Abstract
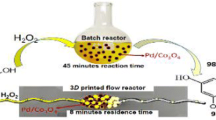
Selective oxidation of hydroxymethylfurfural to diformylfuran was performed in 3D-printed milli-scale porous reactors using pure oxygen in mild operating conditions (T = 60° C, P = 1 atm) and the homogeneous TEMPO/TBN catalytic system. Three different configurations were tested, where a rotation (θ = 22.5°) and/or an inclination (φ = 45°) of the fibers are introduced. An empty tube and a packed bed were also tested as a reference. Out of these designs, the reactor with both parameters varied simultaneously (INSP1) exhibited the highest performance, achieving an efficiency of up to 80%. The maximum conversion of 18.2% was attained for a residence time of 160 s, despite existing mass transfer limitations for this flow rate. The selectivity to DFF was 100% for all the 3D-printed reactors. On the contrary, the packed bed resulted in the highest efficiency, but at the expense of selectivity. Additional oxidation products have been retained in the packing, blocking thus the packed bed after a few hours of operation. The kinetic constant was found based on a (0,1)-order kinetic model from batch experiments. The kinetic information was utilized to evaluate the performance of the 3D-printed porous reactors from a mass transfer and reaction engineering aspect. The 3D-printed reactors were operating almost in kinetic control for total flow rates above 1 mL/min (Ha < 0.3). However, the associated short residence time resulted in small conversion. The 3D-printed reactors show significant potential when operating at higher flow rates. The low conversions can be countered by increasing the residence time, either with multiple passes or by operating them in series.
Highlights
-
Selective formation of DFF from HMF was achieved with the homogeneous, metal-free catalytic system TEMPO/TBN in mild conditions (T = 60° C, P = 1 atm)
-
3D-printed reactors have shown performance comparable to a packed bed achieving an efficiency of 80% at a smaller pressure drop, hence energy dissipation
-
For total flow rates higher than 1 mL/min, higher interfacial areas and mixing resulted in the operation being almost under kinetic control (Ha < 0.3)









Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Gavriilidis A, Constantinou A, Hellgardt K, Hii KK, Hutchings GJ, Brett GL, Kuhn S, Marsden SP (2016) Aerobic oxidations in flow: opportunities for the fine chemicals and pharmaceuticals industries. React Chem Eng 1:595–612. https://doi.org/10.1039/c6re00155f
Stahl SS, Alsters PL (eds) (2016) Liquid phase aerobic oxidation catalysis: industrial applications and academic perspectives. Wiley, New York
van Putten R, van der Waal JC, de Jong E, Rasrenda C, Heeres H, de Vries JG (2013) Hydroxymethylfurfural, a versatile platform chemical made from renewable resources. Chem Rev 113(3):1499–1597
Dai J (2021) Synthesis of 2,5-diformylfuran from renewable carbohydrates and its applications: A review. Green Energy Environ 6(1):22–32. https://doi.org/10.1016/j.gee.2020.06.013
Pal P, Saravanamuguran S (2019) Recent advances in the development of 5-hydroxymethylfurfural oxidation with base (nonprecious)-metal-containing catalysts. ChemSusChem 12(1):145–163. https://doi.org/10.1002/cssc.201801744
Sajid M, Zhao X, Liu D (2018) Production of 2,5-furandicarboxylic acid (FDCA) from 5-hydroxymethylfurfural (HMF): recent progress focusing on the chemical-catalytic routes. Green Chem 20:5427–5453. https://doi.org/10.1039/C8GC02680G
Martin T (1996) Verfahren zum selektiven Herstellen von Furandicarboxaldehyd-2,5 ausgehend von Hydroxymethyl-5-Furancarboxaldehyd 2. Patent Number: DE19615878A1, Germany
Sha J, Kusema BT, Zhou WJ, Yan Z, Streiff S, Pera-Titus M (2021) Single-reactor tandem oxidation–amination process for the synthesis of furan diamines from 5-hydroxymethylfurfural. Green Chem 23:7093–7099. https://doi.org/10.1039/d1gc01621k
Navarro OC, Canos AC, Chornet SI (2009) Chemicals from biomass: aerobic oxidation of 5-hydroxymethyl-2-furaldehyde into diformylfurane catalyzed by immobilized vanadyl-pyridine complexes on polymeric and organofunctionalized mesoporous supports. Top Catal 52:304–314. https://doi.org/10.1007/s11244-008-9153-5
Dimitratos N, Lopez-Sanchez JA, Hutchings GJ (2011) Selective liquid phase oxidation with supported metal nanoparticles. Chem Sci 3:20–44. https://doi.org/10.1039/C1SC00524C
He H, Shen Z, Mo W, Sun N, Hu B, Hu X (2009) TEMPO-tert-butyl nitrite: An efficient catalytic system for aerobic oxidation of alcohols. Adv Synth Catal 351(1–2):89–92. https://doi.org/10.1002/adsc.200800501
Hommes A, Disselhorst B, Janssens HMM, Stevelink RJA, Heeres HJ, Yue J (2021) Mass transfer and reaction characteristics of homogeneously catalyzed aerobic oxidation of 5-hydroxymethylfurfural in slug flow microreactors. Chem Eng J 413:127552. https://doi.org/10.1016/j.cej.2020.127552
Hommes A, Heeres HJ, Yue J (2019) Catalytic transformation of biomass derivatives to valueadded chemicals and fuels in continuous flow microreactors. ChemCatChem 11(19):4671–4708. https://doi.org/10.1002/cctc.201900807
Aellig C, Scholz D, Conrad S, Hermans I (2013) Intensification of TEMPO-mediated aerobic alcohol oxidations under three-phase flow conditions. Green Chem 15:1975–1980. https://doi.org/10.1039/c3gc40159f
Zuo X, Chaudhari AS, Snavely K, Niu F, Zhu H, Martin KJ, Subramaniam B (2017) Kinetics of homogeneous 5-hydroxymethylfurfural oxidation to 2,5-furandicarboxylic acid with Co/Mn/Br catalyst. AIChE J 63(1):162–171. https://doi.org/10.1002/aic.15497
Jeong G-Y, Singh A, Sharma S, Gyak KW, Maurya RA, Kim D-P (2015) One-flow syntheses of diverse heterocyclic furan chemicals directly from fructose via tandem transformation platform. NPG Asia Mater 7:e173. https://doi.org/10.1038/am.2015.21
Potdar A, Thomassen LCJ, Kuhn S (2019) Structured porous millireactors for liquid-liquid chemical reactions. Chem Ing Tech 91(5):592–601. https://doi.org/10.1002/cite.201800128
Potdar A, Thomassen LCJ, Kuhn S (2019) Scalability of 3D printed structured porous milli-scale reactors. Chem Eng J 363:337–348. https://doi.org/10.1016/j.cej.2019.01.082
Parra-Cabrera C, Achille C, Kuhn S, Ameloot R (2018) 3D printing in chemical engineering and catalytic technology: structured catalysts, mixers and reactors. Chem Soc Rev 47(1):209–230. https://doi.org/10.1039/C7CS00631D
Hornung CH, Nguyen X, Carafa A, Gardiner J, Urban A, Fraser D, Horne MD, Gunasegaram DR, Tsanaktsidis J (2017) Use of catalytic static mixers for continuous flow gas-liquid and transfer hydrogenations in organic synthesis. Org Process Res Dev 21(9):1311–1319. https://doi.org/10.1021/acs.oprd.7b00180
Elias Y, Rudolf von Rohr P, Bonrath W, Medlock J, Buss A (2015) A porous structured reactor for hydrogenation reactions. Chem Eng Process: Process Intensif 95:175–85. https://doi.org/10.1016/j.cep.2015.05.012
Maier MC, Valotta A, Hiebler K, Soritz S, Gavric K, Grabner B, Gruber-Woelfler H (2020) 3D printed reactors for synthesis of active pharmaceutical ingredients in continuous flow. Org Process Res Dev 24(10):2197–2207. https://doi.org/10.1021/acs.oprd.0c00228
Potdar A, Protasova LN, Thomassen LCJ, Kuhn S (2017) Designed porous milli-scale reactors with enhanced interfacial mass transfer in two-phase flows. React Chem Eng 2:137–148. https://doi.org/10.1039/C6RE00185H
Charpentier J-C (1981) Mass-transfer rates in gas-liquid absorbers and reactors. Adv Chem Eng 11:1–133. https://doi.org/10.1016/S0065-2377(08)60025-3
Lucile F, Cezac P, Contamine F, Serin JP, Houssin D, Arpentinier P (2012) Solubility of carbon dioxide in water and aqueous solution containing sodium hydroxide at temperatures from (293.15 to 393.15) K and pressure up to 5 MPa: Experimental measurements. J Chem Eng Data 57:784–789. https://doi.org/10.1021/je200991x
Yue J, Chen G, Yuan Q, Luo L, Gonthier Y (2007) Hydrodynamics and mass transfer characteristics in gas–liquid flowthrough a rectangular microchannel. Chem Eng Sci 62(7):2096–2108. https://doi.org/10.1016/j.ces.2006.12.057
Carberry JJ (1976) Chemical and catalytic reaction engineering. McGraw-Hill, New York
Hong M, Min J, Wu S, Cui H, Zhao Y, Li J, Wang S (2019) Metal nitrate catalysis for selective oxidation of 5-hydroxymethylfurfural into 2,5-diformylfuran under oxygen atmosphere. ACS Omega 4:7054–7060. https://doi.org/10.1021/acsomega.9b00391
Schmal M, Pinto JC (2021) Chemical reaction engineering: Parameter estimation, exercises and examples, 2nd edn. Taylor & Francis, London
Gupta AS, Thodos G (1962) Mass and Heat transfer through fixed and fluidized beds. Chem Eng Prog 58(7):608–610. https://doi.org/10.1002/aic.690080509
Saber M, Huu TT, Pham-Huu C, Edouard D (2012) Residence time distribution, axial liquid dispersion and dynamic–static liquid mass transfer in trickle flow reactor containing SiC open-cell foams. Chem Eng J 185–186:294–299. https://doi.org/10.1016/j.cej.2012.01.045
Tourvieille JN, Philippe R, de Bellefon C (2015) Milli-channel with metal foams under an applied gas–liquid periodic flow: External mass transfer performance and pressure drop. Chem Eng J 267:332–346. https://doi.org/10.1016/j.cej.2014.11.084
Serres M, Schweich D, Vidal V, Philippe R (2018) Liquid residence time distribution of multiphase horizontal flow in packed bed milli-channel: Spherical beads versus open cell solid foams. Chem Eng J 190:149–163. https://doi.org/10.1016/j.ces.2018.05.004
Gao H, Tatomir AB, Karadimitriou NK, Steeb H, Sauter M (2023) Effect of pore space stagnant zones on interphase mass transfer in porous media, for two-phase flow conditions. Transp Porous Media 146:639–667. https://doi.org/10.1007/s11242-022-01879-0
Kantarci N, Borak F, Ulgen KO (2005) Bubble column reactors. Process Biochem 40(7):2263–2283. https://doi.org/10.1016/j.procbio.2004.10.004
Al-Rifai N, Galvanin F, Morad M, Cao E, Cattaneo S, Sankar M, Dua V, Hutchings G, Gavriilidis A (2016) Hydrodynamic effects on three phase micro-packed bed reactor performance – gold–palladium catalysed benzyl alcohol oxidation. Chem Eng Sci 149:129–142. https://doi.org/10.1016/j.ces.2016.03.018
van Herk D, Kreitzer MT, Makke M, Moulijn JA (2005) Scaling down trickle bed reactors. Catal Today 106(1–4):227–232. https://doi.org/10.1016/j.cattod.2005.07.180
Sang L, Tu J, Cheng H, Luo G, Zhang J (2020) Hydrodynamics and mass transfer of gas–liquid flow in micropacked bed reactors with metal foam packing. AIChE J 66(2):e6803. https://doi.org/10.1002/aic.16803
Zhang J, Texeira AR, Jensen KF (2018) Automated measurements of gas-liquid mass transfer in micro-packed bed reactors. AIChE J 64(2):564–570. https://doi.org/10.1002/aic.15941
Galvanin F, Sankar M, Cattaneo S, Bethell D, Dua V, Hutchings GJ, Gavriilidis A (2018) On the development of kinetic models for solvent-free benzyl alcohol oxidation over a gold-palladium catalyst. Chem Eng J 342:196–210. https://doi.org/10.1016/j.cej.2017.11.165
Chen S, Cheng Y, Ban H, Zhang Y, Zheng L, Wang L, Li X (2020) Liquid-phase aerobic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid over Co/Mn/Br catalyst. Ind Eng Chem Res 59(39):17076–17084.
Angeli P, Gavriilidis A (2008) Hydrodynamics of Taylor flow in small channels: A review. J Mech Eng Sci 222(5):737–751. https://doi.org/10.1243/09544062JMES776
Thulasidas TC, Abraham MA, Cerro RL (1995) Bubble-train flow in capillaries of circular and square cross section. Chem Eng Sci 50(2):183–199. https://doi.org/10.1016/0009-2509(94)00225-G
Busciglio A, Grisafi F Scargiali F, Brucato A (2013) On the measurement of local gas hold-up, interfacial area and bubble size distribution in gas-liquid contactors via light sheet and image analysis: Imaging technique and experimental results. Chem Eng Sci 102:551–566. https://doi.org/10.1016/j.ces.2013.08.029
Calderbank PH (1958) Physical rate processes in industrial fermentations Part I: The interfacial area in gas-liquid contacting with mechanical agitation. Trans Inst Chem Eng 36:443–463
Laakkonen M, Honkanen M, Saarenrinne P, Aittamaa J (2005) Local bubble size distributions, gas–liquid interfacial areas and gas holdups in a stirred vessel with particle image velocimetry. Chem Eng J 109(1–3):37–47. https://doi.org/10.1016/j.cej.2005.03.002
Sang L, Cao Q, Xie B, Ma C, Zhang J (2021) Investigation of effective interfacial area in micropacked bed reactors. Ind Eng Chem Res 60:9206–9215. https://doi.org/10.1021/acs.iecr.1c00416
Voltolina S, Marín P, Diez FV, Ordóñez S (2017) Open-cell foams as beds in multiphase reactors: Residence time distribution and mass transfer. Chem Eng J 316:323–331. https://doi.org/10.1016/j.cej.2017.01.113
Li M, Niu F, Zuo X, Metelski PD, Busch DH, Subramaniam B (2013) A spray reactor concept for catalytic oxidation of p-xylene to produce high-purity terephthalic acid. Chem Eng Sci 104:93–102. https://doi.org/10.1016/j.ces.2013.09.004
Hikita H, Asai S (1964) Gas absorption with (m, n)-th order irreversible chemical reactions. Int Chem Eng 4:332–340
Prisciandaro M, Pepe F (1997) Absorption with zero and Pseudo-Zero order chemical reaction. Can J Chem Eng 75(2):362–368. https://doi.org/10.1002/cjce.5450750212
Amabile C, Abate T, De Crescenzo C, Sabbarese S, Migliaccio A, Chianese S, Musmarra D (2022) Poly(3-hydroxybutyrate) production from methane in bubble column bioreactors: Process simulation and design optimization. New Biotechnol 70(25):39–48. https://doi.org/10.1016/j.nbt.2022.04.004
Dong Z, Wen Z, Zhao F, Kuhn S, Noël T (2021) Scale-up of micro- and milli-reactors: An overview of strategies, design principles and applications. Chem Eng Sci: X 10:100097. https://doi.org/10.1016/j.cesx.2021.100097
Levenspiel O (1999) Chemical reaction engineering, 3rd edn. Wiley, New York
Otalvaro-Marín HL, Machuca-Martínez F (2020) Sizing of reactors by charts of Damköhler’s number for solutions of dimensionless design equations. Heliyon 6(11):e05386. https://doi.org/10.1016/j.heliyon.2020.e05386
Liu Y, Chen G, Yue J (2020) Manipulation of gas-liquid-liquid systems in continuous flow microreactors for efficient reaction processes. J Flow Chem 10:103–121. https://doi.org/10.1007/s41981-019-00062-9
Guo W, Heeres HJ, Yue J (2020) Continuous synthesis of 5-hydroxymethylfurfural from glucose using a combination of AlCl3 and HCl as catalyst in a biphasic slug flow capillary microreactor. Chem Eng J 381:122754. https://doi.org/10.1016/j.cej.2019.122754
Acknowledgements
The authors acknowledge funding from the Research Foundation Flanders (G0E6314N, 11H8121N) and KU Leuven (CELSA/20/012).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Koufou, D., Smets, J., Ameloot, R. et al. Aerobic oxidation of hydroxymethylfurfural using a homogeneous TEMPO/TBN catalytic system in 3D-printed milli-scale porous reactors. J Flow Chem 13, 169–183 (2023). https://doi.org/10.1007/s41981-023-00264-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41981-023-00264-2