Abstract
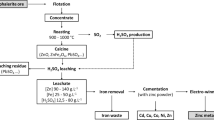
The present study investigates the influence of ferrous iron (as FeSO4) and ferric iron (as Fe2 (SO4)3), and pyrite (FeS2) on the ability of bacterial leaching of a high-grade sulfide Zn–Pb ore. In this regard, shake flask experiments were carried out at 5% (w/v) pulp density of the ore sample (having 40.7% Zn and 12.4% Pb initial metal content) using a consortium of mesophilic iron and sulfur-oxidizing acidophiles. A concentration of 0.04 mol/L of ferric iron in the leaching media was found to be optimum for zinc extraction without affecting growth of the microorganisms. Under this concentration, the dissolution of Zn, Pb, Cd, and As was found to be 57%, 0.2%, 0.03%, and 9.9% in 25 days. Using ferrous iron in the media, 0.16 mol/L of Fe2+ was found to be the optimum concentration for efficient bacterial growth and metal dissolution (54.6% Zn, 0.08% Pb, 0.03% Cd, and 10.2% As) from the sample in 25 days. On the other hand, using pyrite as the source of energy for bacterial growth, an initial 12-day lag period was observed when compared to the effect of ferrous iron in the media. Under the optimum concentration (test with 0.24 mol/L iron in the form of pyrite), the dissolution of Zn, Pb, Cd, and As was found to be 39.8%, 0.1%, 0.03%, and 10% in 25 days. The surface chemistry analysis indicated formation of a sulfur layer over the particle surface that hindered reagent diffusion and affected metal recovery through bioleaching.








Similar content being viewed by others
References
Ahmadi A, Mousavi SJ (2015) The influence of physicochemical parameters on the bioleaching of zinc sulfide concentrates using a mixed culture of moderately thermophilic microorganisms. Int J Miner Process 135:32–39
Akcil A (2004) Potential bioleaching developments towards commercial reality: Turkish metal mining’s future. Miner Eng 17:477–480
Akcil A, Deveci H (2010) Mineral biotechnology of sulphides. In: Jain S, Khan A, Rai MK (eds) Geomicrobiology. Science Publishers, Enfield, pp 101–137
Baba AA, Adekola FA, Atata RF, Ahmed RN, Panda S (2011) Bioleaching of Zn(II) and Pb(II) from Nigerian sphalerite and galena ores by mixed culture of acidophilic bacteria. Trans Nonferrous Metals Soc China 21:2535–2541
Bigham JM, Algur OF, Jones FS, Tuovinen OH (2013) Solid-phase controls on lead partitioning in laboratory bioleaching solutions. Hydrometallurgy 136:27–30
Cheng Y, Guo Z, Liu X, Yin H, Qiu G, Pan F, Liu H (2009) The bioleaching feasibility for Pb/Zn smelting slag and community characteristics of indigenous moderate-thermophilic bacteria. Bioresour Technol 100:2737–2740
Deveci H, Akcil A, Alp I (2004) Bioleaching of complex zinc sulphides using mesophilic and thermophilic bacteria: comparative importance of pH and iron. Hydrometallurgy 73:293–303
Dutrizac JE, Chen TT (1994) Reaction of galena in ferric sulphate-sulphuric acid media. In: Hydrometallurgy ’94. Springer, Dordrecht. doi https://doi.org/10.1007/978-94-011-1214-7_7
Gahan CS, Srichandan H, Kim D, Akcil A (2012) Biohydrometallurgy and biomineral processing technology, a review on its past, present and future. Res J Recent Sci 1:85–99
Ghassa S, Boruomand Z, Abdollahi H, Moradian M, Akcil A (2014) Bioleaching of high-grade Zn-Pb bearing ore by mixed moderate thermophilic microorganisms. Sep Purif Technol 136:241–249
Ghassa S, Boruomand Z, Abdollahi H, Moradian M, Akcil A (2015) Microbial dissolution of Zn-Pb sulfide minerals using mesophilic iron and sulfur-oxidizing acidophiles. Miner Process Extr Metall Rev: Int J 36(2):112–122
Habashi F (1999) A textbook of hydrometallurgy, second edn. Metallurgie Extractive Quebec, Quebec City
Halikia I, Voudouris N (2013) Investigation of zinc dissolution and cadmium precipitation rates in a Cd2+/Zn cementation system. Miner Process Ext Metall 114:95–108
Hawkes RB, Franzmann PD, Plumb J (2006) Moderate thermophiles including “Ferroplasma cupricumulans” sp. nov. dominate an industrial-scale chalcocite heap bioleaching operation. Hydrometallurgy 83:229–236
He Z, Gao F, Zhong H, Hu Y (2009) Effects of L-cysteine on Ni–Cu sulfide and marmatite bioleaching by Acidithiobacillus caldus. Bioresour Technol 100:1383–1387
He Z, Yang Y, Zhou S, Hu Y, Zhong H (2014) Effect of pyrite, elemental sulfur and ferrous ions on EPS production by metal sulfide bioleaching microbes. Trans Nonferrous Metals Soc China 24(4):1171–1178
Johnson DB (1995) Selective solid media for isolating and enumerating acidophilic bacteria. J Microbiol Methods 23:205–218
Lan Z, Hu Y, Qin W (2009) Effect of surfactant OPD on the bioleaching of marmatite. Miner Eng 22:10–13
Leahy MJ, Schwarz MP (2009) Modelling jarosite precipitation in isothermal chalcopyrite bioleaching columns. Hydrometallurgy 98:181–191
Liao MX, Deng TL (2004) Zinc and lead extraction from complex raw sulfides by sequential bioleaching and acidic brine leach. Miner Eng 17:17–22
Liu YG, Zhou M, Zeng GM, Wang X, Li X, Fan T, Xu WH (2008) Bioleaching of heavy metals from mine tailings by indigenous sulfur-oxidizing bacteria: effects of substrate concentration. Bioresour Technol 99:4124–4129
Manafi Z, Abdollahi H, Tuovinen OH (2013) Shake flask and column bioleaching of pyritic porphyry copper sulphide ore. Int J Miner Process 119:16–20
Mousavi SM, Jafari A, Yaghmaei S, Vossoughi M, Roostaazad R (2006) Bioleaching of low-grade sphalerite using a column reactor. Hydrometallurgy 82:75–82
Niu Z, Huang Q, Wang J, Yang Y, Xin B, Chen S (2015) Metallic ions catalysis for improving bioleaching yield of Zn and Mn from spent Zn-Mn batteries at high pulp density of 10%. J Hazard Mater 298:170–177
Olubambi PA (2009) Influence of microwave pretreatment on the bioleaching behavior of low-grade complex sulphide ores. Hydrometallurgy 95:159–165
Panda S, Pradhan N, Mohapatra UB, Panda SK, Rath SS, Rao DS, Nayak BD, Sukla LB, Mishra BK (2013) Bioleaching of copper from pre and post thermally activated low grade chalcopyrite contained ball mill spillage. Front Environ Sci Eng 7:281–293
Panda S, Akcil A, Pradhan N, Deveci H (2015) Current scenario of chalcopyrite bioleaching: a review on the recent advances to its heap leach technology. Bioresour Technol 196:694–706
Panda S, Akcil A, Mishra S, Erust C (2017) Synergistic effect of biogenic Fe3+ coupled to S° oxidation on simultaneous bioleaching of Cu, Co, Zn and As from hazardous pyrite ash waste. J Hazard Mater 325:59–70
Pogliani C, Donati E (2000) Immobilization of Thiobacillus ferrooxidans: importance of jarosite precipitation. Process Biochem 35:997–1004
Qin W, Liu K, Diao M, Wang J, Zhang Y, Yang C, Jiao F (2013) Oxidation of arsenite (As (III)) by ferric iron in the presence of pyrite and a mixed moderately thermophilic culture. Hydrometallurgy 137:53–59
Rawlings DE, Johnson DB (2006) Mesophilic, autotrophic bioleaching bacteria: description, physiology and role, Biomining. Springer, Berlin, pp 229–245
Rehman M, Anwar MA, Iqbal M, Akhtar K, Khalid AM, Ghauri MA (2009) Bioleaching of high grade Pb –Zn ore by mesophilic and moderately thermophilic iron and sulfur oxidizers. Hydrometallurgy 97:1–7
Rodríguez Y, Ballester A, Blázquez ML, González F, Munõz JA (2002) New information on the sphalerite bioleaching mechanism at low and high temperature. Hydrometallurgy 71:57–66
Rohwerder T, Gehrke T, Kinzler K, Sand W (2003) Bioleaching review part A: progress in bioleaching: fundamentals and mechanisms of bacterial metal sulfide oxidation. Appl Microbiol Biotechnol 63:239–248
Sand W, Gehrke T, Jozsa PG, Shippers A (2001) (Bio) chemistry of bacterial leaching-direct vs. indirect bioleaching. Hydrometallurgy 59:159–175
Shi S, Fang S (2004) Bioleaching of marmatite flotation concentrate by Acidithiobacillus ferrooxidans. Hydrometallurgy 75:1–10
Shi S, Fang S, Ni J (2005) Comparative study on the bioleaching of zinc sulphides. Process Biochem 41:438–446
Shi S, Fang Z, Ni J (2006) Comparative study on the bioleaching of zinc sulphides. Process Biochem 41:438–446
Silva G (2004a) Relative importance of diffusion and reaction control during the bacterial and ferric sulphate leaching of zinc sulphide. Hydrometallurgy 73:313–324
Silva G (2004b) Kinetics and mechanism of the bacterial and ferric sulphate oxidation of galena. Hydrometallurgy 75:99–110
Silverman MP, Lundgren DG (1959) Studies on the chemoautotrophic iron bacterium Ferrobacillus ferrooxidans. I An improved medium and harvesting procedure for securing high cell yields. J Bacteriol 77:642–647
Soleimani M, Hosseini S, Roostaazad R, Petersen J, Mousavi SM, Kazemi Vasiri A (2009) Microbial leaching of a low-grade sphalerite ore using a draft tube fluidized bed bioreactor. Hydrometallurgy 99:131–136
Souza AD, Pina PS, Leao VA (2007a) Bioleaching and chemical leaching as an integrated process in the zinc industry. Miner Eng 20:591–599
Souza AD, Pina PS, Leao VA, Silva CA, Siqueira PF (2007b) The leaching kinetics of a zinc sulphide concentrate in acid ferric sulphate. Hydrometallurgy 89:72–81
Wang J, Qiu G, Qin W, Zhang Y (2006) Microbial leaching of marmatite by Acidithiobacillusferrooxidans and Acidithiobacillusthiooxidans. Trans Nonferrous Metals Soc China 16:937–942
Watling HR (2006) The bioleaching of sulphide minerals with emphasis on copper sulphides - a review. Hydrometallurgy 84:81–108
Watling HR, Collinson DM, Fjastad S, Kaksonen AH, Li J, Morris C, Perrot FA, Rea SM, Shiers DW (2014) Column bioleaching of a polymetallic ore: effects of pH and temperature on metal extraction and microbial community structure. Miner Eng 58:90–99
Wei-min Z, Shi-fei GU (2007) Catalytic effect of active carbon on bioleaching of low-grade primary copper sulfide ores. Trans Nonferrous Metals Soc China 17:1123–1127
Xia L, Zeng J, Ding J, Yang Y, Zhang B, Liu J, Qiu G (2007) Comparison of three induced mutation methods for Acidithiobacillus caldus in processing sphalerite. Miner Eng 20:1323–1326
Xia L, Liu J, Xiao L, Zeng J (2008) Single and cooperative bioleaching of sphalerite by two kinds of bacteria Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans. Trans Nonferrous Metals Soc China 18:190–195
Ye M, Li G, Yan P, Ren J, Zheng L, Han D, Sun S, Huang S, Zhong Y (2017) Removal of metals from lead-zinc mine tailings using bioleaching and followed by sulfide precipitation. Chemosphere 185:1189–1196
Acknowledgments
We are grateful to Dr. Fariborz Gharib, Head of Applied Geological Research Center of Iran, (GRCIR) and Marzieh Moradian for providing facilities and technical assistance. Finally, we thank Prof. Olli H. Tuovinen for reviewing and comments on the manuscript.
Funding
This work was supported by the Geological Survey of Iran (GSI) under Grant No. 92-171-459.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ghassa, S., Abdollahi, H., Najafi, E. et al. A Comparative Assessment on the Effect of Different Supplemental Iron Sources on the Bio-dissolution of Zn, Pb, Cd, and As from a High-grade Zn–Pb Ore. Mining, Metallurgy & Exploration 36, 363–374 (2019). https://doi.org/10.1007/s42461-018-0001-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42461-018-0001-2