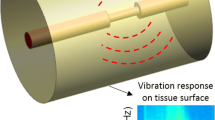
Abstract
Purpose
Intracranial stenosis is a critical cardiovascular disorder which may lead to recurrent ischemic stroke. The flow in a stenosed artery tends to have a chaotic pattern, resulting in fluctuating pressures on the artery wall. These acoustic pressures are known as the sources of arterial noise and vibration. The generated vibro-acoustic signals on the stenosed artery propagate through the surrounding body tissues and reach the epidermal surface. In this study, the stenosis-induced vibration on the epidermal surface is aimed to be utilized for the diagnosis of occlusion in the intracranial artery.
Methods
An intracranial stenosis is modeled considering a simplified head geometry consisted of brain, skull, and skin. The simplified head model has an idealized spherical form. Three different stenosis severities are employed to elucidate the effects of mild, moderate, and severe intracranial stenosis. ADINA finite element analysis software package is used to perform harmonic and modal analyses to determine the biodynamic responses on the epidermal surface of a human head.
Results
According to the results obtained, vibration amplitudes on the epidermal surface tend to increase with increasing stenosis severity. The stenosis-induced effects on the vibration responses are particularly prominent in the frequency range of 400–1000 Hz. Abnormal increase in vibration amplitudes between 400 and 1000 Hz can be an indicator of further occlusion in the intracranial artery.
Conclusions
The stenosis-induced vibro-acoustic effects provide important clues about arterial health. Excessive vibration amplitudes indicate a potential arterial occlusion and can be used as an early diagnostic tool for intracranial stenosis detection.











Similar content being viewed by others
References
Krasteva MP, Lau KK, Mordasini P et al (2020) Intracranial atherosclerotic stenoses: pathophysiology, epidemiology, risk factors and current therapy options. Adv Ther 37:1829–1865
Gutierrez J, Turan TN, Hoh BL, Chimowitz MI (2022) Intracranial atherosclerotic stenosis: risk factors, diagnosis, and treatment. Lancet Neurol 21:355–368
Suri MFK, Qiao Y, Ma X et al (2016) Prevalence of intracranial atherosclerotic stenosis using high-resolution magnetic resonance angiography in the general population. Stroke 47:1187–1193
Turan TN, Makki AA, Tsappidi S et al (2010) Risk factors associated with severity and location of intracranial arterial stenosis. Stroke 41:1636–1640
Chaturvedi S, Turan TN, Lynn MJ et al (2007) Risk factor status and vascular events in patients with symptomatic intracranial stenosis. Neurology 69:2063–2068
Sacco RL, Kargman DE, Gu Q, Zamanillo MC (1995) Race-ethnicity and determinants of intracranial atherosclerotic cerebral infarction. Stroke 26:14–20
Mendelson SJ, Prabhakaran S (2021) Diagnosis and management of transient ischemic attack and acute ischemic stroke: a review. JAMA 325:1088–1098
von Sarnowski B, Schminke U, Tatlisumak T et al (2013) Prevalence of stenoses and occlusions of brain-supplying arteries in young stroke patients. Neurology 80:1287–1294
Wang Y, Zhao X, Liu L et al (2014) Prevalence and outcomes of symptomatic intracranial large artery stenoses and occlusions in China. Stroke 45:663–669
Dearborn JL, Zhang Y, Qiao Y et al (2017) Intracranial atherosclerosis and dementia. Neurology 88:1556–1563
Laviña B (2017) Brain vascular imaging techniques. Int J Mol Sci 18:70
Liang B, Wang S, Shen F et al (2021) Acoustic impact of the human skull on transcranial photoacoustic imaging. Biomed Opt Express 12:1512–1528
Lees RS, Dewey CF (1970) Phonoangiography: A new noninvasive diagnostic method for studying arterial disease. Proc Natl Acad Sci 67:935–942
Salman HE, Yazicioglu Y (2019) Computational analysis for non-invasive detection of stenosis in peripheral arteries. Med Eng Phys 70:39–50
Faragli A, Abawi D, Quinn C et al (2021) The role of non-invasive devices for the telemonitoring of heart failure patients. Heart Fail Rev 26:1063–1080
Guala A, Camporeale C, Ridolfi L, Mesin L (2017) Non-invasive aortic systolic pressure and pulse wave velocity estimation in a primary care setting: An in silico study. Med Eng Phys 42:91–98
Jeger-Madiot N, Gateau J, Fink M, Ing R-K (2017) Non-contact and through-clothing measurement of the heart rate using ultrasound vibrocardiography. Med Eng Phys 50:96–102
Borisyuk AO (2003) Experimental study of wall pressure fluctuations in a pipe behind a stenosis. Int J Fluid Mech Res 30:264–278
Yazicioglu Y, Royston TJ, Spohnholtz T et al (2005) Acoustic radiation from a fluid-filled, subsurface vascular tube with internal turbulent flow due to a constriction. J Acoust Soc Am 118:1193–1209
Salman HE, Sert C, Yazicioglu Y (2013) Computational analysis of high frequency fluid–structure interactions in constricted flow. Comput Struct 122:145–154
Ozden K, Sert C, Yazicioglu Y (2019) Numerical investigation of wall pressure fluctuations downstream of concentric and eccentric blunt stenosis models. Proc Inst Mech Eng 234:48–60
Ozden K, Sert C, Yazicioglu Y (2020) Effect of stenosis shape on the sound emitted from a constricted blood vessel. Med Biol Eng Comput 58:643–658
Khalili F, Gamage PT, Taebi A et al (2021) Spectral decomposition of the flow and characterization of the sound signals through stenoses with different levels of severity. Bioengineering 8:41
Lee SE, Lee S-W, Fischer PF et al (2008) Direct numerical simulation of transitional flow in a stenosed carotid bifurcation. J Biomech 41:2551–2561
Saqr KM, Kano K, Rashad S et al (2022) Non-Kolmogorov turbulence in carotid artery stenosis and the impact of carotid stenting on near-wall turbulence. AIP Adv 12:015124
Tobin RJ, Chang ID (1976) Wall pressure spectra scaling downstream of stenoses in steady tube flow. J Biomech 9:633–640
Salman HE, Yazicioglu Y (2019) Experimental and numerical investigation on soft tissue dynamic response due to turbulence-induced arterial vibration. Med Biol Eng Compu 57:1737–1752
Salman HE, Yazicioglu Y (2017) Flow-induced vibration analysis of constricted artery models with surrounding soft tissue. J Acoust Soc Am 142:1913–1925
Khalili F, Gamage PT, Taebi A et al (2021) Spectral decomposition and sound source localization of highly disturbed flow through a severe arterial stenosis. Bioengineering 8:34
Borisyuk AO (2002) Modeling of noise generation by a vascular stenosis. Int J Fluid Mech Res 29:65–88
Cao DY, Zhang YF, Wang YQ (2022) Wave propagation characteristics in submerged pipes conveying viscous flowing fluid based on the shear deformation shell theory. J Vib Eng Technol 10:827–839
Borisyuk AO (1998) Noise field in the human chest due to turbulent flow in a larger blood vessel. Flow Turbul Combust 61:269–284
Seo JH, Mittal R (2012) A coupled flow-acoustic computational study of bruits from a modeled stenosed artery. Med Biol Eng Comput 50:1025–1035
Guruguntla V, Lal M (2021) An improved biomechanical model to optimize biodynamic responses under vibrating medium. J Vib Eng Technol 9:675–685
Banks HT, Hu S, Kenz ZR et al (2014) Model validation for a noninvasive arterial stenosis detection problem. Math Biosci Eng 11:427–448
Azimpour F, Caldwell E, Tawfik P et al (2016) Audible coronary artery stenosis. Am J Med 129:515–521
Semmlow J, Rahalkar K (2007) Acoustic detection of coronary artery disease. Annu Rev Biomed Eng 9:449–469
Owsley NL, Hull AJ (1998) Beamformed nearfield imaging of a simulated coronary artery containing a stenosis. IEEE Trans Med Imaging 17:900–909
Olson LG, Bathe K-J (1985) Analysis of fluid-structure interactions. A direct symmetric coupled formulation based on the fluid velocity potential. Comput Struct 21:21–32
Sussman T, Sundqvist J (2003) Fluid–structure interaction analysis with a subsonic potential-based fluid formulation. Comput Struct 81:949–962
ADINA R&D Inc (2002) Theory and modeling guide Vol. 1: ADINA. In Report ARD 02‐7
Bathe K-J (1996) Finite element procedures. Prentice-Hall, New Jersey
Bartlett ES, Walters TD, Symons SP et al (2008) Classification of carotid stenosis by millimeter CT angiography measures: effects of prevalence and gender. Am J Neuroradiol 29:1677–1683
Alvarez VS, Halldin P, Kleiven S (2014) The influence of neck muscle tonus and posture on brain tissue strain in pedestrian head impacts. Stapp Car Crash Journal 58:63–101
Ganpule S, Daphalapurkar NP, Cetingul MP, Ramesh KT (2018) Effect of bulk modulus on deformation of the brain under rotational accelerations. Shock Waves 28:127–139
Lillie EM, Urban JE, Lynch SK et al (2016) Evaluation of skull cortical thickness changes with age and sex from computed tomography scans. J Bone Miner Res 31:299–307
Falland-Cheung L, Waddell JN, Chun LK et al (2017) Investigation of the elastic modulus, tensile and flexural strength of five skull simulant materials for impact testing of a forensic skin/skull/brain model. J Mech Behav Biomed Mater 68:303–307
Evans SL, Holt CA (2009) Measuring the mechanical properties of human skin in vivo using digital image correlation and finite element modelling. J Strain Anal Eng Des 44:337–345
El-Barhoun EN, Gledhill SR, Pitman AG (2009) Circle of Willis artery diameters on MR angiography: An Australian reference database. J Med Imaging Radiat Oncol 53:248–260
Zhu G, Yuan Q, Yang J, Yeo JH (2015) Experimental study of hemodynamics in the circle of Willis. Biomed Eng Online 14:1–15
Er U, Fraser K, Lanzino G (2008) The anterior spinal artery origin: a microanatomical study. Spinal Cord 46:45–49
Liu H, Canton G, Yuan C et al (2012) Using in vivo cine and 3D multi-contrast MRI to determine human atherosclerotic carotid artery material properties and circumferential shrinkage rate and their impact on stress/strain predictions. J Biomech Eng 134:011008
Farhoudi M, Kermani SN, Sadeghi-Bazargani H (2010) Relatively higher norms of blood flow velocity of major intracranial arteries in North-West Iran. BMC Res Notes 3:1–5
Celik IB, Ghia U, Roache PJ, Freitas CJ (2008) Procedure for estimation and reporting of uncertainty due to discretization in CFD applications. J Fluids Eng-Trans ASME 130:078001
Saengsuwan J, Suangpho P (2019) Self-perceived and actual risk of further stroke in patients with recurrent stroke or recurrent transient ischemic attack in Thailand. J Stroke Cerebrovasc Dis 28:632–639
Yang B, Shi Z, Wang Q et al (2017) Frequency spectrum of the human head–neck to mechanical vibrations. J Low Freq Noise Vib Active Control 37:611–618
Sun D, Yan B, Han B et al (2016) Vibration characteristic simulation of a pneumatic artificial muscle damping seat. J Low Freq Noise Vib Active Control 35:39–51
Gupta S, Haiat G, Laporte C, Belanger P (2021) Effect of the acoustic impedance mismatch at the bone-soft tissue interface as a function of frequency in transcranial ultrasound: A simulation and in vitro experimental study. IEEE Trans Ultrason Ferroelectr Freq Control 68:1653–1663
Acknowledgements
This study is funded by TÜBİTAK (The Scientific and Technological Research Council of Türkiye) 3501—Career Development Program (Project number: 221M001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Salman, H.E. Computational Modeling and Investigation of the Vibro-Acoustic Effects Induced by Intracranial Stenosis in a Simplified Head Model. J. Vib. Eng. Technol. 11, 1973–1986 (2023). https://doi.org/10.1007/s42417-022-00682-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42417-022-00682-x