Abstract
Ethnopharmacological relevance
For decades, patients in East Africa have used herbal medicine as an alternative and affordable therapeutic option for the treatment of diseases such as Diabetes Mellitus (DM). As a result, the primary objective of this research was to thoroughly investigate the plants employed for treating DM in Uganda, Tanzania, Kenya, and the Democratic Republic of Congo and gather pertinent ethnopharmacological and ethnomedicinal knowledge that could be applied in the development of therapies for DM.
Aim of the study
The study is aimed at critically reviewing the phytochemistry, pharmacology, and toxicology of medicinal plants used for treating diabetes in East African countries, including Uganda, Tanzania, Kenya, and the Democratic Republic of Congo.
Method
A search for relevant articles was conducted on PubMed, ISI Web of Science, Open Theses, Google Scholar, Science Direct, and Scopus, and the identified articles were evaluated for quality, relevance, and taxonomical accuracy before undergoing review.
Results
The study identified 140 plant species used by local communities for the management of DM in East Africa, with decoction and infusion being the most common preparation methods. The leaves, root, and stem bark were the most frequently used parts. The in vitro and in vivo studies demonstrated the antidiabetic effect of medicinal plants such as Kigelia Africana, Hagenia abyssinica (Rosaceae), Physalis peruviana among other plants used for herbal treatment elicited via stimulation of insulin release, and gluconeogenesis. Phytochemicals present were mainly flavonoids, alkaloids, saponins, tannins, and glycosides.
Conclusion
The local communities in Uganda, Tanzania, Kenya and the Democratic Republic of Congo use herbal medicine for managing DM, but only a fraction of these species have scientific evidence. This necessitates additional research to establish the effectiveness, safety and potentially develop novel therapeutics for the management of DM.
Article Highlights
-
A study aimed to review the ethnomedicinal uses, pharmacology and phytochemistry of medicinal plants used for managing diabetes in East Africa.
-
The study found 140 plant species used for this purpose.
-
Decoction and infusion were the most common preparation methods.
-
Asteraceae and Fabaceae are the families with the most species of plants locally used for diabetes management.
-
Phytochemicals present in these plants include flavonoids, alkaloids, saponins, tannins, and glycosides.
-
Some plants were reported to have low toxicity on low and high dosage.
-
Further research is needed to fully evaluate the safety and effectiveness of these plants for managing diabetes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Traditional medicine has played a crucial role in healthcare for centuries, especially in developing regions [1]. However, much of this ancestral knowledge is at risk of disappearing as it is often passed down orally and not documented. The World Health Organization (WHO) recommends conducting ethnopharmacological studies to record this valuable folk knowledge and scientifically validate traditional medicinal claims [1]. This could lead to the development of improved medications. Around 80% of the global population depends on traditional medicine, primarily derived from plants, for their basic healthcare needs [2]. The use of medicinal plants as complementary and alternative medicines (CAM) presents opportunities in both developed and developing societies for managing various illnesses. Diabetes Mellitus (DM) has been a focus of research, with over 800 plant species displaying hypoglycemic activities, offering potential sources for discovering new antidiabetic molecules [3]. Recent data indicates that about 1.7 million people in the Democratic Republic of Congo (DRC) suffer from DM, ranking the country fourth in the top ten countries with diabetes cases in Africa [4]. Also, it is estimated that 478,000 people in Kenya have diabetes, with a treatment cost of USD 61 per person [5]. According to a recent STEP survey, Tanzania has the highest prevalence of DM in the East African region, with an estimated 9.1% among adults aged 25–64 years [6]. This represents a significant increase from the estimated 2.5% in 1984 [7, 8]. In East African member states of the International Diabetes Federation (IDF) in 2021 but South Sudan, there were a total of 128,048,100 adults, with a diabetes prevalence of 30.3% among them, resulting in a total of 6,850,500 cases of diabetes in adults [5] (Table 1). A study in 2022 by Sun et al. [5] reported the prevalence of diabetes among adults aged 20 to 79 over the world was estimated to reach 10.5% (536.6 million) in 2021 and 12.2% (783.2 million) in 2045. The cost of treating diabetes-related illnesses worldwide was estimated at 966 billion USD in 2021 and is expected to rise to 1,054 billion USD by 2045 [5]. There are 24 million diabetics in Africa and 537 million worldwide among the 48 nations that make up the IDF African area. It is anticipated that these figures would rise even higher by 2045[5, 8].
The metabolic syndrome known as DM has several aetiologies and is characterised by persistently high blood sugar levels with a perturbation of carbohydrate, fat and protein metabolism as a result of chronic and/ or relative insulin caused by an acquired or inherited deficiency in the synthesis of insulin by the pancreas, or by the ineffectiveness of the insulin synthesised [9]. The inability of the body to produce insulin is termed as Type 1 diabetes or insulin dependent DM while Type 2 DM or insulin independent DM is characterised by non-respondent of cells to the insulin synthesized by the pancreas. The increased blood sugar precipitate classical symptoms including polydipsia, polyphagia and polyuria. Diabetes has resulted in severe morbidity and death due to microvascular (nephropathy, neuropathy and retinopathy) and macrovascular (stroke, peripheral vascular and heart attack) complications [10, 11]. Type 1 and Type 2 diabetes are both incurable and chronic illnesses that are still among the most feared diseases in the world today. Patients with DM in public health facilities have a severe lack of therapy options, while their private counterparts have several expensive options. Many people with DM in Africa choose to employ spiritual and alternative therapies instead of going to the doctor because they believe in them [12]. Traditional remedies are employed as the major type of basic healthcare in sub-Saharan Africa and include everyday foods and herbs. Herbal medicine is still widely used because it is affordable, easily accessible, less expensive, culturally satisfying, and available in some regions where conventional treatment is not [9].
Despite the widespread use of herbal medicines, there is still a dearth of thorough documentation of these plants and other traditional treatments. The anti-diabetic properties of several plant extracts have been confirmed in numerous animal studies and randomised clinical trials across numerous nations. However, there is still need for documentation and scientific validation. As a result, there still exists a significant need to offer empirical data to legitimise local therapeutic usage. This review aims to investigate the plants utilized for the treatment and control of DM in some parts of East Africa, encompassing pertinent ethnopharmacological and ethnomedicinal data that could offer valuable insights for research on DM therapy.
2 Methods
2.1 Search strategy
From the start of the month of September until December 2022, various databases including PubMed, ISI Web of Science, Open Theses, Google Scholar, Science Direct, and Scopus were searched for relevant articles using the appropriate keywords. Several published papers that highlighted the historical use of plants to manage DM in Uganda, Kenya, Tanzania and DRC were selected for this study. These papers offer valuable insights into the traditional use of plants for treating DM. Whereas, experimental research carried out anywhere in the globe served as the foundation for the scientific evidence to support such usage. The selection of these few East African countries in Africa were selected for study given that they have rich biodiversity of a wide variety of medicinal plants with antidiabetic potentials, furthermore these regions prevalence of DM is high as compared to their counterparts with several studies suggesting so [6, 10, 13,14,15,16,17]. Based on the given criteria, the inclusion criteria for the study on antidiabetic plants include investigating plants traditionally used for treating diabetes, conducting various types of studies such as ex vivo, in vivo, in vitro, and clinical trials, and examining the antidiabetic potential of plant components and bioactive compounds with or without molecular targets. The exclusion criteria involve excluding research on phytochemicals, extracts, and fractions lacking biological activity, as well as computer simulations and thorough reviews of bioactive substances. Additionally, studies that did not examine interesting plants or those with insufficiently reported information are also excluded, along with in vivo studies not utilizing models of diabetes or prediabetes. Reference to Fig. 1 for the selection and screening of articles for study.
3 Results
3.1 A synopsis of the included research
Using the database, a total of 35 articles were found. These articles were checked for eligibility and evaluated for relevance according to inclusion criteria set. Hence, 17 were ultimately added (Fig. 1). The bulk of these papers (11 total) were ethnobotanical survey studies of plant usage in diabetes control. The remaining six publications dealt with in vivo studies on plant extracts or fractions that were antidiabetic.
3.2 Exploring traditional herbal remedies for diabetes
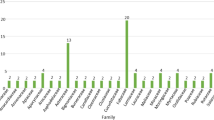
A total of 140 plants were reported to be used traditionally in the management of DM among the local communities of Uganda, Kenya, Tanzania and DRC, (Table 2). Among the reported plant 56.3% were from Kenya, 23.9% DRC, 15.49% and 4.2% from Tanzania. The 140-plant cut across fifty-five (55) different plant families, the most commonly reported family are Asteraceae and Fabaceae with 30.9% each, this was followed closely by Solanaceae 18.18%, Lumiaceae 14.54%, Apolynaceae 12.72%, Amaryllidaceae, Euphorbiaceae, Moraceae, Rutaceae and Urticaceae having 7.27% (Fig. 2).
3.2.1 Plants utilized for managing Diabetes mellitus in Uganda
Uganda's rich cultural heritage has a long-standing tradition of using medicinal plants to treat a range of health issues, including DM. Among the 22 plants reported for DM management in Uganda from this study, notable species include Ageratum conyzoides, Albizia chinensis, Aloe vera var, and Vernonia amygdalina [15, 18, 22, 23], among others (see Table 2). These indigenous plants have a well-established reputation for their efficacy in DM treatment, with deep roots in Ugandan folklore and traditional healing practices. Traditional medicine in Uganda makes use of specific parts of these plants, primarily focusing on the fruits, leaves, and roots [18]. Fruits are central to the traditional methods of managing DM remedies in Uganda. Bitter melon, scientifically known as Momordica charantia, is famed for its bitter fruits, attributed to their potential blood sugar-lowering properties, it is consumed as tea, juice or in cooking. Some individuals in Uganda juice bitter melon and consume it as a beverage, they usually combine it with other fruits or adding a bit of lemon to help mask the bitterness [15, 19]. Many parts of Uganda serve it with a groundnut (peanut) sauce, which adds a creamy and nutty flavour to the dish serve, this serve alongside staples like posho (maize meal) or rice [19]. Anona muricata, is another standout, with its fruits being a key component of DM remedies. The fresh soursop fruit is commonly consumed by either scooping out the sweet, pleasantly flavoured flesh or by juicing it. Anona muricata leaf tea is another popular approach, with dried leaves steeped in hot water to create herbal tea believed to offer antidiabetic benefits when consumed regularly [20, 21]. Some individuals opt for soursop leaf extracts or tinctures, which provide a more concentrated form of the active compounds and can be diluted in water for controlled consumption. Additionally, Anona muricata is incorporated into various traditional recipes, including smoothies, desserts, stews, and curries, as part of the local approach to utilizing soursop for its potential antidiabetic properties [22]. Leaves are another vital plant part utilised in traditional medicine. Aloe vera var, known for its succulent leaves, is processed into a gel for a variety of therapeutic applications, including potential DM management, many Ugandans use it as dietary supplement, and its gel, extracted from the inner leaf, is consumed in various ways, such as mixing it with water or other beverages [22]. Ageratum conyzoides, referred to locally as “billy goat weed,” is also prized for its leaves with anti-diabetic potential. People in Uganda often used to create herbal infusions or teas by drying the leaves and steeping them in hot water, with the belief that this herbal tea possesses medicinal properties for managing diabetes [22]. Additionally, the leaves are processed to extract compounds that are thought to help regulate blood sugar levels, and these extracts are consumed in controlled amounts. Furthermore, Ageratum conyzoides leaves are incorporated into traditional recipes, such as soups and stews, aiming to introduce their potential health benefits into the local diet [23]. Vernonia amygdalina, known as “bitter leaf” is commonly integrated into local cuisine and traditional remedies among the locals of Uganda. They frequently employed it in the preparation of vegetable soups and stews, where the leaves are meticulously washed, finely chopped, and included in these dishes, contributing to their flavour and nutritional content. Additionally, some locals dried the leaves to produce bitter leaf tea, an herbal infusion believed to offer potential health advantages, including potential antidiabetic effects. Some individuals opt for bitter leaf extracts, derived from the leaves, which can be consumed in controlled quantities due to the belief in their ability to help regulate blood sugar levels. Bitter leaf's versatility also extends to its incorporation into various traditional recipes, often combined with other locally available vegetables and ingredients, as an integral component of local dishes[23, 24]. Additionally, the roots of Ficus saussureana are incorporated into traditional formulations, attributed to their anti-diabetic effects [18, 22]. In Uganda, medicinal herbs are traditionally prepared using two primary methods decoction and infusion [25]. These plants offer a diverse array of options for those seeking alternative remedies for diabetes. The choice of specific plant parts and traditional preparation methods reflects the accumulated knowledge and experience of Ugandan traditional healers over generations. The integration of this traditional knowledge with modern scientific research holds promise for advancing diabetes treatment options, offering the potential for effective, evidence-based therapies.
3.2.2 Plants utilized for managing Diabetes mellitus in Kenya
In Kenya, a substantial repertoire of seventy-nine (79) locally employed plant species has been documented for the treatment of DM Table 2. These plants are sourced for their fruits, roots, leaves, and stems, signifying a diverse range of plant parts utilised in traditional remedies. Some notable species include Acacia nilotica, Allium sativum (Garlic), Azadirachta indica (Neem), and Bidens pilosa, among others. In various regions of Kenya, Acacia nilotica, is traditionally utilized as a potential antidiabetic remedy. Its various parts, including the bark, leaves, and pods, are employed in distinct ways. The bark is dried and steeped in hot water to create herbal infusions or teas, believed to have antidiabetic properties [14, 26]. The leaves are dried, crushed, and used in the preparation of teas or extracts for potential medicinal benefits. Additionally, the pods are processed and consumed in various forms, such as boiling, grinding into powder, or integration into traditional recipes, all depending on local customs and practices. Allium sativum, is a versatile and widely used plant with both culinary and potential medicinal applications. As a staple ingredient in Kenyan cuisine, garlic adds flavour and enhances the taste of a diverse array of dishes, spanning from soups and stews to vegetable and meat-based meals [27]. Beyond its culinary role, garlic features prominently in traditional herbal remedies, where it is believed to offer potential antidiabetic and cardiovascular benefits [28]. In these remedies, some individuals either consume garlic cloves directly or incorporate it into herbal preparations. Furthermore, garlic is used to create garlic-infused oils or vinegars, which not only enhance culinary creations but are also consumed as health tonics. Azadirachta indica is employed for potential antidiabetic purposes through various methods in Kenya. Neem leaves are widely used in Kenyan traditional remedies and are either chewed or prepared as a tea or infusion, with neem leaf tea believed to possess antidiabetic properties and often consumed regularly [16, 27]. Neem extracts, encompassing neem leaf extracts and neem oil, are harnessed for their potential medicinal benefits and consumed in controlled amounts. Neem bark and twigs find application in some traditional practices, notably for dental hygiene, a significant consideration given the impact of diabetes on oral health. In Kenya, Bidens pilosa, is employed in various forms for potential antidiabetic purposes. Herbal infusions serve as the most common method of preparation which are commonly made by drying the leaves and steeping them in hot water to create an herbal tea, believed to offer potential antidiabetic benefits and consumed regularly [14, 29]. Most of the herbal preparations include the leaves, stems, or the entire plant often in the form of decoctions or tinctures, are consumed. Additionally, Bidens pilosa is incorporated into traditional Kenyan recipes and local dishes example Sukuma wiki, where it is cooked and forms part of the traditional diet. The comprehensive list also encompasses plant species like Kigelia africana, Olea europaea and Prunus africana, illustrating the varied plant-based approaches to DM treatment in Kenyan traditional medicine [14]. In Kenya, the traditional use of medicinal plants for managing DM is deeply ingrained in the country's cultural heritage. The extensive variety of plant species and plant parts employed for DM treatment reflects the rich indigenous knowledge of the local communities. The use of decoction and infusion as primary preparation methods underlines the versatility and adaptability of traditional remedies to suit the unique characteristics of each plant species [30]. As modern medicine explores the potential benefits of these plants, their integration with scientific research offers a promising avenue for advancing DM treatment options, harnessing the power of nature to tackle a growing global health concern.
3.2.3 Plants utilized for managing Diabetes mellitus in Tanzania
Tanzania, like its East African counterparts, has a rich tradition of utilising medicinal plants for various health applications, including the management of DM. A selection of plant species has garnered significant attention and study in the realm of traditional medicine, with some of them being extensively researched. In this study 10 of these plants were mentioned notable among these are Bersama abyssinica, Dioscorea praehensilis Benth, Hagenia abyssinica, Hypoxis obtusa, and Moringa oleifera Table 2. In Tanzania, Dioscorea praehensilis, particularly its roots and tubers, is esteemed for its potential antidiabetic properties and is incorporated into Tanzanian culinary and traditional practices in various ways. The tubers of Dioscorea praehensilis are commonly cooked or boiled and integrated into Tanzanian meals, offering a delicious and nutritious addition while being believed to possess potential antidiabetic benefits [31]. Furthermore, herbal infusions or teas are crafted from tubers or other parts of the plant, esteemed for their perceived medicinal properties and their consumption for potential antidiabetic effects. Moreover, Dioscorea praehensilis finds its place in traditional Tanzanian recipes, where it becomes an integral ingredient in soups, stews, or side dishes, often harmoniously combined with other components to form a “Mboga na Mtori wa Boga”a local Tanzanian dish. These practices reflect local beliefs in the antidiabetic potential of Dioscorea praehensilis [6, 17]. In traditional Tanzanian antidiabetic management, Moringa oleifera, often referred to as the drumstick tree or horseradish tree, plays a significant role through various preparations and administration methods. Moringa leaf powder, derived from drying and finely grinding the leaves, is frequently used as a dietary supplement and is incorporated into a variety of dishes, such as soups, stews, and smoothies [32]. It is esteemed for its nutritional support and potential in regulating blood sugar levels. Herbal infusions made from dried Moringa leaves are another common practice, where steeping these leaves in hot water yields an herbal tea believed to possess antidiabetic properties. Additionally, Moringa oil, typically recognized for its skincare benefits, is occasionally utilized in culinary preparations or as part of the diet, contributing to overall health. Moringa seeds are consumed directly or included in recipes, believed to offer potential health advantages, including their role in diabetes management [33]. Similarly, in Tanzanian traditional medicine, various parts of Bersama abyssinica, including the bark and leaves, have been utilized for their potential antidiabetic properties [34]. Preparations are done by making herbal infusions or teas this is achieved through drying the bark and steeping it in hot water, resulting in an herbal tea believed to possess antidiabetic benefits and consumed regularly. Additionally, the leaves of the tree are prepared by decoctions or tinctures and consumed in controlled amounts in management of DM [17, 32]. Moreover, Bersama abyssinica may be integrated into various recipes and preparations, often combined with other locally available herbs and plants, as part of traditional practices aimed at managing DM. These plants, among others, have been explored for their potential anti-diabetic properties. In recent years, there has been a growing interest in the scientific validation of the antidiabetic properties of these traditional remedies. A study conducted by Zekeya et al. [34] provided confirmation of the antidiabetic properties of Bersama abyssinica. This research adds a layer of credibility to the traditional knowledge held by Tanzanian communities. It underscores the potential of traditional herbs to contribute to the management of diabetes and highlights the importance of integrating traditional practices with modern scientific research. Decoction and infusion have established themselves as the most commonly employed methods among Tanzanian communities.
3.2.4 Plants utilized for managing Diabetes mellitus in DRC
The Democratic Republic of the Congo (DRC) is home to a diverse array of medicinal plants with potential antidiabetic properties. In this study 38 of these plants were mentioned, these plants, including Allium cepa L. (Onion), Azanza garckeana, Bidens pilosa L., Brassica juncea (L) and Cassia occidentalis L. (Table 2), represent a rich pharmacopoeia deeply rooted in the traditional healing practices of the region [2, 10]. Alongside these, species like Bersama abyssinica (Meliathacea), Ficus exasperata Vahl, and Vernonia amygdalina [34,35,36] have been explored for their antidiabetic potential. While the study by Zekeya et al. [34] supports the antidiabetic properties of Bersama abyssinica, further scientific research is needed to fully comprehend the efficacy and safety of these plants in diabetes management. The preparation of remedies commonly involves traditional methods such as decoction and infusion, which play a vital role in extracting bioactive compounds from plant parts, making these plants valuable assets in the fight against diabetes in DRC. For example, in DRC, traditional practices involving Allium cepa L. as a potential antidiabetic remedy take various forms. Raw onion consumption is a common method, with raw onions frequently featured in salads and various dishes, as it is believed to contribute to the regulation of blood sugar levels [13]. Additionally, onion tea, created by steeping slices or small pieces of onion in hot water, is a popular choice. This aromatic infusion is enjoyed as a beverage and is thought to harbour potential antidiabetic effects. Furthermore, herbal extracts are crafted from onions through the process of maceration or crushing of fresh onion pieces, followed by soaking them in a solvent, usually water. These herbal extracts are administered in controlled amounts and are believed to harness antidiabetic properties, reflecting the rich tradition of using natural remedies like onions in the management of diabetes in the region [2]. Furthermore, in DRC, Azanza garckeana, known as the “African Ordeal Tree” or “Mbugu,” is traditionally administered in various ways to harness its potential antidiabetic benefits. One method involves the consumption of Azanza garckeana fruit, which is incorporated into dishes in raw form or as an ingredient. This practice is rooted in the belief in the fruit's potential to aid in diabetes management [10]. Additionally, Azanza garckeana leaves and other plant parts are used to create herbal infusions and teas, achieved by drying the plant material and steeping it in hot water. The resulting tea is enjoyed as a beverage, with the hope of delivering antidiabetic effects. Herbal extracts from Azanza garckeana are also crafted by macerating or crushing plant material and soaking it in water, offering a controlled means of consumption and further reinforcing the plant's reputation for its perceived antidiabetic properties [2].
Different plant parts were reported to be used in the preparation of herbal extract with leaf forming (50.7%) as the most commonly used part, followed by roots (9.15%) and stem bark (7.74%). The other plant parts used are fruits, whole plants, seeds, tubers, tubules, corm, aerial parts, bulbs and cloves. The decoction was the most desired conventional dosage form, based on the information that was provided. The basic method of preparing the dose was hot water maceration or decoction. For the duration of therapy, a quantity of medicine necessary for a daily dose is typically made each day. Other methods of preparation include eating the whole plant part, infusion, tea or tincture, grinding into powder form and poultice (Table 2). It's interesting to note that plants used to treat traditional methods of managing DM have also been found to be effective in treating other health issues, including erectile dysfunction, allergies, yellow fever, worms, infertility, urinary tract infections, hypertension, stomach-aches, ulcers, malaria, blood purification, stroke, and eye problems [6, 15, 30]. There are several complications of DM that also include many of these illnesses.
3.3 Authentication
3.3.1 In vivo studies of medicinal plants
Of the 17 articles retrieved from data base 8 articles reported in vivo investigation using animal models. Analysis indicated high-fat diet and low-dose streptozotocin (STZ) was the most preferred substance for induction of DM in animals. Different routes of administration and doses were used. Other models used include Alloxan monohydride, glucose-loaded, STZ-nicotinamide [37]. Investigations showed that the plant extract has distinct mechanisms of action for managing DM and reducing blood glucose levels. Some of these mechanisms include, but are not limited to: decreasing total cholesterol (TC), triglycerides (TG), low density lipoprotein cholesterol (LDL-c) and very-low density lipoprotein cholesterol (VLDL-c), stimulating glycogenesis in the liver and/or inhibition of glucagon secretion, promoting the regeneration of b-cells of the islets of Langerhans, ameliorating oxidative stress, and facilitating glucose-induced insulin release [9, 13, 38]. Furthermore, different extracts were used for the preparation of doses of which ethanolic extract was the most used, other extract used include ethyl acetate, methanolic extract, aqueous and crude extract. All the plants investigated had a glucose-lowering effect at different doses of extracts and fractions. The eight (8) plant species traditionally used for DM in Uganda, Kenya, Tanzania and DRC as per this study are; Kigelia africana, Hagenia abyssinica (Rosaceae), Physalis peruviana, Chenopodium ambrosioides, Mangifera indica L. Lonchocarpus eriocalyx, Urtica massaica Mildr. Schkuhria pinnata, Launaea cornuta, Rotheca myricoides (Hochst.), Maerua decumbens (Brongn.) [1, 38,39,40, 44, 52] which have been reported to have in vivo antidiabetic effect (Table 3). For instance, Kigelia africana fruit extract caused significant improvements in blood glucose levels, haematological parameters, and kidney and liver health, suggesting its potential protective effect against diabetes-related complications [1]. Similarly, Hagenia abyssinica leaf extract induced hypoglycaemic, antihyperglycemic, and anti-dyslipidaemic effects in Streptozotocin-Induced Diabetic mice, contributing to the lowering of blood glucose levels and improvement of dyslipidaemia [38]. Additionally, the leaf extract of Physalis peruviana L. effectively lowered blood glucose levels without causing toxicity in mice [39]. Chenopodium ambrosioides, on the other hand, exhibited a significant hypoglycaemic effect in mice [40]. Lonchocarpus eriocalyx extracts displayed significant anti-inflammatory, anti-nociceptive, and antioxidant effects without acute oral toxicity, highlighting their potential as safe and effective natural alternatives for treating inflammatory and oxidative stress-related diseases [41]. The ethyl acetate extract of Launaea cornuta showed hypoglycaemic efficacy in diabetic mice, while the aqueous extract had adverse effects on body weight and haematological parameters, indicating the potential of the ethyl acetate extract as a safe and effective option for managing diabetes [42]. Freeze-dried extracts of Rotheca myricoides exhibited hypoglycaemic, hypolipidemic, and hypo insulinemic effects in a rat model of type 2 diabetes, suggesting the potential as a natural treatment for diabetes and the ability to modulate peroxisome proliferator-activated gamma receptor and reduce hepatic triglycerides [43]. Maerua decumbens methanolic root extract demonstrated antidiabetic activity without signs of toxicity, including decreased fasting blood glucose levels, reduced liver malondialdehyde levels, and cytoprotective effects on the liver and pancreas in diabetic rats [9]. The assessment of Mangifera indica leaves extract, both alone and in combination with gliclazide, in diabetic rats showed significant reductions in fasting blood glucose levels [44]. The collective findings of these studies highlight the promising effects of various plant extracts on different aspects of diabetes, including blood glucose levels and associated complications.
3.3.2 Phytochemicals identified
A number of phytochemicals were identified to be present in all the plants reported to possess antidiabetic activities. The most commonly reported phytochemicals from this review include; tannins, flavonoids, saponin glycosides, phenolic acids, alkaloids, anthraquinones, alkaloids sterols triterpenoids and glycoalkaloids (Table 2). Fagbohon et al. [1] reported some phytochemicals (Table 4) from the n-hexane fruit extract of K. africana to possess antidiabetic potentials, they identified phytochemicals were docked against AMP-activated protein kinase (AMPK) and mitochondrial glycerophosphate dehydrogenase (mGPDH) with metformin a reference standard. Benzene-1,2,3,5-tetramethyl- was reported to have a higher binding affinity of − 6.4 kcal/mol and can activate mitochondrial glycerophosphate dehydrogenase better than metformin with binding affinity of − 6.3 kcal/mol. Several studies have shown that certain phytochemicals can help in the management of diabetes by improving insulin sensitivity, reducing inflammation, and lowering blood glucose levels [3, 45,46,47,48]. For example, flavonoids, such as quercetin and kaempferol, have been shown to improve insulin sensitivity and reduce inflammation, which can help in the management of diabetes [49]. Other phytochemicals, such as resveratrol and curcumin, have been shown to improve glucose metabolism and reduce blood glucose levels [50]. GC–MS analysis of some selected plants reveal some compounds having antidiabetic potentials (Table 4).
3.3.3 In vitro studies
Several studies conducted on extracts derived from Kigelia africana, Hagenia abyssinica, Physalis peruviana, Chenopodium ambrosioides, Mangifera indica L., Lonchocarpus eriocalyx, Urtica massaica Mildr., Schkuhria pinnata, Launaea cornuta, Rotheca myricoides (Hochst.), and Maerua decumbens (Brongn.) have provided further evidence supporting their efficacy in managing diabetes [38, 39, 51]. For instance, Fagbohun et al., [1] identified new compounds in fruit extracts of Kigelia africana and found that the hexane and ethyl acetate fractions showed the highest antioxidant activities. The hexane fraction also exhibited the lowest IC50 value against α-amylase. The methanol extract and its fractions of Chenopodium ambrosioides showed moderate inhibitory activity on α-glucosidase, an enzyme involved in carbohydrate digestion [52, 53]. Mangifera Indica leaf (MIL) extract demonstrated an inhibitory effect on alpha-amylase activity, reducing it by 51.4% at a concentration of 200 µg/mL. This suggests the extract can potentially slow down the breakdown of complex carbohydrates by inhibiting amylase, leading to a slower release of glucose [54]. The antioxidant activity of the aqueous root extract of Launaea cornuta using the ferric-reducing antioxidant power (FRAP) and 2,2-diphenyl-2-pycrylhydrazyl (DPPH) radical scavenging test methods demonstrated significant antioxidant effects, as evidenced by its ability to scavenge DPPH radicals and its FRAP activity [55]. This suggests that the extract has the potential to protect cells from oxidative damage caused by free radicals, which are known to contribute to various health conditions, including DM, inflammation and other chronic diseases. The findings of these studies suggest that the identified plant extracts possess various mechanisms of action that contribute to their antidiabetic properties. One such mechanism involves the inhibition of alpha-amylase and alpha-glucosidase enzymes. Alpha-amylase is an enzyme responsible for the breakdown of complex carbohydrates into simpler sugars, while alpha-glucosidase aids in the final digestion of carbohydrates by breaking down disaccharides into absorbable monosaccharides. Inhibiting the activity of these enzymes can help reduce the rate of carbohydrate breakdown and subsequent glucose absorption, leading to improved glycemic control [56]. Moreover, the investigated plant extracts have demonstrated the ability to stimulate insulin secretion. Insulin plays a crucial role in regulating blood glucose levels by facilitating the uptake of glucose into cells and promoting its storage as glycogen [40]. The stimulation of insulin secretion by these extracts suggests a potential therapeutic benefit in individuals with impaired insulin production or function.
Furthermore, the studies have revealed that the examined plant extracts exhibit the capacity to reduce blood glucose levels. Hyperglycaemia, or high blood glucose levels, is a hallmark characteristic of diabetes and is associated with various complications [57]. The ability of these extracts to lower blood glucose levels indicates their potential in managing this metabolic disorder. It is important to note that these in vitro studies provide valuable preliminary evidence of the antidiabetic effects of the identified plant extracts. However, further research is necessary to validate these findings and elucidate the underlying mechanisms in in vivo models and, eventually, human trials. The in vitro studies conducted on the extracts have demonstrated promising antidiabetic effects. The inhibition of alpha-amylase and alpha-glucosidase enzymes, stimulation of insulin secretion, and reduction of blood glucose levels suggest the potential of these plant extracts in managing diabetes. Further research is warranted to validate these findings, explore the underlying mechanisms, and identify the bioactive components responsible for these beneficial effects.
3.3.4 Toxicological studies
Toxicological studies on the listed plants have been limited, and most of the available studies have focused on acute toxicity in animal models. These studies have generally shown that these plants have low toxicity and are relatively safe when consumed in moderate doses. However, it is essential to note that these findings may not necessarily translate to human consumption and further toxicological studies, including chronic toxicity and genotoxicity, are needed.
For instance, in a study on Kigelia africana, the aqueous and methanol extracts were found to have no observable toxic effects in rats at doses of up to 5000 mg/kg [58]. Kasali et al. [39] investigated the acute and subchronic toxicities of Kigelia africana bark extract in rats. The study reported that the extract did not induce significant acute toxicity or adverse effects in the treated animals. However, more comprehensive toxicological studies are necessary to evaluate its chronic toxicity and long-term effects. Similarly, a study on Hagenia abyssinica showed no significant acute toxicity in mice at doses up to 5000 mg/kg [38]. In another study on Mangifera indica, the ethanol extract was found to be safe in rats at doses of up to 2000 mg/kg [59]. Similarly, Kifle et al. [38] examined the acute and subchronic toxicities of Hagenia abyssinica leaf extracts in mice did not show toxicity at a dose of 2 g/kg in mice. Therefore, the LD50 of the extract is greater than 2 g/kg. The findings revealed no signs of acute toxicity, but the subchronic toxicity study indicated mild haematological alterations in the treated animals. Regarding Physalis peruviana, Kasali et al., [39] conducted an acute oral toxicity study on the methanol extract of the plant's leaves with an LD50 of 2000 mg/kg. The results suggested that the extract had a high safety margin, as no mortality or significant adverse effects were observed in the treated animals.
In contrast, some studies have reported potential toxicity concerns with some of these plants. For instance, a study on Chenopodium ambrosioides found that the essential oil from this plant had acute toxic effects on mice at doses higher than 2000 mg/kg [52]. Similarly, a study on Lonchocarpus eriocalyx found that the aqueous extract had toxic effects on rats at doses of 4000 mg/kg and above [41]. While some of the listed plants have shown low toxicity in animal studies, further toxicological studies are needed to determine their safety profile, particularly with long-term exposure and in human consumption.
4 Discussion
In the pursuit of alternative remedies for managing diabetes mellitus, traditional medicinal plants have exhibited promise. Our research findings unveil that local communities in Uganda, Kenya, DRC, and Tanzania employ a minimum of 140 different plant species to handle DM. While some of these species have undergone extensive investigation of their phytochemicals for DM management with encouraging results, other species necessitate additional scientific validation. This revelation may open avenues for discovering valuable drug lead compounds by concentrating research efforts on traditionally utilized plant species.
In vitro studies demonstrate that the investigated species exert their effects on multiple cellular and molecular targets in the liver, skeletal muscles, gastrointestinal tract, pancreas, and adipocytes. These plants inhibit enzymes such as α-amylase, α-glucosidase, sucrase, and maltase, regenerate pancreatic β-cells, and diminish fat accumulation in adipocytes by activating signalling pathways like PPAR-α and PPAR-δ [60]. Additional molecular actions involve inhibiting the nuclear inflammatory signalling pathway through the modulation of nuclear factor-erythroid 2-related factor Nrf2/NFκB signalling and suppressing pro-inflammatory cytokines such as IL-1a, IL-1b, and tumour necrosis factor (TNF)-α [33, 61, 62].
In recent years, it has been acknowledged that the pathophysiology of DM involves multiple cellular and molecular abnormalities. Consequently, drugs with the potential to target multiple sites have become the focal point for effective DM management [46]. Nevertheless, while in vitro studies on plant preparations have reported dose ranges up to 1000 μg/mL and much higher doses up to 1500 mg/kg, there is no standardized cut-off dose for either extract in either in vitro or in vivo studies [3, 47]. Recognizing that the tested dose should be sufficient to yield a meaningful pharmacological response and realistically convertible to a clinical dose, the large doses tested may pose challenges in translating them to human equivalent doses.
For the pharmacological assessment of herbal products, Heinrich et al. [63] proposed a plant extract dosage ranging from 100 to 200 mg/kg, significantly lower for its pure constituent. Despite the potential benefits of these traditional medicinal herbs, numerous methodological flaws have been identified in research analyses. Notably, certain studies documented considerable variation in effect size between standard medication and test substances. Methodological issues such as insufficient dosage optimization, improper preparation techniques, and inadequate ethnomedical data, including a lack of a comprehensive description of the identification process and voucher specimen, have been noted. Our study underscores the substantial potential of traditional medicinal herbs as complementary treatments for DM management. However, to sufficiently affirm the safety and efficacy of these plants, additional scientific evidence is imperative.
5 Conclusion
In conclusion, the traditional use of 140 plant species by local communities in Uganda, Kenya, DRC, and Tanzania for the management of DM is a valuable source of knowledge that deserves further exploration. While some of these plant species have been investigated scientifically, many have not, highlighting the need for additional research in this area. Despite the lack of scientific evidence for some of these plants, toxicological studies have shown that many of them are safe for human consumption at both low and high doses. Therefore, it is essential to conduct more in vitro, in vivo, and phytochemical investigations to fully understand the therapeutic potential of these plant species for DM management. Such research could help identify novel and effective plant-derived therapies that could be incorporated into existing diabetes management strategies. Furthermore, engaging with local communities to better understand their traditional knowledge could help in the discovery of new plant species with promising anti-diabetic properties.
Overall, bridging the gap between traditional knowledge and modern scientific investigation is crucial to improving diabetes management in these regions and beyond. The potential of these plant species as a source of new anti-diabetic drugs cannot be overstated, and future research in this area should be a priority.
Data availability
The data that support the findings of this study are available from the corresponding author, RSD, upon reasonable request.
Abbreviations
- DM:
-
Diabetes mellitus
- IDF:
-
International diabetes federation
- IL-1:
-
Interleukin-1
- LDL-c:
-
Low-density lipoprotein cholesterol
- LMICs:
-
Low- to middle-income countries
- NCDs:
-
Non-communicable diseases
- NF-kB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- PI3K:
-
Phosphoinositide 3-kinase
- PKC-β:
-
Protein kinase C β isoform
- PPAR-α:
-
Peroxisome proliferator-activated receptor alpha
- PPAR-δ:
-
Peroxisome proliferator-activated receptor gamma
- PPG:
-
Postprandial glucose
- PTP1B:
-
Protein tyrosine phosphatase 1 B
- SOD:
-
Superoxide dismutase
- SSA:
-
Sub-Saharan Africa
- STZ:
-
Streptozotocin
- T2DM:
-
Type 2 diabetes mellitus
- TC:
-
Total cholesterol
- TGs:
-
Triglycerides
- TNF-α:
-
Tumor necrosis factor- α
- VLDL-c:
-
Very low-density lipoprotein cholesterol
- WHO:
-
World health organization
References
Fagbohun OF, Oriyomi OV, Adekola MB, Msagati TAM. Biochemical applications of Kigelia africana (Lam.) Benth. fruit extracts in diabetes mellitus. Comp Clin Path. 2020;29:1251–64.
Amuri B, Maseho M, Simbi L, Okusa P, Duez P, Byanga K. Hypoglycemic and antihyperglycemic activities of nine medicinal herbs used as antidiabetic in the region of Lubumbashi (DR Congo). Phytother Res. 2017;31(7):1029–33.
Ahangarpour A, Sayahi M, Sayahi M. The antidiabetic and antioxidant properties of some phenolic phytochemicals: a review study. Diabetes Metab Syndr. 2019;13(1):854–7.
Kasali FM, et al. Antidiabetic medicinal plants used in Democratic Republic of Congo: a critical review of ethnopharmacology and bioactivity data. Front Pharmacol. 2021;12: 757090.
Sun H, et al. IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183: 109119. https://doi.org/10.1016/j.diabres.2021.109119.
Peter EL, et al. Pharmacological reflection of plants traditionally used to manage diabetes mellitus in Tanzania. J Ethnopharmacol. 2021;269:113715. https://doi.org/10.1016/j.jep.2020.113715.
WH Organization, Classification of diabetes mellitus. 2019
Williams R, et al. Global and regional estimates and projections of diabetes-related health expenditure: results from the International Diabetes Federation Diabetes Atlas. Diabetes Res Clin Pract. 2020;162: 108072.
Kiptisia RT, Maiyoh GK, Macharia BN, Tuei VC. Safety evaluation and antihyperglycaemic effect of root extract of Maerua decumbens (Brongn.) DeWolf in Wistar rats. Sci Afr. 2020;8: e00379. https://doi.org/10.1016/j.sciaf.2020.e00379.
Katemo M, et al. Ethnopharmacological survey of plants used against diabetes in Kisangani city (DR Congo). J Ethnopharmacol. 2012;144(1):39–43. https://doi.org/10.1016/j.jep.2012.08.022.
Ota A, Ulrih NP. An overview of herbal products and secondary metabolites used for management of type two diabetes. Front Pharmacol. 2017;8:1–14. https://doi.org/10.3389/fphar.2017.00436.
Schultz F, Anywar G, Wack B, Quave CL, Garbe LA. Ethnobotanical study of selected medicinal plants traditionally used in the rural Greater Mpigi region of Uganda. J Ethnopharmacol. 2020;256:112742. https://doi.org/10.1016/j.jep.2020.112742.
Amuri B, Maseho M, Simbi L, Okusa P, Duez P, Byanga K. Hypoglycemic and antihyperglycemic activities of nine medicinal herbs used as antidiabetic in the region of Lubumbashi (DR Congo). Phytother Res. 2017;31(7):1029–33. https://doi.org/10.1002/ptr.5814.
Chege IN, Okalebo FA, Guantai AN, Karanja S, Derese S. Management of type 2 diabetes mellitus by traditional medicine practitioners in Kenya-key informant interviews. Pan Afr Med J. 2015;22:1–8. https://doi.org/10.11604/pamj.2015.22.90.6485.
Gang R, Matsabisa M, Okello D, Kang Y. Ethnomedicine and ethnopharmacology of medicinal plants used in the treatment of diabetes mellitus in Uganda. Appl Biol Chem. 2023;66(1):39.
Kamau LN, Mbaabu MP, Mbaria JM, Karuri GP, Kiama SG. Knowledge and demand for medicinal plants used in the treatment and management of diabetes in Nyeri County, Kenya. J Ethnopharmacol. 2016;189:218–29. https://doi.org/10.1016/j.jep.2016.05.021.
Lunyera J, et al. Traditional medicine practices among community members with diabetes mellitus in Northern Tanzania: an ethnomedical survey. BMC Complement Altern Med. 2016. https://doi.org/10.1186/s12906-016-1262-2.
Ssenyange CW, Namulindwa A, Oyik B, Ssebuliba J. Plants used to manage type ii diabetes mellitus in selected districts of central Uganda. Afr Health Sci. 2015;15(2):496–502. https://doi.org/10.4314/ahs.v15i2.24.
Nkambo W, Anyama NG, Onegi B. Ein vivo hypoglycemic effect of methanolic fruit extract of Momordica charantia L. Afr Health Sci. 2013;13(4):933–9. https://doi.org/10.4314/ahs.v13i4.11.
Okullo JBL, et al. Journal of medicinal plants studies ethno-medicinal uses of selected indigenous fruit trees from the lake victoria basin districts in Uganda. J Med Stud. 2014;2(1):78–88.
Gumisiriza H, Birungi G, Olet EA, Sesaazi CD. Medicinal plant species used by local communities around Queen Elizabeth national park, Maramagambo central forest reserve and Ihimbo central forest reserve, South western Uganda. J Ethnopharmacol. 2019;239:111926. https://doi.org/10.1016/j.jep.2019.111926.
Obakiro SB, et al. Ethnobotanical study of plants used in management of diabetes mellitus in Eastern Uganda. Phytomedicine Plus. 2023;3(4): 100486.
Ssenyange CW, Namulindwa A, Oyik B, Ssebuliba J. Plants used to manage type II diabetes mellitus in selected districts of central Uganda. Afr Health Sci. 2015;15(2):496–502.
Kiguba R, Ononge S, Karamagi C, Bird SM. Herbal medicine use and linked suspected adverse drug reactions in a prospective cohort of Ugandan inpatients. BMC Complement Altern Med. 2016;16(1):1–8. https://doi.org/10.1186/s12906-016-1125-x.
Tugume P, Nyakoojo C. Ethno-pharmacological survey of herbal remedies used in the treatment of paediatric diseases in Buhunga parish, Rukungiri District, Uganda. BMC Complement Altern Med. 2019;19:1–10.
Kamau LN, Mbaabu MP, Mbaria JM, Karuri GP, Kiama SG. Knowledge and demand for medicinal plants used in the treatment and management of diabetes in Nyeri County, Kenya. J Ethnopharmacol. 2016;189:218–29.
Keter LK, Mutiso PC. Ethnobotanical studies of medicinal plants used by Traditional Health Practitioners in the management of diabetes in Lower Eastern Province, Kenya. J Ethnopharmacol. 2012;139(1):74–80. https://doi.org/10.1016/j.jep.2011.10.014.
Hengame Y, Development of a Food Supplement for Diabetes Type II Management With Enhanced Antidiabetic and Antioxidant Properties From Selected Local Foods. University of nairobi, 2019
Kiage-Mokua BN, et al. Effect of extracts from selected Kenyan plants on traits of metabolic syndrom in Wistar rats fed a high-fat high fructose diet. Trop J Pharm Res. 2020;19(10):2137–46.
Mogwasi R, Zor S, Kariuki DK, Getenga MZ, Nischwitz V. Sequential extraction as novel approach to compare 12 medicinal plants from Kenya regarding their potential to release chromium, manganese, copper, and zinc. Biol Trace Elem Res. 2018;182(2):407–22. https://doi.org/10.1007/s12011-017-1083-2.
Vinceti B, Eyzaguirre P, Johns T, The nutritional role of forest plant foods for rural communities. In: Human Health and Forests, Routledge, 2012, pp 63–96
Moshi MJ, Otieno DF, Weisheit A. Ethnomedicine of the Kagera Region, north western Tanzania. Part 3: plants used in traditional medicine in Kikuku village, Muleba District. J Ethnobiol Ethnomed. 2012;8(1):1–11.
Bao Y, Xiao J, Weng Z, Lu X, Shen X, Wang F. A phenolic glycoside from Moringa oleifera Lam. improves the carbohydrate and lipid metabolisms through AMPK in db/db mice. Food Chem. 2020;311: 125948.
Zekeya N, et al. Potential of natural phenolic antioxidant compounds from Bersama abyssinica (Meliathacea) for treatment of chronic diseases. Saudi J Biol Sci. 2022;29(6): 103273.
Soji-Omoniwa O, Oloyede HOB, Antidiabetic activity of bioactive-rich fractions of ficus exasperata vahl leaf in fructose and streptozotocin-induced diabetic rats. 2020
Ugbogu EA, et al. The phytochemistry, ethnobotanical, and pharmacological potentials of the medicinal plant-Vernonia amygdalina L.(bitter Leaf). Clin Complement Med Pharmacol. 2021;1(1):100006.
Tian S, Li X, Wang Y, Lu Y. The protective effect of sulforaphane on type II diabetes induced by high-fat diet and low-dosage streptozotocin. Food Sci Nutr. 2021;9(2):747–56.
Kifle ZD, Yesuf JS, Atnafie SA. Evaluation of in vitro and in vivo anti-diabetic, anti-hyperlipidemic and anti-oxidant activity of flower crude extract and solvent fractions of hagenia abyssinica (rosaceae). J Exp Pharmacol. 2020;12:151–67.
Kasali FM, et al. Phytochemical constituents found in Physalis peruviana L. leaf extracts and their ability to inhibit alpha-glucosidase and scavenge DPPH free radicals in vitro. Trends Phytochem Res. 2022;6(1):3–10.
Song Y, et al. Metabolites identification of chemical constituents from the eggplant (Solanum melongena L.) Calyx in Rats by UPLC/ESI/qTOF-MS Analysis and their cytotoxic activities. Front Pharmacol. 2021;12: 655008.
Moriasi GA, Ireri AM, Nelson EM, Ngugi MP. “In vivo anti-inflammatory, anti-nociceptive, and in vitro antioxidant efficacy, and acute oral toxicity effects of the aqueous and methanolic stem bark extracts of Lonchocarpus eriocalyx (Harms.). Heliyon. 2021;7(5):07145.
Karau MG, Njagi E, Machocho AK, Wangai LN, Kamau PN. Efficacy and safety assessment of Launaea cornuta extracts in the management of diabetes mellitus. Ame J Med Med Sci. 2014;4(1):1–7.
Chege BM, Waweru MP, Frederick B, Nyaga NM. The freeze-dried extracts of Rotheca myricoides (Hochst.) Steane & Mabb possess hypoglycemic, hypolipidemic and hypoinsulinemic on type 2 diabetes rat model. J Ethnopharmacol. 2019;244: 112077. https://doi.org/10.1016/j.jep.2019.112077.
Madhuri AS, Mohanvelu R. Evaluation of antidiabetic activity of aqueous extract of Mangifera indica leaves in alloxan induced diabetic rats. Biomed Pharmacol J. 2017;10(2):1029–35.
Rath P, et al. A critical review on role of available synthetic drugs and phytochemicals in insulin resistance treatment by targeting PTP1B. Appl Biochem Biotechnol. 2022;194(10):4683–701.
Kong M, et al. Anti-inflammatory phytochemicals for the treatment of diabetes and its complications: lessons learned and future promise. Biomed Pharmacother. 2021;133: 110975.
Gandhi GR, et al. Citrus flavonoids as promising phytochemicals targeting diabetes and related complications: a systematic review of in vitro and in vivo studies. Nutrients. 2020;12(10):2907.
Kasali FM, Kadima JN, Tusiimire J, Agaba AG. Hypoglycemic, antihyperglycemic, and toxic effects of Physalis peruviana L. Aqueous and Methanolic Leaf Extracts in Wistar Rats. J Exp Pharmacol. 2022;14:185–93.
Sok Yen F, et al. Hypoglycemic effects of plant flavonoids: a review. Evidence-Based Complementary and Alternative Medicine. 2021;2021:1–12.
Faal M, Manouchehri H, Changizi R, Bootorabi F, Khorramizadeh MR. Assessment of resveratrol on diabetes of zebrafish (Danio rerio). J Diabetes Metab Disord. 2022;21(1):823–33.
Njogu SM, Arika WM, Machocho AK, Ngeranwa JJN, Njagi ENM. In Vivo Hypoglycemic Effect of Kigelia africana (Lam): studies with alloxan-induced diabetic mice. J Evid Based Integr Med. 2018;23:1–10. https://doi.org/10.1177/2515690X18768727.
Ali N, et al. GC/MS analysis, anti-leishmanial and relaxant activity of essential oil of Chenopodium ambrosioides (L.) from Malakand region. Pak J Pharm Sci. 2021;34(2):577–83.
Kasali FM, et al. GC-FID and GC-MS profiling and in vitro antidiabetic and antioxidant activities of Chenopodium ambrosioides L. Natural Resources for Human Health. 2023;3(2):219–27.
Ngo D-H, Ngo D-N, Vo TTN, Vo TS. Mechanism of action of Mangifera indica leaves for anti-diabetic activity. Sci Pharm. 2019;87(2):13.
Akimat EK, Omwenga GI, Moriasi GA, Ngugi MP. Antioxidant, anti-inflammatory, acute oral toxicity, and qualitative phytochemistry of the aqueous root extract of launaea cornuta (hochst. Ex oliv. & hiern.). J Evid Based Integr Med. 2021;26:2515690X211064585.
Makuttan S, Fernandes J, Anti-diabetic potential of fruit extracts of Flacourtia indica (Burm. F.) Merr-An in-vitro study. Indian Journal of Natural Products and Resources (IJNPR)[Formerly Natural Product Radiance (NPR)], vol. 13, no. 4, pp. 491–496, 2022
Sharma P, Hajam YA, Kumar R, Rai S. Complementary and alternative medicine for the treatment of diabetes and associated complications: a review on therapeutic role of polyphenols. Phytomedicine Plus. 2022;2(1): 100188.
Imran IZ, Elusiyan CA, Agbedahunsi JM, Omisore NO. Bioactivity-directed evaluation of fruit of Kigelia africana (Lam.) Benth. Used in treatment of malaria in Iwo, Nigeria. J Ethnopharmacol. 2021;268: 113680.
Villas-Boas GR, Paes MM, Gubert P, Oesterreich SA. Evaluation of the toxic potential of the aqueous extract from Mangifera indica Linn. (Anacardiaceae) in rats submitted to experimental models of acute and subacute oral toxicity. J Ethnopharmacol. 2021;275: 114100.
Peter EL, et al. Pharmacological reflection of plants traditionally used to manage diabetes mellitus in Tanzania. J Ethnopharmacol. 2021;269: 113715.
Oyenihi AB, Langa SOP, Mukaratirwa S, Masola B. Effects of Centella asiatica on skeletal muscle structure and key enzymes of glucose and glycogen metabolism in type 2 diabetic rats. Biomed Pharmacother. 2019;112: 108715.
Tekula S, Khurana A, Anchi P, Godugu C. Withaferin-A attenuates multiple low doses of Streptozotocin (MLD-STZ) induced type 1 diabetes. Biomed Pharmacother. 2018;106:1428–40.
Heinrich M, et al. Best practice in research–overcoming common challenges in phytopharmacological research. J Ethnopharmacol. 2020;246: 112230.
Keter LK, Mutiso PC. Ethnobotanical studies of medicinal plants used by Traditional Health Practitioners in the management of diabetes in Lower Eastern Province, Kenya. J Ethnopharmacol. 2012;139(1):74–80.
Kamboj A, Saluja AK. Ageratum conyzoides L.: A review on its phytochemical and pharmacological profile. Int J Green Pharm (IJGP). 2008;2(2):59.
Kokila K, Priyadharshini SD, Sujatha V. Phytopharmacological properties of Albizia species: a review. Int J Pharm Pharm Sci. 2013;5(3):70–3.
Katemo M, et al. Ethnopharmacological survey of plants used against diabetes in Kisangani city (DR Congo). J Ethnopharmacol. 2012;144(1):39–43.
Nalimu F, Oloro J, Peter EL, Ogwang PE. Acute and sub-acute oral toxicity of aqueous whole leaf and green rind extracts of Aloe vera in Wistar rats. BMC Complement Med Ther. 2022;22(1):16.
Mogwasi R, Zor S, Kariuki DK, Getenga MZ, Nischwitz V. Sequential extraction as novel approach to compare 12 medicinal plants from Kenya regarding their potential to release chromium, manganese, copper, and zinc. Biol Trace Elem Res. 2018;182:407–22.
Seke F, Manhivi VE, Shoko T, Slabbert RM, Sultanbawa Y, Sivakumar D. Effect of freeze drying and simulated gastrointestinal digestion on phenolic metabolites and antioxidant property of the Natal plum (Carissa macrocarpa). Foods. 2021;10(6):1420.
Schultz F, Anywar G, Wack B, Quave CL, Garbe L-A. Ethnobotanical study of selected medicinal plants traditionally used in the rural Greater Mpigi region of Uganda. J Ethnopharmacol. 2020;256: 112742.
Njogu SM, Arika WM, Machocho AK, Ngeranwa JJN, Njagi ENM. In vivo hypoglycemic effect of Kigelia africana (Lam): studies with alloxan-induced diabetic mice. J Evid Based Integr Med. 2018;23:2515690X18768727.
Kiptisia RT, Maiyoh GK, Macharia BN, Tuei VC. Safety evaluation and antihyperglycaemic effect of root extract of Maerua decumbens (Brongn.) DeWolf in Wistar rats. Sci Afr. 2020;8: e00379.
Krawinkel MB, Ludwig C, Swai ME, Yang R, Chun KP, Habicht SD. Bitter gourd reduces elevated fasting plasma glucose levels in an intervention study among prediabetics in Tanzania. J Ethnopharmacol. 2018;216:1–7.
Oketch-Rabah HA. Mondia whitei, a medicinal plant from Africa with aphrodisiac and antidepressant properties: a review. J Diet Suppl. 2012;9(4):272–84.
Chege BM, Waweru MP, Frederick B, Nyaga NM. The freeze-dried extracts of Rotheca myricoides (Hochst.) Steane & Mabb possess hypoglycemic, hypolipidemic and hypoinsulinemic on type 2 diabetes rat model. J Ethnopharmacol. 2019;244: 112077.
Ayyanar M, Subash-Babu P. Syzygium cumini (L.) Skeels: a review of its phytochemical constituents and traditional uses. Asian Pac J Trop Biomed. 2012;2:240–6.
Okello D, Kang Y. Ethnopharmacological potentials of Warburgia ugandensis on antimicrobial activities. Chin J Integr Med. 2021;27(8):633–40.
Saleem M, Tanvir M, Akhtar MF, Iqbal M, Saleem A. Antidiabetic potential of Mangifera indica L. cv. anwar ratol leaves: medicinal application of food wastes. Medicina (Lithuania). 2019;55(7):353. https://doi.org/10.3390/medicina55070353.
Sharaibi OJ, Omolokun KT, Oluwa OK, Ogbe AA, Adebayo AO. Comparative studies of chemical compositions and antioxidant potentials of Dysphania ambrosioides L (Amaranthaceae) and Hybanthus enneaspermus (L) F. Muell (Violaceae). J Pharmacogn Phytochem. 2023;12(1):573–81.
Acknowledgements
This work is part of a Ph.D. thesis of Reuben Samson Dangana. Kampala International University (KIU) supported the Ph.D. training under the staff development scholarship scheme. The authors wish to thank the management of KIU for the scholarship.
Funding
No specific grant from funding agencies in the public, commercial, or not-for-profit sectors was received for the work.
Author information
Authors and Affiliations
Contributions
Reuben Samson Dangana: Conceptualization, Writing – original draft, Investigation, Visualization, Data analysis. Ibrahim Babangida Abubakar: Writing – original draft, Supervision, Writing– review & editing. Tijjani Salihu Shinkafi: Writing – review & editing, Validation, Supervision. Patrick Maduabuchi Aja: Writing – review & editing, Supervision. Sarah Onyenibe Nwozo: Writing–review & editing, Supervision. All data were generated in-house, and no paper mill was used. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethical approval is not required for the work.
Competing interest
No conflict of interest from the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dangana, R.S., Abubakar, I.B., Shinkafi, T.S. et al. Ethnobotany, pharmacology and phytochemistry of medicinal plants used for management of Diabetes mellitus in Uganda, Kenya, Tanzania and the Democratic Republic of Congo. Discov Appl Sci 6, 312 (2024). https://doi.org/10.1007/s42452-024-05970-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-024-05970-7