Abstract
Drying oilseeds to reduce their moisture content is crucial in order to preserve the seeds and their contents. However, due to the nature of conventional storage facilities, it is ideal to dry seeds just before using them for oil extraction and/or in situ biodiesel production as the seeds dried in advance might recover the equilibrium moisture content due to the humidity from the air. Thus, drying the seeds immediately before oil extraction is vital to reduce the moisture content to its minimum. In the present study, the effects of five drying temperatures (313, 323, 333, 343 and 353 K) on the degree of moisture loss from Jatropha seeds at storage and the suitability of the drying processes to reduce the seed moisture to its minimum were investigated. The drying experiments of non-pretreated (whole seeds) and pretreated (crushed seeds) seeds were performed in a heating furnace. It was found that increasing in drying temperature promoted the rate of moisture loss, and the evaporation of moisture from the crushed seeds was faster than that of the whole seeds. However, the largest weight loss (6.47%) and the smallest seed residual moisture content (0.34%) were obtained when the whole seeds dried at 353 K. The findings of the present experiments suggested that drying the whole seeds of Jatropha at 353 K could provide dried seeds with suitable moisture content for oil extraction and/or in situ biodiesel production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Biodiesel is an alternative to petroleum-based fuels and can be produced from a variety of feedstock, including vegetable oils, animal fats, and waste cooking oils [1]. Currently, more than 95% of the biodiesel is being produced from edible vegetable oils such as rapeseed, sunflower, palm and soybean [2,3,4]. The price of edible oils has significantly increased [3, 5] as a result of the increasing of vegetable oil demands caused by the ever-increasing global population [5]. Using expensive edible vegetable oils for biodiesel production makes the process costly as the feedstock alone account up to 75% of the total price [4, 6]. Thus, the cost of the biodiesel feedstock becomes the main obstacle for the commercialization of the product [7]. Moreover, extensive conversion of edible oils to biodiesel may lead to food crisis [1] and causes major environmental problems such as serious destruction of vital soil resources, deforestation and usage of much of the available arable land [4]. Therefore, production of biodiesel from low cost non-edible oil feedstock such as Jatropha curcas L. seeds and waste cooking oils would be a potential solution [1, 8].
The oil content of Jatropha seed is reported to be ranged from 30 to 50% by weight of the seed and 45–60% by weight of the kernel [9]. Due to the presence of major toxic compound (the phorbol esters) in Jatropha seed oil, biodiesel production from the seeds does not compete with human consumption [10, 11]. Production of biodiesel from non-edible feedstock such as Jatropha could be a good alternative to overcome the food versus fuel crisis that could occur due to continuous conversion of edible oils to biodiesel [3, 4].
Jatropha seeds are storable and its further processing can be delayed, and this makes the production of the crop suitable to remote areas [12]. However, the moisture of contents of the seeds should be reduced just after harvest to minimize loss of quality [13]. According to Almeida et al. [14], due consideration should be given to the seed moisture content during storage as this parameter affects seed chemical composition and the speed of seed metabolic activities. Moisture content of oilseeds is also one of the important parameters that determine the quantity of oil which can be obtained from the seeds [15]. Thus, its measurement is an inevitable operation during postharvest processing such as handling, storage, milling and oil extraction [16]. Moisture contents of oilseed may depend on the conditions of collections, stages of seed maturation [17], postharvest processing [16] and storage conditions [15].
It has been reported that Jatropha seeds that were harvested from green, yellow and brown fruits differed in their moisture content, but the moisture contents of the seeds reach values between 7.6 and 8.3% after natural drying at room conditions, independent of the harvest stages [17]. The seeds should be shade dried for sowing but dried in the sun for oil production, and its moisture content should be reduced to around 6–10% for storage. If kept dry and stored at optimum conditions, the seeds may be stored for up to 12 months without loss of oil content [12].
The drying of seeds is a required step to achieve the desired moisture content. Knowing the drying characteristics of Jatropha seeds is very essential in its handling and as a preventive protection approach to preserve the material in its current form [18]. Drying processes are also very important for easy extraction of oil from the seeds [19]; and dried seeds with low moisture content give higher percentage of oil than the wet seeds [18]. For instance, in the study of oil extraction from soybean, using hexane as solvent, Lawson et al. [20] obtained the oil yield of 15% and 11.81% from seeds with moisture content of 10% and 20%, respectively. According to the authors, non-polar solvents such as hexane are immiscible in water and oil extraction efficiency of these solvent from seeds with high moisture contents would be reduced, and thus, resulted in lower oil yield.
Drying oilseeds to a lower moisture content, particularly to the moisture content for oil extraction and/or in situ biodiesel production, and keeping it up at this moisture content in storage is impractical. During seed storage, drying oilseeds to very low moisture content is not of such use as the seeds return back to the equilibrium moisture content in relation to the relative humidity of storage environment [21]. Thus, drying the seeds at storage prior to oil extraction and/or in situ biodiesel production may be needed.
There were several research reports on the optimum moisture contents of Jatropha seeds for various oil extraction methods. It was found that the rate of Jatropha oil extraction by screw press increased with increase in temperature and decreased with increasing of seed moisture content [22]. Oil extraction by this method also demonstrated that Jatropha seeds with low moisture content provide higher oil yield than seeds with high moisture content [23]. In the extraction of oil by mechanical press, drying of fresh Jatropha seeds to moisture content of about 5.9% and deshelling the seeds to kernels with moisture content of 3.8% and pressing the kernels gave a maximum oil yield of 87.4% (at applied pressure of 19 MPa, 90 °C pressing temperature and 10 min of pressing) [24]. This moisture content was also reported as the optimum moisture level for the extraction method used. Moreover, in the mechanical extraction of oil from Nyamplung (Calophyllum inophyllum L.) by screw press, the larger oil yield (33.39%) was obtained from seeds with moisture content of 1.2% (dry based, d.b) compared to 15.56% oil yield from seeds with moisture contents of 20% [25].
Higher moisture content of the oilseeds is also one of the important factors that could cause oil hydrolysis and the formation free fatty acids (FFA) in the seeds, which reduces the oil quality. The post extraction pre-treatment of the oil to reduce its FFA gives lower oil yield; and this does also make the oil production process more costly [26]. Oil with lower FFA content is often preferred for direct utilization in alkaline catalyzed transesterification of biodiesel production [27], and the above limit of FFA in the oil should be 1% for satisfactory base catalyzed transesterification [28]. Higher moisture and FFA contents (15% FFA) of Jatropha oil have resulted in the current commercial alkaline catalyst based transesterification not being suitable to be employed [29]. The acid-catalyzed transesterification of Jatropha oil is an alternative, but it is much slower than the base catalyzed one, and thus, not efficient [30]. The complicity of multistage processing involved (extraction, drying, degumming and deacidification) would also add to the production cost and thus contribute negatively to the biodiesel cost effectiveness [31].
In the extraction of Jatropha oil from ground kernel with moisture content of 0.912%, using Soxhlet extractor and hexane as solvent, a maximum of 45% oil that contained 0.9% FFA was obtained [32]. It was also reported that the oil obtained by this method did not need pretreatment with acid for basic catalyzed biodiesel productions [32], as its free fatty acid content is less than 1% [33].
In basic catalyzed in situ biodiesel production, tolerance to moisture content of the feedstock depends on the in situ technology to be adopted and the amount of alcohol being used [34]. However, reports from several investigations showed that seeds with moisture content less than 1% is suitable for in situ transesterification of biodiesel production. For instance, in an integrated extraction and transesterification process from Jatropha seeds using hexane and methanol via base catalyzed transesterification, seeds with moisture content less than 1% was used, and the highest yield obtained was of 90.8% at optimum conditions [35]. Kartika et al. [36] reported that in a solvent extraction and in situ transesterification by alkaline catalyst, seeds with moisture content less than 1% was used as increase in water content of the oilseeds can cause ester saponification [36, 37].
In the study on direct in situ alkaline-catalyzed transesterification of cottonseeds, Qian et al. [37] found that decreasing in seed moisture content had increased the amount of oil dissolved in methanol. Moreover, in reactive extraction technologies using hexane as co-solvent, it was found that seeds with moisture content of 1% gave the maximum biodiesel yield (68.3%) compared to seeds with 3 and 5% moisture contents [38]. It was also reported that reducing moisture content of the seeds did lower the amount of chemicals requirements during in situ transesterification process. Accordingly, a reduction of 60% methanol and 56% sodium hydroxide was found when soybean flakes with moisture contents of 7.4% were dried in a convection oven until the water content reached 0% prior to in situ transesterification [39].
Research papers that deal with drying process of oilseeds in general and that of Jatropha in particular, mainly deal with drying of freshly collected seeds for storage and/or for oil extraction. However, to our knowledge, no research investigation has been found on the influence of different temperatures on the degree of moisture removal from Jatropha seeds at a storage moisture level.
In the present study, we investigated the impact of five different drying air temperatures (313, 323, 333, 343, and 353 K) on the moisture removal process of Jatropha seeds under different pretreatment conditions in order to produce the dried seeds with minimum moisture content. In addition, the physical appearance of the seeds during drying was investigated.
2 Materials and method
2.1 Materials
Seven Jatropha seed collections at storage: Kurkura, Bira, Salmene1, Salmene2, Salmene3, Shekla and Gerbi were used for the experiments. For the crushing pretreatment process of Jatropha seeds, a bowl-shaped mortar and pestle made from stone were used. After crushing, the particle sizes of the pulverized seeds were estimated by three different stainless steel sieves with openings of 500 µm, 1 mm and 2 mm woven cloth (Control Group, 15-D2245/J, 15-D2215/J and 15-D2185/J). All the seed samples were weighed on a digital balance machine having 0.01 mg accuracy (Mettler-Toledo, PG 5002 Delta Range, Switzerland). The seed samples were placed on Petri dishes (100 mm × 15 mm) during drying. The drying experiments and determination of initial and residual moisture contents of the seed samples were performed using a heating furnace (Narbetherm P300, Germany).
2.2 Experimental procedures
To determine the effects of temperature and pretreatments on the drying of Jatropha seeds at the storage moisture content, the methodology employed previously in the group has been followed [40]. From the seven seed collections, Salmene3 seed collection was selected and used for the drying experiment as base line for comparison. This collection was selected based on the information recorded during seed collection, and it was chosen as the best one since it was obtained from the fruits at the physiological maturity stage. It has been reported that maximum physiological quality and lipid content of Jatropha seeds occur at physiological seed maturity, which corresponds to seeds obtained from yellow fruits [41, 42].
In the drying experiment, the non-pretreated whole seeds (WS) and the pretreated crushed seeds (CS) of Salmene3 collection were used. The seeds were cleaned manually by removing all foreign matter such as stones, dirt and broken seeds before the experiment. For all drying experiments, ca. 15 g of Jatropha seeds were used. Four 15 g seed samples were weighed separately for the drying at a particular drying temperature (two samples for each treatment in order to have duplicate measurements). The first two WS samples were dried without any pretreatment with its seed coat. The second two seed samples were crushed and grinded mechanically by hands, using mortar and pestle. The particle sizes of the crushed seeds (PSCS) were estimated using three different sieve sizes as it was described in the previous study published elsewhere [43]. Thus, the crushed seeds used in this experiment was a mixture of four particles sizes. The average proportions (%) of the PSCS: PSCS > 2 mm, 2 mm > PSCS > 1 mm, 1 mm > PSCS > 500 µm and PSCS < 500 µm were 14.95 ± 8.2, 29.96 ± 4.38, 35.32 ± 10.5 and 19.77 ± 4.74, respectively.
Two different dimensional properties (geometric and arithmetic mean diameters) of the whole seeds of the selected seed collection (Salmene3 collection) were determined as these properties could affect the drying process of the seeds. The geometric and arithmetic mean diameters (mm) of the selected seed collection were calculated by measuring the length, width and thickness of 50 whole seeds with measuring calipers, and using Eqs. 1 and 2, respectively [44, 45].
where Dg = geometric mean diameter, Da = arithmetic mean diameter, L = length, W = width and T = thickness of the whole seeds in millimeter.
Five different air temperatures (313, 323, 333, 343, and 353 K) that were used to dry Hass avocado seeds were adopted for drying the Jatropha seed samples [40]. The influence of these five air temperatures was selected for the investigation as several studies reported that air temperature is the dominant parameter that affecting the drying process of the seeds [40, 46, 47]. All the seed samples were kept in separate Petri dishes (100 mm × 15 mm), uniformly distributed on it and then placed in the heating furnace for drying.
The effectiveness of the drying temperatures on the moisture evaporation from Jatropha seeds was systematically recorded for 5760 min to ensure the achievement of the critical moisture level at which no more moisture loss occurred. To understand the rate of moisture evaporation, the weight loss data were recorded following the methods used in related studies by Perea-Flores et al. [46] and Avhad and Marchetti [40]. Accordingly, the seeds samples were taken out from the heating furnace and weighed on a digital balance at a predetermined time intervals by taking less than 15 s to weigh the samples. Thus, the samples were exposed to the room temperature for very short instant during weighing, and then, put back in the hearing furnace. The samples were weighed until no more weight loss occurred in three consecutive weight data. Based on the information obtained for Salmene3 seeds for the different temperatures, the WS samples of all the remaining six seed collections were also dried at 353 K following the aforementioned procedures. This was done to compare the methodology for different seed collections.
All experiments were replicated twice to obtain reproducibility in the experimental findings. Although the result obtained from the replicated experiments did not significantly vary, the average values were used in data analysis and reporting. The weight losses (wt%) of the seed samples (both the WS and CS) were determined relative to the initial weight of the seeds just before drying (wet based). The residual moisture remained in the dried seeds (wt%), which could also be referred as the experimental equilibrium moisture content (EMC) of the dried seeds was calculated using Eq. 3 below. The initial moisture contents (M0) of the seeds were determined by drying 15 g of the seed samples at 105 °C for 24 h [48, 49].
where M0 = the initial moisture contents of the seeds and Mf = the final moisture loss from the seeds at the time when no more moisture (weight) loss occurred.
As it was also very important to investigate the impact of drying air temperatures on the physical properties of seeds as it impacts the seed drying process [40], the physical appearance of the samples were also investigated. For this purpose, the drying seeds were carefully observed, and photographed using a digital photo camera (Canon, DS126061, Japan) every 24 h (1440 min) during the drying experiment for all drying temperatures.
3 Results and discussion
3.1 Moisture loss of crushed and whole seeds
Table 1 presents the dimensional properties of the whole seeds of Salmene3 collection. As it could be observed from Table 1, the mean value for the seed length, width, thickness, geometric mean diameter, and arithmetic mean diameter were found to be 15.46 ± 0.74, 10.24 ± 0.54, 7.28 ± 0.57, 10.47 ± 0.501 and 10.99 ± 0.48 mm, respectively. All the seed mean dimensions found in the current study were in the range of the values determined by Mousa et al. [50] for Jatropha seeds collected from Egypt. The initial moisture content of the selected seed was also found to be 6.81%. This result demonstrated that the moisture content of the selected seed was in the range of the safe seed storage moisture content (6–10%) recommended for Jatropha seeds [12].
From the result of the drying experiment, it was found that weight loss by the CS was faster than that of the WS at the beginning of seed drying and slowed down after some hours depending on the drying temperatures. Accordingly, the weight losses (wt%) by the CS were larger than that of the WS before the drying times of around 1440, 480, 330, 270 and 240 min for the drying temperatures of 313, 323, 333, 343 and 353 K, respectively. After the indicated drying times, the weight losses (wt%) by the CS was found to be lower than that of the WS for all respective temperatures. The time at which the weight losses of WS and CS became about equal for the same drying temperature, and the respective weight loss recorded are presented in Table 2.
As it is evident from Table 2, the time taken for the weight losses (wt%) by CS and WS to be about equal was the longest for the lowest temperature of drying (around 1440 min for 313 K,) and the shortest for the highest temperature (around 240 min for 353 K). This shows that the time needed for the weight losses (wt%) of CS and WS dried at 353 K to be about equal was 6 times shorter compared to that of the same samples dried at 313 K. The reason for this is that due to faster seed moisture evaporation rate at higher temperature [48], and the process rapidly reached at the falling drying rate period compared to the drying process at lower temperature.
3.2 Effects of temperatures on the physical appearances of seed samples
The experiment on the effects of drying temperatures on the physical appearance of Jatropha seed samples was conducted to observe if charring of the seeds surface would occur and prevent the moisture loss during drying the samples at higher temperature as reported for the non-pretreated whole Hass avocado seeds [40]. In the present study, after a close examination of the seeds before and after drying, and evaluating the photos taken at the same times, only a blackening of the seed coat on the WS and a similar transformation over the upper surfaces of the CS were observed. After drying, the crushed seeds seemed to be stickier than at the beginning of the study. Except those previously mentioned changes, no other major modification of physical appearances was seen as presented in Fig. 1.
The results obtained in the present study was in contrast with the physical appearance of Hass avocado seeds dried at 313, 323, 333 K, 343 and 353 K, where rupturing of the seeds into two pieces, and hardening and charring of the outer surface of the seeds occurred at 333 K and higher temperatures [40]. The authors also reported that rupturing of the seeds at higher temperatures promoted moisture evaporation; however, the higher temperatures damaged the seeds due to charring and resulted in incomplete evaporation of water from the seeds at the end of drying.
3.3 Effects of temperatures and seed pretreatment on seed drying
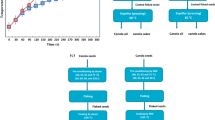
The effect of five different drying air temperatures on the extent of moisture evaporation from the WS and CS are graphically represented in Figs. 2 and 3, respectively. As it is evident from Figs. 2 and 3, the rise in operative air temperature accelerated the drying process of the seeds. After 360 min of drying, air temperature of 353 K resulted in 6.15% reduction in the weight of the WS, which was about twice the weight reduced using 313 K at the same drying time. Weight loss of CS at drying temperature of 353 K was also much higher than that of 313 K. This behavior was in agreement with the findings in which fresh Jatropha seeds were dried at 313 and 333 K, and found that evaporation of water from the seeds at 333 K was 2 times faster than those dried at 313 K [24]. In another drying experiments using freshly collected Jatropha seeds with initial moisture content of 61% (d.b), the evaporation of water from the seeds was found to be 8 times faster when temperature was increased from 309 to 378 K [48]. In drying of fresh Hass avocado seeds (with initial moisture content of 51.2%), air temperature of 353 K resulted in 4.3 times weight reduction compared to that of 313 K after 720 min of drying time [40]. Thus, the rise in operating temperature minimizes the time required for the drying process. The larger weight reduction at higher temperature in the latter two investigations compared to the present study might be due to larger initial moisture contents of the fresh seeds.
As it could be seen from Figs. 2 and 3, for all temperatures, the weight losses of the CS and WS were found to increase with time until it reached a critical value above which there was no further increase of the weight loss. With increasing of time of drying, the rate of weight loss decreased gradually, and finally, no more weight loss was observed. This happened when moisture content of a seed would equilibrate with the relative humidity of the air surrounding it [51]; and the residual moisture remained in the seed at this time is the equilibrium moisture content (EMC) of the dried seed. Table 3 shows the final weight loss (at EMC) by WS and CS, their EMC for different temperatures and the time taken to reach the EMC.
As it is evident from Figs. 2, 3 and Table 3, the weight losses (wt%) by CS were faster than that of WS for higher temperatures, particularly for 333, 343 and 353 K as the total time required to reach at EMC for WS is 2 times longer than that of CS. The results obtained in this study was in agreement with the findings reported for the drying experiment using crushed kernels (paste), kernel with particles size of 4 and 8 mm, and the whole kernels of Buccholzia coriacea and Butyrospermum parkii using drying temperatures of 318 and 333 K [52]. From this investigation, it was found that the decline in weight of the paste was very rapid compared to those of other treatments. According to the authors, the paste gave the highest drying rate, at each drying temperature compared to samples with larger particle sizes. The findings of the present study was also in line with the results reported from the drying experiments of Hass avocado seeds [40], where the weight loss of crushed avocado seeds was faster than that of the whole seeds. The faster moisture loss of crushed seeds could be due to the fact that grinding of the seeds provoked rupture of the cell membranes, thus, providing a larger surface area for rapid moisture loss to occur [52].
In the current study, although weight loss (wt%) of the CS was faster than that of the WS, the final weight losses (wt%) of the WS were larger than that of the CS, for all respective temperatures (Table 3). The maximum final weight loss of 6.47% for WS, which was obtained at 353 K, was also greater than that of the CS (6.21%). Moreover, the lowest residual moisture content (0.34%) was found in the WS dried at 353 K.
The smaller final weight loss (wt%) of the CS compared to the WS might be due to the hardening of pulverized particles of Jatropha seeds due to larger surface area that could be exposed to the drying temperatures. Some amount of moisture might also be lost by adhering to the surface of mortar and pestle during crushing the seeds [40], and to the surface of sieves during the estimation of CS particle size. These two activities were not applied to the WS prior to drying. Although the same amount of the whole and crushed seeds (15 g) and similar Petri dishes (100 mm × 15 mm) were used during the drying experiments, the thicknesses of the bed of CS and WS on the Petri dishes were different. There was also larger spaces for drying air circulation between individual whole seeds than that of the particles of crushed seeds. These might also be resulted in smaller final weight loss of the crushed seeds.
As it could be observed from Table 3, drying temperature of 353 K could reduce the moisture content of both WS and CS to below 1% while other drying temperatures could not do so. As it has been previously mentioned, reducing the moisture content of seeds to 0.912% allows the production of quality oil with FFA content of 0.9% [32] which could be directly used in alkaline catalyzed transesterification for biodiesel production [32, 33]. Dried seeds with moisture content less than 1% could also be used in alkaline catalyzed in situ transesterification for biodiesel production [35, 36]. Moreover, although the drying temperature of 353 K could reduce the moisture content of WS and CS to less than 1%, the moisture loss by WS (6.47%) was larger than that of CS (6.21%). Drying of the seed samples at a temperature higher than 353 K has not been tested based on the work done by Subroto et al. [24], where it has been shown that temperatures higher than 333 K could increase oil peroxidation in shell unprotected Jatropha seeds. Oil peroxidation reduces the quality of oil as Jatropha seed has high unsaturated oil content [24]. Therefore, drying the whole Jaatropha seeds at 353 K for 2880 min drying time was considered as optimal for satisfactory drying of Jatropha seeds for oil extraction and in situ biodiesel production.
From the above results and the information from literature, it is possible to say that reducing the moisture contents of the whole Jatropha seeds at storage to its minimum using the aforementioned methods prior to oil extraction has several benefits. For instance, reducing the moisture content of the seed to below 1% could ease oil extraction using non-polar solvents [19, 20]. It could also avoid the post extraction boiling of the oil to remove the oil moisture, and thus, it reduces the possibility of oil peroxidation. According to Subroto [24], Jatropha oil is highly unsaturated and susceptible to lipid peroxidation. Lowering the seed moisture to its minimum could also lower the amount of chemicals required during in situ transesterification process using alkaline catalysts [39]. Moreover, drying Jatropha seeds using the standard heating furnace does not require very specialized expert, and it could be easily performed.
3.4 Drying different collections of the whole seeds
Table 4 shows the initial moisture content (wt%), final moisture loss (wt%) and residual moisture contents/equilibrium moisture contents (EMC) of different whole Jatropha seeds at 353 K. As it could be seen from Table 4, all the seven seed collections had different initial moisture contents. The initial moisture contents of the seed collections named Gerbi, Salmene1, Shekla, Salmene3, Kurkura, Bira and Salmene2 were found to be 8.72, 7.44, 7.12, 6.81, 6.80, 6.71 and 6.51% (wt%), respectively. The differences in moisture contents of the seed collections might be due to differences of the climatic conditions of the area from where the seeds were collected, stage of seed maturity [17] and differences of postharvest processing of the seeds [16]. Moreover, the moisture contents of all the seven seed collections were in the range of safe storage moisture content (6–10%) recommended for Jatropha seed [12].
Figure 4 also presents the weight loss (wt%) of different collections of the whole Jatropha seeds during drying at 353 K as the function of drying time. As it is evident from Fig. 4, the trends of weight loss in the seven seed collections was almost similar. However, the amount of moisture loss by different seed collections differed. As it could be observed from Table 4 and Fig. 4, the final moisture loss by Gerbi collection was the largest (8.58%) and that of Salmene2 (5.36%) was the lowest. The differences in the amount of moisture loss (%) for different collection of Jatropha seeds might be due to the differences of the initial moisture contents, seed sizes and other physical traits for various seed collections. It was reported that seed grain size [53], porosity, shape and surface area [54] are some of the important seed physical properties that affect the drying profile of the seeds. Factors such as the climatic conditions of the seed collection sites, stage of seed maturity and the differences in postharvest processing of the seeds could also influence the moisture loss (%) of the seed collections as these factors could affect the moisture contents of the seeds [16, 17].
As it could be seen from Table 4, drying temperature of 353 K reduced the residual moisture contents of the six seed collections to below 1%, however the residual moisture content of Salmene2 (1.15%) was above the recommended value for oil extraction and in situ transesterification for biodiesel production.
4 Conclusions
Moisture content of Jatropha seeds is one of the important parameters that determine the quantity and quality of oil produced from the seeds. As it is impractical to dry oilseeds such as Jatropha to lower moisture content and keep it up at storage, drying of the stored seed for oil extraction and/or in situ biodiesel production is foremost important. From the investigation of the effects of drying air temperature ranged from 313 to 353 K on the drying process of the whole and crushed Jatropha seeds at storage condition, it was found that the rise in operating temperature accelerated the drying process of the seeds. The weight loss (wt%) of crushed seeds due to moisture evaporation was faster than that of the whole seeds, however, the drying process of the whole seed at 353 K resulted in the maximum weight loss of 6.47%, which was larger than that of the crushed seeds (6.21%). The lowest residual moisture content (0.34%) was also found in case of the whole seeds of Jatropha dried at 353 K. Thus, it could be concluded that drying the whole seeds of Jatropha at 353 K for 288 min was considered as the optimal for the satisfactory drying of the seeds for oil extraction and/or for in situ biodiesel production.
References
Demirbas A, Bafail A, Ahmad W, Sheikh M (2016) Biodiesel production from non-edible plant oils. Energy Explor Exploit 34(2):290–318. https://doi.org/10.1177/0144598716630166
Bhuiya M, Rasul M, Khan M, Ashwath N, Azad A (2016) Prospects of 2nd generation biodiesel as a sustainable fuel—Part: 1 selection of feedstocks, oil extraction techniques and conversion technologies. Renew Sustain Energy Rev 55:1109–1128. https://doi.org/10.1016/j.rser.2015.09.086
Sajjadi B, Raman AAA, Arandiyan H (2016) A comprehensive review on properties of edible and non-edible vegetable oil-based biodiesel: composition, specifications and prediction models. Renew Sustain Energy Rev 63:62–92. https://doi.org/10.1016/j.rser.2016.05.035
Atabani AE, Silitonga AS, Badruddin IA, Mahlia T, Masjuki H, Mekhilef S (2012) A comprehensive review on biodiesel as an alternative energy resource and its characteristics. Renew Sustain Energy Rev 16(4):2070–2093. https://doi.org/10.1016/j.rser.2012.01.003
Ajala AS, Adeleke SA (2014) Effect of drying temperatures on physicochemical properties and oil yield of African star apple (Chrysophyllum albidum) seeds. GJEDT 3(3):12–16
Ahmad A, Yasin NM, Derek C, Lim J (2011) Microalgae as a sustainable energy source for biodiesel production: a review. Renew Sustain Energy Rev 15(1):584–593. https://doi.org/10.1016/j.rser.2010.09.018
Shikha K, Chauhan YR (2012) Biodiesel production from non edible-oils: a review. J Chem Pharm Res 4(9):4219–4230
Supardan MD, Fahrizal RM, Moulana R, Safrida D, Satriana, Mustapha WAW (2017) Optimisation of process parameter conditions for biodiesel production by reactive extraction of jatropha seeds. JESTEC 12(3):847–859
Chhetri AB, Tango MS, Budge SM, Watts KC, Islam MR (2008) Non-edible plant oils as new sources for biodiesel production. Int J Mol Sci 9(2):169–180
He Y, Peng T, Guo Y, Li S, Guo Y, Tang L, Chen F (2017) Nontoxic oil preparation from Jatropha curcas L. seeds by an optimized methanol/n-hexane sequential extraction method. Ind Crops Prod 97:308–315. https://doi.org/10.1016/j.indcrop.2016.12.034
Amkul K, Laosatit K, Somta P, Shim S, Lee S-H, Tanya P, Srinives P (2017) Mapping of QTLs for seed phorbol esters, a toxic chemical in Jatropha curcas (L.). Genes 8(8):205. https://doi.org/10.3390/genes8080205
Brittaine R, Lutaladio N (2010) Jatropha: a smallholder bioenergy crop: the potential for pro-poor development, vol 8. FAO, Rome
Lang A, Farouk H (2013) Jatropha oil production for biodiesel and other products—a study of issues involved in production at large scale. Aeronautical Research Centre, Khartoum, Sudan; World Bioenergy Association, Stockholm, Sweden
Almeida FdA, Morais AMd, Carvalho JM, de Gouveia JP (2002) Cryoconservation of nordestina and pernambucana varieties of castor bean seeds. Rev Bras Eng Agr Amb 6(2):295–302
Allah FUM (2015) Determining the moisture content of Jatropha seeds, iodine vale and peroxide value of Jatropha oil. Ann Valahia Univ Târgovişte Geogr Ser 15(1):68–74
Soltani M, Takavar A, Alimardani R (2014) Moisture content determination of oilseeds based on dielectric measurement. Agric Eng Int CIGR J 16(1):313–318
Silva LJd, Dias DCFdS, Milagres CdC, Dias LAdS (2012) Relationship between fruit maturation stage and physiological quality of physic nut (Jatropha curcas L.) seeds. Ciênc Agrotec 36(1):39–44
Uthman F, Onifade T (2016) Effects of drying conditions on fuel property of physic nut (Jatropha Curcas). ASRJETS 18(1):53–66
Onifade T, Jekayinfa S, Uthman F Exergy analysis of thin layer drying of physic nuts (Jatropha curcas) using response surface methodologyy. In: Proceedings of the OAU, faculty of technology conference. pp 321–328. http://www.oautekconf.org/proceedings/47Onifade%20et%20al.pdf Accessed 28 June 2019
Lawson OSOA, Ologunagba FO, Ojomo AO (2010) Evaluation of the parameters affecting the solvent extraction of soybean oil. ARPN J Eng Appl Sci 5(10):51–55
Nagaraj G (2009) Oilseeds: properties, processing, products and procedures. New India Publishing, New Delhi
Willems P, Kuipers N, De Haan A (2008) Hydraulic pressing of oilseeds: experimental determination and modeling of yield and pressing rates. J Food Eng 89(1):8–16. https://doi.org/10.1016/j.jfoodeng.2008.03.023
Kabutey A, Herak D, Hanus J (2010) Screw press performance for oil extraction from Jatropha curcas L. seeds of different moisture content. Sci Agric Bohem 41(4):225–230
Subroto E (2015) Optimization of Jatropha curcas pure plant oil production. PhD thesis. University of Groningen, Netherlands. https://www.rug.nl/research/portal/files/23864987/Complete_thesis.pdf.Accessed 28 June 2019
Fadhlullah M, Widiyanto SNB, Restiawaty E (2015) The potential of nyamplung (Calophyllum inophyllum L.) seed oil as biodiesel feedstock: effect of seed moisture content and particle size on oil yield. Energy Procedia 68:177–185. https://doi.org/10.1016/j.egypro.2015.03.246
Brlek Savić T, Krička T, Voća N, Jurišić V, Matin A (2009) Effect of storage temperature on rapeseed quality. Agric Conspec Sci (ACS) 74(3):143–147
Akowuah J, Addo A, Kemausuor F (2012) Influence of storage duration of Jatropha curcas seed on oil yield and free fatty acid content. ARPN J Agric Biol Sci 7(1):41–45
Kombe GG, Temu AK, Rajabu HM, Mrema GD (2012) High free fatty acid (FFA) feedstock pre-treatment method for biodiesel production. In: Kombe GG et al. (eds) High free fatty acid (FFA) feedstock pre-treatment method for biodiesel production. Proc 2nd Int Conf Adv Eng Technol. pp 176–182
Berchmans HJ, Hirata S (2008) Biodiesel production from crude Jatropha curcas L. seed oil with a high content of free fatty acids. Bioresour Technol 99(6):1716–1721. https://doi.org/10.1016/j.biortech.2007.03.051
Traoré S, Magassouba S, Camara M (2016) Characteristics of Jatropha oil and prospective for its valorization as feedstock for the development of biodiesel technology in Guinea. Int Rev App Sci 3(1):1–11. https://doi.org/10.20448/journal.513/2016.3.1/513.1.1.11
Lim S, Hoong SS, Teong LK, Bhatia S (2010) Supercritical fluid reactive extraction of Jatropha curcas L. seeds with methanol: a novel biodiesel production method. Bioresour Technol 101(18):7169–7172. https://doi.org/10.1016/j.biortech.2010.03.134
Kadry GA (2015) Biodiesel production from Jatropha seeds. Am J Chem Eng 3(6):89–98. https://doi.org/10.11648/j.ajche.20150306.13
Azhari FM, Yunus R, Ghazi TM, Yaw T (2008) Reduction of free fatty acids in crude Jatropha curcas oil via an esterification process. Int J Eng Technol 5(2):92–98. https://doi.org/10.1016/j.jfoodeng.2008.03.023
Go AW, Sutanto S, Ong LK, Tran-Nguyen PL, Ismadji S, Ju Y-H (2016) Developments in in situ (trans) esterification for biodiesel production: a critical review. Renew Sustain Energy Rev 60:284–305. https://doi.org/10.1016/j.rser.2016.01.070
Hawash S, Kader EA, El Diwani G (2015) Biodiesel production from Jatropha seeds using heterogeneous integrated extraction reaction process. Afinidad 72(570):151–154
Kartika IA, Yani M, Ariono D, Evon P, Rigal L (2013) Biodiesel production from Jatropha seeds: solvent extraction and in situ transesterification in a single step. Fuel 106:111–117. https://doi.org/10.1016/j.fuel.2013.01.021
Qian J, Wang F, Liu S, Yun Z (2008) In situ alkaline transesterification of cottonseed oil for production of biodiesel and nontoxic cottonseed meal. Bioresour Technol 99(18):9009–9012. https://doi.org/10.1016/j.biortech.2008.04.059
Supardan MD, Satriana S, Fahrizal F, Moulana R (2013) Reactive extraction of Jatropha seed for biodiesel production: effect of moisture content of Jatropha seed and co-solvent concentration. Int J Adv Sci Eng Inf Technol 3(3):218–221
Haas MJ, Scott KM (2007) Moisture removal substantially improves the efficiency of in situ biodiesel production from soybeans. J Am Oil Chem Soc 84(2):197–204. https://doi.org/10.1007/s11746-006-1024-2
Avhad MR, Marchetti JM (2015) Temperature and pretreatment effects on the drying of Hass avocado seeds. Biomass Bioenergy 83:467–473. https://doi.org/10.1016/j.biombioe.2015.10.028
Zavala-Hernández J, Córdova-Téllez L, Martínez-Herrera J, Molina-Moreno J (2015) Physiological quality and chemical profile of developing Jatropha curcas seeds. Seed Sci Technol 43(3):433–444. https://doi.org/10.15258/sst.2015.43.3.12
Santoso BB, Budianto A, Aryana I (2012) Seed viability of Jatropha curcas in different fruit maturity stages after storage. Nusant Biosci 4(3):113–117. https://doi.org/10.13057/nusbiosci/n040305
Keneni YG, Hvoslef-Eide AK, Marchetti JM (2019) Mathematical modelling of the drying kinetics of Jatropha curcas L. seeds. Ind Crops Prod 132:12–20. https://doi.org/10.1016/j.indcrop.2019.02.012
Ramashia S, Gwata E, Meddows-Taylor S, Anyasi T, Jideani A (2018) Some physical and functional properties of finger millet (Eleusine coracana) obtained in sub-Saharan Africa. Food Res Int 104:110–118. https://doi.org/10.1016/j.foodres.2017.09.065
Mansouri A, Mirzabe AH, Ráufi A (2017) Physical properties and mathematical modeling of melon (Cucumis melo L.) seeds and kernels. J Saudi Soc Agric Sci 16(3):218–226. https://doi.org/10.1016/j.jssas.2015.07.001
Perea-Flores M, Garibay-Febles V, Chanona-Perez JJ, Calderon-Dominguez G, Mendez-Mendez JV, Palacios-González E, Gutierrez-Lopez GF (2012) Mathematical modelling of castor oil seeds (Ricinus communis) drying kinetics in fluidized bed at high temperatures. Ind Crops Prod 38:64–71. https://doi.org/10.1016/j.indcrop.2012.01.008
Kashaninejad M, Mortazavi A, Safekordi A, Tabil LG (2007) Thin-layer drying characteristics and modeling of pistachio nuts. J Food Eng 78(1):98–108. https://doi.org/10.1016/j.jfoodeng.2005.09.007
Siqueira VC, Resende O, Chaves TH (2012) Drying kinetics of Jatropha seeds. Revista Ceres 59(2):171–177
Bamgboye IA, Adebayo SE (2012) Seed moisture dependent on physical and mechanical properties of Jatropha curcas. J Agric Tech 8(1):13–26
Mousa A, Elkaoud NS, Dosoky SH (2016) Physical, mechanical and aerodynamic properties of Jatropha seeds. Misr J Ag Eng 33(4):1477–1496
Rao NK, Hanson J, Dulloo ME, Ghosh K, Nowell A (2006) Manual of seed handling in genebanks, vol 8. Bioversity International, Rome
Kapseu C, Bup DN, Tchiegang C, Abi C, Broto F, Parmentier M (2007) Effect of particle size and drying temperature on drying rate and oil extracted yields of Buccholzia coriacea (MVAN) and Butyrospermum parkii ENGL. Int J Food Sci Technol 42(5):573–578. https://doi.org/10.1111/j.1365-2621.2006.01277.x
Onwuka UN, Nwachukwu G (2013) Grain size and heat source effect on the drying profile of cocoa beans. Niger J Technol 32(3):417–423
Pradhan RC, Meda V, Naik SN, Tabil L (2010) Physical properties of Canadian grown flaxseed in relation to its processing. Int J Food Prop 13(4):732–743. https://doi.org/10.1080/10942910902818137
Acknowledgements
The authors would like to express their gratitude to the EnPe/NORHED Project of Norad at the Norwegian University of Life Sciences, Faculty of Sciences and Technology for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Keneni, Y.G., Marchetti, J.M. Temperature and pretreatment effects on the drying of different collections of Jatropha curcas L. seeds. SN Appl. Sci. 1, 943 (2019). https://doi.org/10.1007/s42452-019-0969-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-019-0969-3




 ) 313 K, (
) 313 K, (
 ) 323 K, (
) 323 K, (
 ) 333 K, (
) 333 K, (
 ) 343 K and (
) 343 K and (
 ) 353 K
) 353 K

 ) Salmene2, (
) Salmene2, (
 ) Bira, (
) Bira, (
 ) Kurkura, (
) Kurkura, (
 ) Salmene3, (
) Salmene3, (
 ) Shekla, (
) Shekla, (
 ) Salmene1 and (
) Salmene1 and (