Abstract
Chemical exudates from some plants to the environment have been reported as causative agents of adverse effect on growth of neighboring plants and thus affecting normal growth in their natural environment. The mode of action of Chemical exudates on plants is involves a large number of biochemical reactions. This research aimed to investigate the effect of foliar application of different levels of two bacterial species and five aqueous plant extracts on the yield and yield components of Oryza sativa L. and Echinochloa crus-galli L. This research was conducted as a factorial based on randomized complete block design (RCBD) with three factors with three replications during the 2017–2018 crop year. The main factor included aqueous extracts of Sorghum (Sorghum bicolor L. Moench), Broccoli (Brassica oleracea var. Italica), Nettle (Urtica dioica), Eucalyptus (Eucalyptus globulus Labill), Elderberry (Sambucus nigra L.), and Streptomyces sp-albos containing thaxtomin, Xanthomonas campestris, and control (without aqueous plant extracts and surfactant). The sub-factor includes foliar application treatment with different levels of zero (control), 5, 10, and 15% of extracts, and the third experimental factor included Oryza sativa L. and E. crus-galli L. The results indicated the plant height, leaf area index (LAI), grain yield, biological yield, chlorophyll a, chlorophyll b, total chlorophyll, and grain protein content decreased, and the grain starch content increased with increasing the extract concentration; the highest effect was obtained at 15% of extracts. The lowest value of leaf area index (3.13) were found in higher concentration of X. campestris followed by Elderberry extract (5%, 10% and 15%) and Nettle (10%) extracts. It was also found that using extract reduced yield characteristics and components of Oryza sativa L., E. crus-galli L., and Elderberry extract showed the highest negative effect. According to the results, Oryza sativa L. had higher plant height, LAI, grain yield, biological yield, harvest index, chlorophyll, grain protein content, and grain starch content than E. crus-galli L. In all extractions, control treatment had the highest number of panicles plant−1 in both plants (rice and E. crus-galli L., 260.1 and 117.1 mg g−1 FW). The results suggested that various extracts, especially Nettle, and 15% of Elderberry can control E. crus-galli L. at the germination stage under field conditions.
Article highlights
-
The Elderberry and Nettle extracts had the highest impact on rice and suruf.
-
The negative impact of extract reduced yield and yield components of rice.
-
The reduction in grain weigh correlated to in leaves and plant height.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Oryza sativa L., belongs to the Oryza genus and Gramineae family [1] and is the most important crop after wheat (Triticum aestivum L.), which provides about 40% of the nutritional energy of more than half of the world’s population [2]. Echinochloa crus-galli L., E. crus-galli L., is an annual summer plant of the Gramineae family that seeds propagate. Echinochloa crus-galli L. grows mostly in warm temperate regions and is moisture-friendly. This plant is among the most important weeds grown in rice fields in Iran and other countries. Echinochloa crus-galli L. is a C4 plant and, therefore, has rapid growth and tillering and quickly dominates rice fields [3]. Echinochloa crus-galli L. is usually found in rice fields, imposes new biological limitations on rice productivity, and has adverse effects on rice grain growth and yield [4]. Widespread use of chemical herbicides and severe dependence on them has caused problems such as weed resistance to herbicides and adverse effects on human health and the environment [5]. Therefore, it is essential to find a way that is not only safe for the humans and environment but also cost-effective for weed control.
Using the allelopathic property of some plants to control weeds is of particular importance [6]. Allelopathy is a phenomenon, by which a plant produces allelochemicals and can influence the growth of itself and other plants. These substances can have negative or positive effects on the germination, vegetative, and reproductive stages of other plants [7]. Allelochemicals are released into the environment by shoot leaching and volatilization, root exudation, and plant litter decomposition [8]. Known effects of allelochemicals include anatomical abnormalities, decreased absorption, germination, and bud growth, chlorosis, and necrosis. The inhibitory effect of these substances depends on the aqueous extract concentration of the plant [9].
In their study, Dastres et al. [10] found that allelochemicals of aqueous extracts of pagoda tree and creeping jenny reduced plant height and wheat dry weight. Hatami Hampa et al. [11] investigated the allopathic effect of aqueous extracts of Sorghum and Russian knapweed on the seedling growth and activity of antioxidant enzymes in the wheat, sugar beet, common lambsquarter, and redroot pigweed, and reported that Sorghum and Russian knapweed extracts reduced seedling growth and increased soluble carbohydrates, proline concentration, and catalase activity in the studied plants compared to the control. Sadat Asilan et al. [12] found that a number of rice cultivars had a stimulatory effect, and some had an inhibitory effect on E. crus-galli L. growth. Increasing the concentration of rice cultivar extract increased the inhibition and stimulation of E. crus-galli L. growth [13,14,15]. A comprehensive list of allelochemicals and secondary metabolites present in different plant parts of sorghum (roots, stems, foliage and panicle) is documented by Hussain et al. [16]. Broccoli is well known for their allelopathic potential as most of them endogenously produce potent allelochemicals such as glucosinolates, allyl isothiocyanates, and brassinosteroids [17]. Extracts of fresh nettle (Urtica dioica L.) showed greater harmful effects than those from dried plants and generally reduced the germination of cereals from control values of 90–93 to 50–93% and decreased root length in barley from 426 to 455 to 162–397 mm [18].
Weeds cause many problems in different land uses. The growing number of herbicide-resistant weeds and prohibiting the use of pesticides and herbicides have provided a strong incentive to develop new weed control strategies. Using bacteria, fungi, and viruses to achieve this goal has received increasing attention over the last 3 decades. The benefits of this strategy include reducing environmental impacts, decreasing development costs compared to conventional herbicides, and identifying new herbicide mechanisms [19]. It has been recommended to use bacteria for the biological control of weeds due to the faster growth of biological antiseptics, relatively simple propagation requirements, and suitability for genetic modification through mutagenesis or gene transfer [20]. Biological control, as a general term, refers to introducing organisms into an ecosystem in order to control one or more undesirable species [21]. Xanthomonas campestris is a Gram-negative rod-shaped bacterium with a mobile polar flagellum belonging to the Pseudomonas family and seed-borne plant pathogens [22, 23]. The Streptomyces genus belongs to Actinomystals and is commonly found in soil. This genus is considered a potential source for producing a variety of antibiotics and other secondary metabolites due to its ability to secrete large amounts of extracellular enzymes [24]. Chen et al. [25] determined the role of endophytic bacteria of C. equietifolia, involved in synthesis of allelochemicals. The results of Abbas et al. [26] indicated that dry matter of broad-leaved dock, wild oat, little seed canary grass, and common lambs’ quarter were reduced by strains of rhizobacteria and some strains suppressed the growth of wheat, some strains increased its dry matter and some strains remained ineffective.
Research on allelopathy and plant pathogens can be among the basic ways to control weeds in the sustainable agricultural system. Allelopathic weeds may cause positive, negative, or even neutral effects on crops by releasing chemical compounds, mainly secondary metabolites, through various ways such as root exudation, litter decomposition, leaching, and volatilization. Therefore, considering the importance of this issue in terms of protection and sustainable production, this study tried to take a step towards sustainable agriculture by implementing the above plan, reducing the application of pesticides and herbicides, and establishing non-chemical methods to improve the quality and quantity of crops by controlling weeds and moving from conventional agriculture to sustainable agriculture.
2 Materials and methods
This factorial experiment was carried out using randomized complete block design with three replications an arable land located in one of the villages of northern Iran, Amol, Mazandaran province (52° 25′ E, 36° 25′ N, 110 m above sea level), in the 2017–2018 crop year. There were three experimental factors:
-
1.
Aqueous extracts in seven levels: Sorghum (Sorghum bicolor L. Moench), Broccoli (Brassica oleracea var. Italica), Nettle (U. dioica), Eucalyptus (Eucalyptus globulus Labill), Elderberry (Sambucus nigra L.), and Streptomyces sp-albos containing thaxtomin, Xanthomonas campestris, and control (without aqueous plant extracts and surfactant).
-
2.
Concentration of extraction included: 0 (control), 5%, 10%, and 15% of pure extraction.
-
3.
Two plants included the land-race rice (cv. Fereydunkenar) and E. crus-galli L.
The bacteria were purchased as lyophilized in coordination with Islamic Azad University and the Center for Scientific and Industrial Research of Iran. Plates containing solid medium and inoculated bacteria were incubated at 30 °C for 24–48 h for X. campestris and 1–2 weeks for Streptomyces to provide sufficient time for the bacteria to grow and produce their metabolites. After this period, the sterilized seeds of the studied plants were placed on the grown plates on opposite sides. A plate containing the medium without a bacterial culture with seeds should always be considered as the control.
According to the conditions, the leaves and stems of Sorghum, Broccoli, Nettle, Eucalyptus, and Elderberry were placed in an oven at 70 °C for 48 h and, then, ground by a mill to prepare the aqueous extracts with sterile distilled water. The aqueous extracts of the intended plants were obtained by pouring 50 g of the powdered aerial parts of each plant into an Erlenmeyer flask and adding 500 ml of distilled water, which had reached 70–80 °C. The flask’s neck was then covered, and it was placed in a bain-marie at 60 °C. After 24 h, all the mixtures inside the flask were squeezed, and the obtained extract was filtered using filter paper and a funnel to get a stock solution with a concentration of 0.1 g ml−1 (100%). From the stock solution, four concentrations; 5%, 10%, and 15%, were then made on the basis of percentage. The extract was then sprayed on 4–7-leaf plants and kept in a petri dish as a liquid.
Land preparation operations, including plowing, harrowing, and leveling were carried out in a desirable manner before planting, terracing the nursery, and sowing rice and E. crus-galli L. seeds in the nursery. rice and E. crus-galli L. seeds were brought from Amol rice Research Institute and disinfected with Vitavax fungicide before sowing. Urea fertilizer (150 kg ha−1) and phosphorus fertilizer (90 kg P2O5 ha−1 in the form of single superphosphate) were applied in three stages along with transplanting the seedlings to the main plot, tillering, and clustering. After transplanting the seedlings, the field was kept submerged for 2 weeks; since then, until 2 weeks before the harvest, intermittent irrigation was carried out. All the preparation operations performed for experimental plots and the studied rice and E. crus-galli L. seedlings were similar. The seeding density was 300 g seeds m−2 in the nursery in the Japanese form, the planting density was 9 plants m−2 in the main plot; then, the result was generalized. Herbicides and pesticides were not used during the growing season.
Plant extracts were prepared according to the experimental protocol and kept in a graduated container along with two species of bacteria and sprayed on the rice nursery in three stages after germination until the seedling stage. The seedlings were then transplanted to the main plot until ripening and harvesting, and sampling and measurements were performed. After the growing period, complete physiological maturity, and 14% grain moisture content, harvesting was carryon out. When the crops ripened, 10 plants were randomly sampled from each plot, the yield and yield components of which were examined.
The leaf area was measured using the leaf area meter. To determine chlorophyll content, 0.2 g of the leaf sample was extracted in 80% acetone. The extract was then filtered with the filter paper, and the acetone was added until reaching a volume of 25 ml. Chlorophyll was completely extracted from the leaf tissue. The light absorption of total chlorophylls was read at 645 and 663 nm, respectively, and the total chlorophyll content was obtained using the following equation (Eq. (1)) [27]:
The grain protein content was measured using the Kjeldahl method by Kjeltec 1030 autoanalyzer (made in Sweden). The total nitrogen content was first obtained, and, then, the grain protein content was estimated by multiplying the total nitrogen value by 6.25. To determine the grain starch content, 0.1 g of the fully powdered sample was poured into 15 ml of 80% ethanol. After 20 s, the vortex was filtered by filter paper and placed in an oven at 50 °C until the ethanol evaporated. To remove excess sediments and other compounds, 5 ml of 5% zinc sulfate solution and 4.7 ml of 0.3 N normal barium hydroxide solution were added. After centrifugation, 2 ml of the floating extract and 1 ml of 5% phenol solution were added. After the mixture was stirred well, 5 ml of the concentrated sulfuric acid was added; after 45 min, light absorption was read at 485 nm by the UV-VIS spectrophotometer [28]. At the end of the experiments, the data were analyzed using Statistical Package for the Social Sciences (SPSS) statistical software, and a comparison of the means was performed by LSD’s test at the significance level of 5% [3].
3 Results
The analysis variance results (Table 1) indicated that the main effect of extraction concentration and plant were significant on all studied traits (Table 1). The interaction between extraction and concentration treatment was also statistically significant on all studied traits. Also, the interaction between extraction and plant was significant in all studied traits except grain protein content. In addition, the interaction between concentration and plant was significant on LAI, a number of grains panicle−1, 1000-grain weight, grain yield, and biological yield. Based the on analysis variance table, none of the parameters were affected by the main effect of year and the four-way interaction of year, extraction, concentration, and plant (Table 1).
3.1 Total chlorophyll
The two plants (rice and E. crus-galli L.) responded differently to extraction and their concentration (Table 2). In all extractions, the control treatment had the highest content of total chlorophyll in both plants (rice and E. crus-galli L., 8.38 and 5.69 mg g−1 FW). Increasing in extract concentration decreased the content of total chlorophyll. The results show that the different concentrations (5–15%) of extraction significantly decreased the total chlorophyll content of rice as compared to untreated plants (0%), and high inhibitory impacts were observed in Nettle and Elderberry extraction. Maximum reduction in total chlorophyll of rice was in the highest concentration of Eucalyptus and X. campestris (15%), Nettle, and Elderberry with a concentration of 10% that decreased 30–34% compared to the control. The results showed that there is E. crus-galli L. was a high total chlorophyll accumulation in control plants whereby the highest value was 5.69 mg g−1 FW. However, the total leaf chlorophyll content was remarkably lowered to 3.07 mg g−1 FW in Elderberry extract (15%) while in 15% of sorghum, Broccoli, Nettle, Eucalyptus, S. sp-albos, and X. campestris, the total chlorophyll content was reduced to 4.43, 3.26, 3.14, 4.07, 3.19 and 4.07 3.07 mg g−1 FW which was lower than control (0%) by 22.1, 42.7, 44.8, 28.4, 43.9 and 28.4% respectively.
3.2 Plant height
Relative to the control, the lowest plant height of rice plants was observed at concentrations of 10% and 15% of Elderberry extraction, which was lower by average of 24% and 22% than the control. Averaged over the extract concentration (5%,10%, and 15%), the plant height of rice was 113.6 cm, 111.1 cm, 101.8 cm, 108.1 cm, 95.4 cm, 113.8 cm, and 100.1 cm while rice plants were treated by sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos, and X. campestris, respectively and decreased by 6.9%, 9.0%, 16.6%, 11.4%, 21.8%, 6.7% and 17.9% at control treatment, respectively (Table 2). Regarding E. crus-galli L. and related with control plants which had the highest plant height (104.4 cm), the length of treated plants, was negatively affected by an increase in the level of extract concentration, the highest inhibitory effects were observed at the highest concentration of Elderberry, S. sp-albos and Nettle (57.56 cm, 67.62 cm, and 69.37 cm).
3.3 LAI
In comparison with the control plants, the LAI of rice and E. crus-galli L. plants were negatively influenced by extracts (Table 2). Concerning the rice plants, the highest LAI (4.5) was found in the control treatments related to the plants, which were treated with concentrations of extracts. The lowest value (3.13) were found in higher concentration of X. campestris followed by Elderberry extract (5%, 10% and 15%) and Nettle (10%) extracts which were at the same statistical level. The results showed that LAI was significantly reduced in E. crus-galli L. plants treated with different concentration of extract treatments (Table 2). High inhibitory effects were shown in Elderberry extract with 10% and 15% concentrations (2.05 and 1.68, respectively), followed by 15% concentration of Nottle (2.00). Averaged over the extract concentrations (5%, 10%, and 15%), the LAI of E. crus-galli L. was 3.32, 2.52, 2.32, 2.82, 1.99, 2.50 and 3.08 when E. crus-galli L. plants were treated by sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos and X. campestris, respectively and decreased by 14.8, 35.38, 40.5, 27.6, 48.9, 35.8 and 21.02% at control treatment, respectively (Table 2). It should be noted that LAI of E. crus-galli L. (on average across extract) was 2.93, 2.66 and 2.35, at the 5%, 10%, and 15% and reduced by 24.8, 31.7 and 39.7%, at treated E. crus-galli L. plants with distil water (control), respectively (Table 2).
3.4 Number of panicles m−2
The two plants (rice and E. crus-galli L.) responded differently to extraction and their concentration (Table 2). In all extractions, control treatment had the highest number of panicles plant−1 in both plants (rice and E. crus-galli L., 260.1 and 117.1 mg g−1 FW, respectively). Increasing in extract concentration decreased the number of panicles plant−1. The results show that the different concentrations (5–15%) of extraction significantly decreased the number of panicles plant−1 in rice as compared to untreated plant (0%) and high inhibitory impacts were observed in Elderberry extraction (10% and 15%, 165.4 and 176.1, respectively) which decreased by 36% and 32% compared the control. The results showed that in E. crus-galli L. there was a higher number of panicles plant−1 in control plants whereby the highest value was 117.1. However, the total number of panicles plant−1 was remarkably lowered to 33.6 in Elderberry extract (15%) while in highest extract concentration (15%) of sorghum, Broccoli, Nettle, Eucalyptus, S. sp-albos and X. campestris, the total number of panicles plant−1 was reduced to 92.04, 68.7, 52.6, 75.6, 53.1 and 80.9 which was lower than control (0%) by 21.4, 41.3, 55.0, 35.4, 54.6 and 30.9% respectively.
3.5 No. of grains panicle−1
The decrease in number of grains panicle−1 of rice associated with Elderberry at 10% and 15% (reduced to 72.8) followed by X. campestris at 15% (77.2) which were at same statistical level. Averaged over the extract concentrations (5%, 10% and 15%), the number of grains panicle−1 of rice was 102.7, 89.7, 83.7, 97.7, 77.4, 102 and 81.8 when rice plants were treated by sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos and X. campestris, respectively and decreased by 7%, 18.7%, 24.2%, 11.5%, 29.9%, 7.6% and 25.9% at control treatment, respectively (Table 3). It should be noted that the number of grains panicle−1 of rice (on average across extract) was 94.25, 89.98 and 88, at the 5%, 10% and 15% and reduced by 24.6%, 31.5% and 39.6%, at treated rice plants with distil water (control), respectively (Table 3). Relative to the controls, the maximum reduction in number of grains panicle−1 of E. crus-galli L. was achieved at concentration of 10% and 15% of Elderberry extract and Nettle (15%) which were at same statistical levels and lower by 44.1% and 41.5% than control. Averaged across the extract treatments, the concentration of 15% in all extracts had the maximum inhibitory effects on number of grains panicle−1 (Table 3). Among the extracts, maximum reduction in number of grains panicle−1 was observed in Elderberry (Table 3).
3.6 1000-grain weight
The 1000-grain weight of both plants treated with extracts deceased as the concentration of all extracts increased. The 1000-grain weight of rice of was lowered to 18.7 g in treatment of Elderberry extract with 10% which was lower than control by 23.6% (Table 3). Averaged by concentration, the extract of sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos and X. campestris decreased 1000-grain weight of rice by 7.7%, 14.6%, 20.0%, 14.6%, 21.3%, 8.7% and 20.2%, respectively compared to control. All concentration of extract treatments reduced the 1000-grain weight of E. crus-galli L. The mean maximum and minimum 1000-grain weight of E. crus-galli L. (recorded in plants grown in control and plants treated with 15% of Elderberry extraction), were 4.76 and 3.19 g, respectively (Table 3). Averaged across the extract treatments, the concentration of 15% in all extracts had the maximum inhibitory effects on biological yield (Table 3). Among the extracts, maximum reduction in 1000-grain weight was observed in Elderberry, S. sp-albos, and Nettle which were found statistically at per with Elderberry (Table 3).
3.7 Grain yield
The decrease in grain yield of rice associated with Elderberry at 10% (reduced to 2.58 ton ha−1) followed by Elderberry at 15% (3.25 ton ha−1) and Nettle at 10% and 15% (3.28 and 3.15 ton ha−1, respectively). Averaged over the extract concentrations (5%, 10% and 15%), the grain yield of rice was 4.43, 4.06, 3.37, 4.22, 3.15, 4.25 and 3.7 ton ha−1 when rice plants were treated by sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos and X. campestris, respectively and decreased by 11.54, 18.9, 32.7, 15.7, 37.1, 15.1 and 26.1% at control treatment, respectively (Table 3). It should be noted that grain yield of rice (on average across extract) was 4.28, 3.76 and 3.60 ton ha−1, at the 5, 10 and 15% and reduced by14.57, 24.95 and 28.14%, at treated rice plants with distil water (control), respectively (Table 3). Relative to the control, the maximum reduction in grain yield of E. crus-galli L. was achieved at highest concentration of Elderberry and Nettle (15%) which were at same statistical levels and lower by 74.1% and 69.5% than control. Averaged across the extract treatments, the concentration of 10% in all extracts had the maximum inhibitory effects on grain yield (Table 3). Among the extracts, maximum reduction in grain yield was observed in Elderberry and Nettle (Table 3).
3.8 Biological yield
The capacity of both plants treated with extracts to accumulated biomass deceased as the concentration of all extracts increased. In rice plants, the biological yield was finally affected and significantly reduced (P < 0.01) in plants treated with all extracts (Table 1). High inhibitory effects were observed at 10% of all extracts. The biological yield rice of was lowered to 5.49 ton ha−1 in treatment of Elderberry extract with 10% which was lower than control by 52.6% (Table 3). Averaged by concentration, the extract of sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos and X. campestris decreased biological yield of rice by 12.8%, 22.1%, 24.8%, 20.6%, 29.4%, 12.6% and 24.1%, respectively compared to control. All concentration of extract treatments reduced the biological yield of E. crus-galli L. The mean maximum and minimum biological yield of E. crus-galli L. (recorded in plants grown in control and plants treated with 15% of Elderberry extraction), were 5.69 and 3.07 ton ha−1, respectively (Table 3). Averaged across the extract treatments, the concentration of 15% in all extracts had the maximum inhibitory effects on biological yield (Table 3). Among the extracts, maximum reduction in biological yield was observed in Nettle, S. sp-albos and Braccoli which were found statistically at per with Elderberry (Table 3).
3.9 Grain protein content
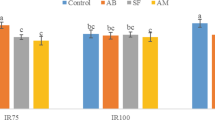
The results presented in Fig. 1 showed clearly the significant different (P < 0.01) of grain protein content in rice and E. crus-galli L. Regarding to rice, the grain protein content (9.82%) was significantly higher than E. crus-galli L. (8.91%) by averaged of 9.26%. The results showed that there was high grain protein content (10%) in control plants (Fig. 2). However, the grain protein content of both plants (rice and E. crus-galli L.) was significantly decreased for those plants treated with extract treatments. High inhibitory impacts were exhibited at 15% of S. sp-albos extract and 10% of Nettle extract (8.9%).
3.10 Grain starch content
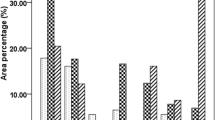
The results showed that there was high grain starch content (61.7%) in control plants (Fig. 3). However, the grain protein content of both plants (rice and E. crus-galli L.) was significantly increased for those plants treated with extract treatments. High stimulate effects were exhibited at 10% of Elderberry (80.7%) and 15% of Nottle extract (79.4%). In addition, the results showed that there was high grain starch content in Nettle, Eucalyptus and Elderberry with content 74.87%, 76.8% and 74.62%, respectively (for rice) and 70.04%, 70.91% and 69.3%, respectively (for E. crus-galli L.). When compared with rice plant, the grain starch content of E. crus-galli L. decreased 7.16, 8.11, 6.45, 7.76, 7.12, 1.80 and 11.59% by application of sorghum, Broccoli, Nettle, Eucalyptus, Elderberry, S. sp-albos and X. campestris (Fig. 4).
4 Discussion
When allelochemicals were stressed, the abscisic acid (ABA) and ethylene concentrations increased [22], and these hormones had a stimulating effect on the chlorophylase activity. Chlorophylase plays an important role in the process of chlorophyll degradation. Allelopathic compounds may either inhibit chlorophyll synthesis pathways, stimulate chlorophyll consumption pathways, or cause both reactions, leading to reduced chlorophyll accumulation and, consequently, decreased photosynthesis and plant growth [29]. Another possible reason for the decreased chlorophyll in plants under allelopathic stress is a change in nitrogen metabolism. Allelopathic compounds cause glutamate, which is the common precursor of chlorophyll and proline synthesis, to be less involved in chlorophyll synthesis [30].
Hormonal imbalance is among the most important reasons for decreased growth of plant shoots and roots. Some of the mechanisms of action of allelopathic substances are similar to those of plant hormones; for example, phenolic acids and polyphenols reduce auxin-stimulated growth by stopping its oxidative decarboxylation [10]. Auxin, as the hormone regulating cell longitudinal growth and its divisions, is among the factors affecting longitudinal growth. Therefore, any disturbance in the action of this hormone inhibits or reduces plant growth [30]. Studies have shown that some of the allelopathic substances interfere with normal levels of the hormone by inhibiting auxin transport, leading to growth arrest and the development of abnormal structures in the growing organ [31]. It seems that with increasing the extract concentration, the number of allelopathic substances increased and prevented the full growth of the leaves; therefore, the leaves remained smaller, and the leaf area decreased [32]. Decreased leaf area can be due to the inhibitory effect of allelopathic substances, reduced cell division, and tissue development [33]. Production of panicles m−2 was considerably reduced when treated with different concentrations of extraction which might be due to translocation of active ingredients of allelochemics [7]. Further, the higher concentrations of extraction might have controlled or arrested the various metabolic processes in leaves causing lesser photosynthates [34] and thus decreased rate in assimilation of growth regulators, which might have checked the formation of panicles. Decrease in number of grains panicle−1 of rice over control shows that stimulatory allelopathic compounds might be responsible for this increase. Furthermore, the decrease in number of grains panicle−1 can also be attributed to decrease in LAI and length of shoots of rice over control [35]. Also, this inhibitory effect in number of grains panicle−1 might be due to checking or inhibition of biosynthesis of gibberellins [36], which are responsible for cell division and cell-elongation. It is expected that the phenolics might have checked the increase of number of grains panicle−1 by preventing the synthesis of protein, nucleic acid and other metabolites responsible for the synthesis of growth regulators [37]. The present findings agree with the report of Tibugari et al. [38]. The allelochemicals released from the extraction might have checked the synthesis and translocation and/or accumulation of plant hormones responsible for production of assimilate for grain filling. Grain setting inside the plant is another important factor of yield per plant. The development of grain depends on the photosynthates available in leaves and their translocation to grains [39]. The allelochemicals might have lowered 1000 grain wight. Allelopathic compounds affect nitrogen metabolism and, consequently, biochemical interactions, photosynthesis, dry matter accumulation in shoots, and grain yield components. However, changes in the nitrogen metabolism rate depend on the type of allelopathic compounds and plant species [40]. Probably over time, due to the decrease in leaf activity and their fall at the end of the growing season, the growth rate of the plant gradually decreases and becomes negative, which results in reduced yield [12]. Decreased biological yield can be due to reduced nutrient and water uptake by roots, reduced leaves for photosynthesis, and decreased photosynthesis by allelochemicals, which resulted in reduced biomass production in plants [9]. The inhibitory effect of allelochemicals on plant growth is also related to the reduced photosynthesis. Photosynthesis reduction leads to the decreased amount of carbohydrates, which ultimately decreases dry matter accumulation in plant organs [41]. Decreased protein content can be due to their high degradation or denaturation rate, changes in their three-dimensional structure and spatial shape, as well as disruption of the protein synthesis process during stress. Protein degradation and formation of molecules with lower weight as well as conversion of polypeptides into amino acids such as proline, glycine, and betaine have also been reported [41]. Some of the respiratory enzymes may be inactivated by allelopathic substances existing in the leaves of this plant. In this case, decomposition of soluble sugars in the treated plants decreases, which leads to the increased levels of soluble sugars [42]. Accumulating carbohydrates such as sugars (glucose, fructose, and sucrose) and starch play an important role in osmotic protection and regulation. The increase in carbohydrates is probably due to inhibiting respiratory enzymes, decomposing soluble sugars, and reducing cell energy levels [43,44,45,46,47,48].
5 Conclusion
Nowadays, weed control programs are designed to reduce toxin consumption and avoid the negative consequences of herbicide use including incomplete weed control, environmental pollution, adverse effects on human health, increasing weed resistance to herbicides, and elimination of natural enemies. Allelopathy and plant pathogens can be among the basic ways to control weeds in the sustainable agricultural system. Maximum reduction in total chlorophyll of rice was in the highest concentration of Eucalyptus and X. campestris (15%) that decreased 30% compared to the control. The lowest height of E. crus-galli L. was observed at highest concentration of Elderberry, S. sp-albos and Nettle (57.56 cm, 67.62 cm, and 69.37 cm). The lowest LAI (3.13) were found in higher concentration of X. campestris. It could be concluded that among the extractions, 15% of Nettle and Elderberry extracts can be used to control E. crus-galli L. at the germination stage under field conditions.
Data availability
The datasets used during the current study available from the corresponding author on reasonable request.
References
Sreenivasulu N (2019) Oryza sativa L. Grain Quality: methods and protocols (methods in Molecular Biology), vol 1892. Humana Press, New York, p 338. https://doi.org/10.1007/978-1-4939-8914-0
Gbanguba AU, Ismaila U, Kolo MGM, Umar A (2011) Effect of cassava-legumes intercrop before Oryza sativa L. on weed dynamics and Oryza sativa L. grain yield at Badeggi Nigeria. Afr J Plant Sci 5(4):264–267
Khanh TD, Anh LH, Nghia LT, Trung KH, Hien PB, Trung DM, Xuan TD (2018) Allelopathic responses of Oryza sativa L. seedlings under some different stresses. Plants 7:40–51. https://doi.org/10.3390/plants7020040
Kong CH, Wang L, Wang P, Nic HW, Mengc XR (2012) Reproduction allocation and potential mechanism of individual allelopathic Oryza sativa L. plants in the presence of competing Echinochloa crus-galli L. Pest Manag Sci 69:142–148. https://doi.org/10.1002/ps.3380
Das CR, Mondal NK, Aditya P, Datta K, Banerjee A, Das K (2012) Allelopathic potentialities of leachates of leaf litter of some selected tree species on gram seeds under laboratory conditions. Asian J Exp Biol Sci 3(1):59–65
Mubeen K, Nadeem MA, Tanveer A, Zahir ZA (2012) Allelopathic effects of Sorghum and sunflower water extractson germination and seedling growth of Oryza sativa L. (Oryza sativa L.) and three weed species. J Anim Plant Sci 22(3):738–746
Tantiado RG, Saylo MC (2012) Allelopathic potential of selected grasses (Family Poaceae) on the germination of Lettuce Seeds (Lactuca sativa). Int J Biol Sci 4:2
Song Y, Liu H (2014) The allelopathic effect of ginseng root exudates on Oryza sativa L. seeds. J Chem Pharm Sci 6(2):785–789
Mishra A (2015) Allelopathic properties of Lantana camara. Int Res J Basic Clin Stud 3:13–28
Dastres E, Safari M, Maghsoudimoud AA (2015) Allelopathic effects of aqueous extract of pagoda tree (Sophora alopecuriodes L.) and creeping jenny (Convolvulus arvensis L.) on five crop plants at seedling growth stage. J Plant Process Funct 4(11):45–58
Hatami Hampa A, Javanmard A, Alebrahim MT, Sofalian O (2018) Allelopathic effects of aqueous extracts from Sorghum (Sorghum bicolor L.) and russian knapweed (Acroptilon repens L.) on seedling growth and enzymes activity of wheat, sugar beet, common lambsquarters and redroot pigweed. J Plant Prot 32(1):101–119
Sadat Asilan K, Modarres Sanavy SAM, Ghahary S, Moradi Ghahderijani M, Panahi M (2015) The evaluation allelopathic effects of Iranian Oryza sativa L. (Oryza sativa L.) cultivars on growth factors of barnyard grass (Echinochloa cruss-galli L.). Environ Sci 12(4):37–46
Kim SY, Madrid AV, Park ST, Yang SJ, Olofsdotter M (2005) Evaluation of rice allelopathy in hydroponics. Weed Res 45(1):74–79. https://doi.org/10.1111/j.1365-3180.2004.00438.x
Lin W, He H, Guo Y, Liang Y, Chen F (2001) Rice allelopathy and its physiobiochemical characteristics. J Appl Ecol 12(6):871–875
Ebana K, Yan W, Dilday RH, Namai H, Okuno K (2001) Variation in the allelopathic effect of rice with water soluble extracts. Agron J 93(1):12–16. https://doi.org/10.2134/agronj2001.93112x
Hussain MI, Danish S, Sánchez-Moreiras AM, Vicente Ó, Jabran K, Chaudhry UK, Branca F, Reigosa MJ (2021) Unraveling sorghum allelopathy in agriculture: concepts and implications. Plants 10(9):1795https://doi.org/10.3390/plants10091795
James D, Devaraj S, Bellur P, Lakkanna S, Vicini J, Boddupalli S (2012) Novel concepts of broccoli sulforaphanes and disease: induction of phase II antioxidant and detoxification enzymes by enhanced-glucoraphanin broccoli. Nutr Rev 70(11):654–665. https://doi.org/10.1111/j.1753-4887.2012.00532.x
Bojovic BM, Jakovljevic DZ, Curcic SS, Stankovic MS (2018) Phytotoxic potential of common nettle (Urtica dioica L.) on germination and early growth of cereals and vegetables. Allelopath J 43(2):175–186. https://doi.org/10.26651/allelo.j./2018-43-2-1139
Harding DP, Raizada M (2015) Controlling weeds with fungi, bacteria and viruses: a review. Front Plant Sci 6:1–14. https://doi.org/10.3389/fpls.2015.00659
Mithila J, Hall JC, Johnson WG, Kelley KB, Riechers DE (2011) Evolution of resistance to auxinic herbicides: historical perspectives, mechanisms of resistance, and implications for broadleaf weed management in agronomic crops. Weed Sci 59:445–457. https://doi.org/10.1614/WS-D-11-00062.1
Bailey KL, Pitt WM, Falk S, Derby J (2014) The effects of Phoma macrostoma on non-target plant and target weed species. Biol Control 58(3):379–386. https://doi.org/10.1016/j.biocontrol.2011.06.001
Zabot GL, Silva MF, Terra LD, Foletto EL, Jah SL, Dal Pra MF (2012) Simulation of the xanthan gum production in continuous fermentation systems. Biocatal Agric Biotechnol 1:301–308. https://doi.org/10.1016/j.bcab.2012.08.002
He Y-W, Wu J, Zhou L, Yang F, He Y-Q, Jiang B-L (2011) Xanthomonas campestris Diffusible factor is 3-hydroxybenzoic acid and is associated with xanthomonadin biosynthesis, cell viability, antioxidant activity, and systemic Invasion. Mol Plant-microbe Interact 24(8):948–957. https://doi.org/10.1094/MPMI-02-11-0031
Valan Arasu M, Duraipandiyan V, Agastian P, Ignacimuthu S (2009) In vitro antimicrobial activity of Streptomyces spp. ERI-3 isolated from western ghats rock soil (India). J Mycol Méd 19:22–28. https://doi.org/10.1016/j.mycmed.2008.12.002
Chen P, Huang R, Zuo LZ, Zhang YQ, Li L (2022) Allelopathic potential of root endophytic bacterial metabolites on seeds germination of Casuarina equisetifolia. Allelopath J 55(2):261–276. https://doi.org/10.26651/allelo.j/2021-52-2-1321
Abbas T, Zahir ZA, Naveed M, Abbas S, Alwahibi MS, Elshikh MS, Mustafa A (2020) Large scale screening of rhizospheric allelopathic bacteria and their potential for the biocontrol of wheat-associated weeds. Agronomy 10(10):1469. https://doi.org/10.3390/agronomy10101469
Arnon DI (1949) Copper enzymes in isolated chloroplasts, polyphenoxidase in beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Sivakumar P, Sharmila P, Pardha Saradhi P (2000) Proline alleviates salt stress induced enhancement in the activity of ribulose-1, 5-bisphosphate oxygenase. Biochem Biophys Res Commun 279:512–515. https://doi.org/10.1006/bbrc.2000.4005
Elisante F, Tarimo MT, Ndakidemi PA (2013) Allelopathic effect of seed and leaf aqueous extracts of Datura stramonium on leaf chlorophyll content, shoot and root elongation of Cenchrus ciliaris and Neonotonia ightii. Am J Plant Sci 4:2332–2339. https://doi.org/10.4236/ajps.2013.412289
Zuo S, Li X, Ma Y, Yang S (2014) Soil microbes are linked to the allelopathic potential of different wheat genotypes. Plant Soil 378:49–58. https://doi.org/10.1007/s11104-013-2020-6
Mirsky SB, Ryan MR, Teasdal JR, Curran WS, Reberg-Horton CS, Spargo JT, Wells MS, Keene CL, Moyer JW (2013) Overcoming weed management challenges in cover crop-based organic rotational no-till soybean production in the eastern United States. Weed Technol 27:193–200. https://doi.org/10.1614/WT-D-12-00078.1
Azizbeigi S, Khara J (2014) The allelopathic effects of aqueous extract of walnut (Juglans regia) leaves on some physiological and biochemical characteristics of parsley plants inoculated by mycorrhizal fungus Glomus versiforme. J Plant Process Funct 2(6):65–76
Jabran K, Mahajan G, Sardana V, Chauhan BS (2015) Allelopathy for weed control in agricultural systems. Crop Prot 72:57–65. https://doi.org/10.1016/j.cropro.2015.03.004
Macías AF, Mejías FJR, Molinillo JMG (2019) Recent advances in allelopathy for weed control: from knowledge to applications. Pest Manag Sci 75:2413–2436. https://doi.org/10.1002/ps.5355
Hussain MI, Reigosa MJ (2021) Secondary metabolites, ferulic acid and p-hydroxybenzoic acid induced toxic effects on photosynthetic process in Rumex acetosa L. Biomolecules 11:233. https://doi.org/10.3390/biom11020233
Farooq M, Khan I, Nawaz A, Cheema MA, Siddique KH (2020) Using sorghum to suppress weeds in autumn planted maize. Crop Prot. https://doi.org/10.1016/j.cropro.2020.105162
Marchiosi R, dos Santos WD, Constantin RP, de Lima RB, Soares AR, Finger-Teixeira A, Mota TR, de Oliveira DM, Foletto-Felipe MDP, Abrahão J et al (2020) Biosynthesis and metabolic actions of simple phenolic acids in plants. Phytochem Rev 19:865–906. https://doi.org/10.1007/s11101-020-09689-2
Tibugari H, Chiduza C, Mashingaidze AB, Mabasa S (2020) High sorgoleone autotoxicity in sorghum (Sorghum bicolor (L.) Moench) varieties that produce high sorgoleone content. S Afr J Plant Soil 37:160–167. https://doi.org/10.1080/02571862.2020.1711539
Ayeni JM, Kyode J (2021) The effects of aqueous extracts from maize roots and sorghum stem on the germination and radicle growth of Sphenosylis sternocarpa hochst ex. Rich.(African Yam beans). Afr J General Agri 5:117–121
Young SL, Pierce FJ, Nowak P (2014) Introduction: scope of the problem-rising costs and demand for environmental safety for weed control. In: Young SL, Pierce FJ (eds) Automation: the future of Weed Control in Cropping Systems. Springer, Dordrecht, pp 1–8. https://doi.org/10.1007/978-94-007-7512-1_1
Le-Thi H, Lin CH, Smeda RJ, Leigh ND, Wycoff WG, Fritschi FB (2014) Isolation and identification of an allelopathic phenylethylamine in Oryza sativa L. Phytochemistry 108:109–121. https://doi.org/10.1016/j.phytochem.2014.08.019
Niroomand A, Seyyednejad M, Abrahimpour F, Gilani A, Bakhshikhaniki G (2017) Study on the effect of applying different levels of olive pomace (Olea europaea L.) on the grain yield of three Oryza sativa L. (Oryza sativa L.) cultivars in climatic conditions of Khuzestan. J Iran Plant Ecophysiol Res 12(46):31–41
Aasifa G, Badruzzaman SM (2014) Evaluation of allelopathic effect of Eclipta Alba (L.) Hass.k on biochemical activity of Amaranthus spinosus L., Cassia tora L. and Cassia sophera L. Afr J Environ Sci Technol 8(1):1–5. https://doi.org/10.5897/AJEST2013.1617
Keshavarz Mirzamohammadi H, Modarres-Sanavy SAM, Sefidkon F, Mokhtassi-Bidgoli A, Mirjalili MH (2021a) Irrigation and fertilizer treatments affecting rosmarinic acid accumulation, total phenolic content, antioxidant potential and correlation between them in peppermint (Mentha piperita L.). Irrig Sci 39:671–683. https://doi.org/10.1007/s00271-021-00729-z
Keshavarz Mirzamohammadi H, Tohidi-Moghadam HR, Hosseini SJ (2021b) Is there any relationship between agronomic traits, soil properties and essential oil profile of peppermint (Mentha piperita L.) treated by fertiliser treatments and irrigation regimes? Ann Appl Bio 179(3): 331–344. https://doi.org/10.1111/aab.12707
Hosseini SJ, Tahmasebi-Sarvestani Z, Mokhtassi-Bidgoli A, Keshavarz H, Kazemi Sh, Khalvandi M, Pirdashti H, Hashemi-Petroudi SH, Nicola S (2023a) Functional Quality, Antioxidant Capacity and Essential Oil Percentage in Different Mint Species Affected by Salinity Stress. Chem Biodivers 20(4): e202200247. https://doi.org/10.1002/cbdv.202200247
Hosseini SJ, Tahmasebi-Sarvestani Z, Mokhtassi-Bidgoli A, Keshavarz H, Kazemi Sh, Khalvandi M, Pirdashti H, Bovand F, Abassian A (2023b) Do various levels of salinity change chlorophyll fluorescence, nutrient uptake, and physiological characteristics of Mentha ecotypes. Ind Crop Prod 203:117199. https://doi.org/10.1016/j.indcrop.2023.117199
Sabourifard H, Estakhr A, Bagheri M, Hosseini SJ, Keshavarz H (2023) The quality and quantity response of maize (Zea mays L.) yield to planting date and fertilizers management. Food Chem Adv 2:100196. https://doi.org/10.1016/j.focha.2022.100196
Acknowledgements
The authors thank Dr. Luis Avila (Associate Professor, Weed Science Mississippi State University) and Dr. Fernanda Reolon Souza (Research Associate at Mississippi State University, Plant and Soil Sciences Department, Mississippi State University) for critical reading and editing of the manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
M.E.: Wrote the main manuscript text. PK: Corresponding author, Data analyzer. HRL: Read and edit the manuscript. HRT-M: Laboratory operator. SH-J: Farm operator.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Plant Guidelines
The authors confirm that the use of plants in the present study complies with international, national and/or institutional guidelines.
Permissions to collect the plants/plant parts
All plant specimens collected with permissions.
Source of the plant used in your study
All plants seeds name and he source of them are in the Methods section.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ebrahimpour, M., Kasraie, P., Larijani, H.R. et al. The impact of two bacterial species and five aqueous plant extracts on yield and yield components of rice (Oryza sativa L.) and surof (Echinochloa crus-galli L.). SN Appl. Sci. 5, 278 (2023). https://doi.org/10.1007/s42452-023-05500-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-023-05500-x