pharm Sci 84(3):552–559
Oyetayo FL, Ojo OA (2017) Seeds inhibiting ferrous sulfate-induced oxidative stress in rat tissues. Oxidants Antioxi Med Sci 6(2):35–39Dennettia tripetala
Gyesi JN, Opoku R, Borquaye LS (2019) Chemical composition, total phenolic content, and antioxidant activities of the essential oils of the leaves and fruit pulp of Annona muricata L. (Soursop) from Ghana.Biochem Res Int1–9. https://doi.org/10.1155/2019/4164576
Phuyal N, Jha PK, Raturi PP, Rajbhandary STotal (2020) phenolic, flavonoid contents, and antioxidant activities of fruit, seed, and bark extracts of Zanthoxylum armatum DC. The Sci World J 2020, ID e8780704, 1–7
Kifle ZD, Yesuf JS, Atnafie SA (2020) Evaluation of in vitro and in vivo anti-diabetic, anti-hyperlipidemic and anti-oxidant activity of flower crude extract and solvent fractions of Hagenia abyssinica (Rosaceae). J Exp Pharmacol 12:151–167
Borquaye LS, Laryea MK, Gasu EN, Boateng MA, Baffour PK, Kyeremateng A, Doh G (2020) Anti-inflammatory and antioxidant activities of extracts of Reissantia indica,Cissus cornifolia and Grosseria vignei. Cogent Biol 6(1):1785755. https://doi.org/10.1080/23312025.2020.1785755
Kumari S, Phogat D, Sehrawat KD, Choudhary R, Rajput VD, Ahlawat J, Karunakaran R, Minkina T, Sehrawat AR (2021) The Effect of Ascophyllum nodosum Extract on the nutraceutical antioxidant potential of Vigna radiata sprout under salt stress. Plants 10:1216. https://doi.org/10.3390/plants10061216
Dibacto REK, Tchuente BRT, Nguedjo MW, Tientcheu YMT, Nyobe EC, Edoun FLE, Kamini MFG, Dibanda RF, Medoua GN (2021) Total Polyphenol and Flavonoid Content and Antioxidant Capacity of Some Varieties of Persea americana Peels Consumed in Cameroon. The Sci World J 2021, 8882594, https://doi.org/10.1155/2021/8882594
Mechchate H, Es-safi I, Louba A, Alqahtani AS, Nasr FA, Noman OM, Farooq M, Alharbi MS, Alqahtani A, Bari A, Bekkari H, Bousta D (2021) In Vitro Alpha-Amylase and Alpha-Glucosidase Inhibitory Activity and in vivo antidiabetic activity of Withania frutescens L. Foliar Extract. Molecules 26:293
Nguelefack TB, Fofie CK, Nguelefack-Mbuyo EP, Wuyt AK (2020) Multimodal alpha-glucosidase and alpha-amylase inhibition and antioxidant effect of the aqueous and methanol extracts from the trunk bark of Ceiba pentandra. Biomed Res Int 3063674. https://doi.org/10.1155/2020/3063674
Jame R (2018) Phytochemical and pharmacological uses of acacia nilotica-a review. Seeds 1:15–21
Chiang SH, Yang KM, Lai YC, Chen CW (2021) Evaluation of the in vitro biological activities of Banana flower and bract extracts and their bioactive compounds. Int J Food Properties 24(1):1–16. https://doi.org/10.1080/10942912.2020.1856134
Ojo OA, Oloyede OI, Olarewaju OI, Ojo AB (2013) Vitro antioxidant activity and estimation of total phenolic content in Ethyl acetate extract of Ocimum gratissimum. Pharmacol Online 3:37–44
Foyzun T, Mahmud AA, Ahammed MS, Manik MIN, Hasan MK, Islam KM, Lopa SS, Al-Amin MY, Biswas K, Afrin MR et al Polyphenolics with strong antioxidant activity from Acacia nilotica ameliorate some biochemical Signs of Arsenic-Induced neurotoxicity and oxidative stress in mice.Molecules 2022, 27,1037, doi: https://doi.org/10.3390/molecules27031037
Kifle ZD, Debeb SG, Belayneh YM (2021) InVitroα-Amylase and α-Glucosidase Inhibitory and Antioxidant Activities of the Crude Extract and Solvent Fractions of Hageniaabyssinica Leaves. Biomed Res Int 2021, 6652777, https://doi.org/10.1155/2021/6652777
Zia-Ul-Haq M, Ćavar S, Qayum M, Khan I, Ahmad S (2013) Chemical composition and antioxidant potential of Acacia leucophloea Roxb. Acta Bot Croatica 72(1):133–144
Ojo OA, Ajiboye BO, Olayide I, Fadaka A, Olasehinde OR (2016) Ethyl acetate fraction of bark of Bridelia ferruginea Benth. Inhibits carbohydrate hydrolyzing enzymes associated with type 2 diabetes (α-glucosidase and α-amylase). Adv Biores 7(3):126–133. https://doi.org/10.15515/abr.0976-4585.7.3.126133
Ajiboye BO, Ojo OA, Oyinloye BE, Akuboh O, Okesola MA, Idowu O, Kappo AP (2020) vitro Antioxid inhibitory activities polyphenolic-rich extracts Syzygium cumini (Linn) Skeels leaf two important enzymes relevant type II diabetes mellitus Pak J Pharm Sci 33(2):523–529
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
FLO and OAO conceptualized the study, ASO, VOO, and OAO performed the antioxidant and antidiabetic analysis. FLO is major supervisor of ASO and VOO research project. FLO and OAO participated in the technical writing of the manuscript. FLO and OAO conducted a critical review and editing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors wish to confirm that there is no competing interest related to this work.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ojo, O.A., Oyetayo, F.L., Oladipo, A.S. et al. Polyphenolic contents, free radical scavenging properties, and enzyme inhibitory activities of Acacia nilotica (L.) delile seed and pod extracts. Vegetos 37, 296–304 (2024). https://doi.org/10.1007/s42535-023-00599-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42535-023-00599-0