Abstract
Purpose
Dopamine agonists (DA) comprise first-line treatment for prolactinomas. However, some patients show no response to DA and are considered resistant. In this study, we retrospectively analyzed the outcomes of DA-resistant prolactinoma patients after transsphenoidal surgery (TSS).
Methods
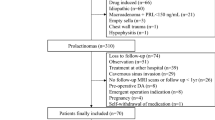
A total of 94 consecutive patients with DA-resistant prolactinomas who underwent TSS were retrospectively enrolled in the present study. Early postoperative remission rate, prolactin (PRL) levels, and recurrence rate were analyzed.
Results
Of 94 DA-resistant patients, 47 (50%) achieved early remission 1 week post-surgery, including 41.18% of macroprolactinoma patients and 73.08% of microprolactinoma patients. PRL levels on the first postoperative day were significantly lower than preoperative PRL levels (p < 0.001). Total resection rate in macro- and microprolactinomas were, respectively, 75 and 96.15%. A recurrence of hyperprolactinemia and tumor was, respectively, found in 31.91 and 19.15% of patients with a follow-up of 39.53 ± 2.172 months (range 3–86). A higher hyperprolactinemia recurrence was observed in patients with invasive prolactinomas (p = 0.021) or preoperative PRL levels ≥ 200 ng/ml (p = 0.029). Univariate logistic regression indicated that larger maximum tumor diameter (p = 0.045), invasive prolactinomas (p = 0.002), and absence of early postoperative remission (p = 0.004) were significant predictors of tumor recurrence. However, using multivariate stepwise logistic regression, only invasiveness and early postoperative remission remained significant.
Conclusion
Tumor invasiveness and preoperative PRL levels were significant predictors of hyperprolactinemia recurrence after TSS. For tumor recurrence, invasiveness and early postoperative remission were independent predictors.


Similar content being viewed by others
Data availability
Data are available on request due to local (academic) restrictions.
References
Amar AP, Couldwell WT, Chen JC, Weiss MH (2002) Predictive value of serum prolactin levels measured immediately after transsphenoidal surgery. J Neurosurg 97(2):307–314. https://doi.org/10.3171/jns.2002.97.2.0307
Babey M, Sahli R, Vajtai I et al (2011) Pituitary surgery for small prolactinomas as an alternative to treatment with dopamine agonists. Pituitary 14(3):222–230. https://doi.org/10.1007/s11102-010-0283-y
Chanson P, Maiter D (2019) The epidemiology, diagnosis and treatment of Prolactinomas: the old and the new. Best Pract Res Clin Endocrinol Metab 33(2):101290. https://doi.org/10.1016/j.beem.2019.101290
Del Basso De Caro M, Solari D, Pagliuca F et al (2017) Atypical pituitary adenomas: clinical characteristics and role of ki-67 and p53 in prognostic and therapeutic evaluation. A series of 50 patients. Neurosurg Rev 40(1):105–114. https://doi.org/10.1007/s10143-017-0818-z
Donegan D, Atkinson JL, Jentoft M et al (2017) Surgical outcomes of prolactinomas in recent era: results of a heterogenous group. Endocr Pract 23(1):37–45. https://doi.org/10.4158/EP161446.OR
Gillam MP, Molitch ME, Lombardi G, Colao A (2006) Advances in the treatment of prolactinomas. Endocr Rev 27(5):485–534. https://doi.org/10.1210/er.2005-9998
Zielinski G, Ozdarski M, Maksymowicz M et al (2020) Prolactinomas: prognostic factors of early remission after transsphenoidal surgery. Front Endocrinol (Lausanne) 11:439. https://doi.org/10.3389/fendo.2020.00439
Glezer A, Bronstein MD (2015) Prolactinomas. Endocrinol Metab Clin North Am 44(1):71–78. https://doi.org/10.1016/j.ecl.2014.11.003
Grimm F, Maurus R, Beschorner R et al (2019) Ki-67 labeling index and expression of p53 are non-predictive for invasiveness and tumor size in functional and nonfunctional pituitary adenomas. Acta Neurochir (Wien) 161(6):1149–1156. https://doi.org/10.1007/s00701-019-03879-4
Han YL, Chen DM, Zhang C et al (2018) Retrospective analysis of 52 patients with prolactinomas following endoscopic endonasal transsphenoidal surgery. Medicine (Baltimore) 97(45):e13198. https://doi.org/10.1097/MD.0000000000013198
Knosp E, Steiner E, Kitz K, Matula C (1993) Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery 33(4):610–618. https://doi.org/10.1227/00006123-199310000-00008
Kreutzer J, Buslei R, Wallaschofski H et al (2008) Operative treatment of prolactinomas: indications and results in a current consecutive series of 212 patients. Eur J Endocrinol 158(1):11–18. https://doi.org/10.1530/EJE-07-0248
Primeau V, Raftopoulos C, Maiter D (2012) Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol 166(5):779–786. https://doi.org/10.1530/EJE-11-1000
Gerges MM, Rumalla K, Godil SS et al (2020) Long-term outcomes after endoscopic endonasal surgery for nonfunctioning pituitary macroadenomas. J Neurosurg 31:1–12. https://doi.org/10.3171/2019.11.JNS192457
Souteiro P, Belo S, Carvalho D (2020) Dopamine agonists in prolactinomas: when to withdraw? Pituitary 23(1):8–44. https://doi.org/10.1007/s11102-019-00989-1
Panigrahi MK, Chandrasekhar YBVK (2020) Vooturi S (2020) Current status of surgery in management of prolactinomas. Neurol India 68(Supplement):S39–S43. https://doi.org/10.4103/0028-3886.287668
Vilar L, Vilar CF, Lyra R, Freitas MDC (2019) Pitfalls in the diagnostic evaluation of hyperprolactinemia. Neuroendocrinology 109(1):7–19. https://doi.org/10.1159/000499694
Hamilton DK, Vance ML, Boulos PT, Laws ER (2005) Surgical outcomes in hyporesponsive prolactinomas: analysis of patients with resistance or intolerance to dopamine agonists. Pituitary 8(1):53–60. https://doi.org/10.1007/s11102-005-5086-1
Smith TR, Hulou MM, Huang KT et al (2015) Current indications for the surgical treatment of prolactinomas. J Clin Neurosci 22(11):1785–1791. https://doi.org/10.1016/j.jocn.2015.06.001
Oh MC, Aghi MK (2011) Dopamine agonist-resistant prolactinomas. J Neurosurg 114(5):1369–1379. https://doi.org/10.3171/2010.11.JNS101369
Raverot G, Wierinckx A, Dantony E et al (2010) Prognostic factors in prolactin pituitary tumors: clinical, histological, and molecular data from a series of 94 patients with a long postoperative follow-up. J Clin Endocrinol Metab 95(4):1708–1716. https://doi.org/10.1210/jc.2009-1191
Tyrrell JB, Lamborn KR, Hannegan LT et al (1999) Transsphenoidal microsurgical therapy of prolactinomas: initial outcomes and long-term results. Neurosurgery 44(2):254–263. https://doi.org/10.1097/00006123-199902000-00006
Maiter D (2019) Management of dopamine agonist-resistant prolactinoma. Neuroendocrinology 109(1):42–50. https://doi.org/10.1159/000495775
Donoho DA, Laws ER Jr (2019) The role of surgery in the management of prolactinomas. Neurosurg Clin N Am 30(4):509–514. https://doi.org/10.1016/j.nec.2019.05.010
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was approved by Ethics Committee of the Second People’s Hospital of Qinzhou.
Consent to participate
Informed consent was signed by all patients or their guardians.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wei, L., Wei, X. Outcomes of transsphenoidal surgery in dopamine agonist-resistant prolactinomas: a retrospective study. Hormones 20, 745–752 (2021). https://doi.org/10.1007/s42000-021-00309-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-021-00309-y