Abstract
Background
Childhood steroid-sensitive nephrotic syndrome is a frequently relapsing disease with significant short- and long-term complications, leading to high healthcare costs and reduced quality of life for patients. The majority of relapses are triggered by upper respiratory tract infections (URTIs) and evidence shows that daily low-dose prednisolone at the time of infection may reduce the risk of relapse.
Objective
The aim of this study was to assess the cost effectiveness of a 6-day course of low-dose prednisolone at the start of a URTI when compared with placebo.
Methods
A state-transition Markov model was developed to conduct a cost-utility analysis with the outcome measured in quality-adjusted life-years (QALYs). Resource use and outcome data were derived from the PREDNOS2 trial. The analysis was performed from a UK National Health Service perspective and the results were extrapolated to adulthood. Model parameter and structural uncertainty were assessed using sensitivity analyses.
Results
The base-case results showed that administering low-dose prednisolone at the time of a URTI generated more QALYs and a lower mean cost at 1 year compared with placebo. In the long-term, low-dose prednisolone was associated with a cost saving (£176) and increased effectiveness (0.01 QALYs) compared with placebo and thus remained the dominant treatment option. These findings were robust to all sensitivity analyses.
Conclusion
A 6-day course of low-dose prednisolone at the time of a URTI in children with steroid-sensitive nephrotic syndrome has the potential to reduce healthcare costs and improve quality of life compared with placebo.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This study examined the cost effectiveness of a 6-day course of low-dose prednisolone at the time of a respiratory infection for treating children with steroid-sensitive nephrotic syndrome and found that it is the preferred option when compared with placebo. |
The high hospital costs and reduced health-related quality of life associated with a relapse, combined with the low cost of prednisolone, were the main reasons for this result. |
This study raises an interesting question on how to interpret economic evidence when it is at odds with the clinical-effectiveness evidence from the same trial. |
1 Introduction
Idiopathic nephrotic syndrome is the most common childhood glomerular disease, with an annual incidence of 2 per 100,000 children [1]. Children develop the disease at a median age of 2–3 years and its incidence rate is higher among boys and South Asian populations [1]. It presents with heavy urine protein losses that lead to low plasma levels of protein (predominantly albumin) and oedema.
According to current practice, the presenting nephrotic syndrome episode is treated with high-dose corticosteroids, usually administered as 60 mg/m2 or 2 mg/kg prednisone or prednisolone, and the disease that responds (around 90%) is then labelled as steroid-sensitive nephrotic syndrome (SSNS) [2]. Following initial successful treatment, at least 80% of children with SSNS will experience disease relapses [3] and about 50% will develop frequently relapsing nephrotic syndrome (two or more relapses within 6 months) [4]. Treatment of relapses requires further courses of high-dose corticosteroid and most patients with frequently relapsing disease will receive additional prophylactic immunosuppressive treatment due to the adverse effects experienced or anticipated with repeated courses of high-dose corticosteroids [3, 5].
SSNS relapses and their treatment with high-dose corticosteroids are associated with major short- and long-term adverse effects, including sepsis, thrombosis, malnutrition, hypertension, diabetes and behavioural problems [5, 6]. Such adverse effects often lead to substantial costs and morbidity for patients and their families.
The majority of SSNS relapses follow intercurrent upper respiratory tract infections (URTIs), and previous studies showed that at least half of URTIs in children with SSNS will trigger a relapse [7,8,9]. Previous studies have also shown that administering a 5- to 7-day course of daily low-dose prednisolone (around 0.5 mg/kg or 15 mg/m2) at the time of a URTI reduces the risk of subsequent relapses [10,11,12,13]. However, the generalisability of these findings were limited as most studies focused on children already taking background alternate-day prednisolone and the studies were carried out in India, Sri Lanka and the Middle East where patterns of intercurrent infection are different from Europe and North America. Additionally, there was no evaluation of the cost effectiveness.
A multicentre, placebo-controlled, randomised controlled trial (PREDNOS2) recently reported that administering daily low-dose prednisolone at the time of a URTI leads to no reduction in URTI-associated relapse [14]. However, it is still unknown if this leads to a cost-effective use of public resources, therefore the objective of this study was to conduct an economic evaluation using the data from the PREDNOS2 trial [15]. Unlike clinical-effectiveness analysis, which is based on the concept of classic statistical inference, economic evaluation is about decision analysis and is recommended by International Society for Pharmacoeconomics and Outcomes Research (ISPOR) guidance for trials that report a non-statistically significant difference in primary outcome [16].
2 Methods
2.1 Patients and Data Collection
Overall, 365 children with relapsing SSNS and a mean age of 7.6 years were recruited to the PREDNOS2 trial (ISRCTN10900733; EudraCT 2012-003476-39) from 91 of 122 UK paediatric departments that were set up for the trial. After exclusion, data for 271 children aged 1–17 years who had experience of at least one URTI during the 12-month follow-up period were included. Reasons for exclusion were withdrawal from the trial prior to a URTI and absence of URTIs during follow-up. The participants were randomised in a 1:1 ratio, stratified by background therapy, either to a 6-day course of low-dose prednisolone (15 mg/m2) at the start of a URTI (n = 134) or to an equivalent course of placebo (n = 137). An intention-to-treat analysis was adopted.
Individual patient-level data on both resource use and outcomes were collected on case report forms within the PREDNOS2 trial. Health-related quality of life (HRQoL) was measured using the CHU-9D, PedsQL and EQ-5D-3L instruments [17,18,19], depending on the age of the participants. Where appropriate, questionnaires were proxy-completed by parents or guardians. HRQoL and resource use data were collected at baseline and 3, 6, 9, and 12 months of follow-up.
2.2 Economic Model
Due to the fluctuating severity of the disease and frequent changes to background treatment, a decision analytic model was considered appropriate to accurately capture disease progression. A state transition Markov cohort model was developed using Treeage Pro (TreeAge Software, Inc, Williamstown, MA, USA). The model made a direct comparison of administering a 6-day daily course of prednisolone at the time of a URTI versus no change in current treatment (with use of placebo for blinding), from a UK National Health Service (NHS) perspective. The time horizon for the primary analysis was 12 months based on the trial follow-up period. Costs and outcomes were extrapolated in the secondary analysis until the cohort had reached adulthood (18 years).
2.3 Model Structure
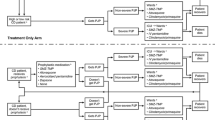
The model structure (Fig. 1) was informed by the treatment pathways from the PREDNOS2 trial and clinical input. SSNS is not considered a fatal disease therefore transition to ‘death’ was not included in the primary analysis but was added for the extrapolation in the form of an all-cause mortality risk derived from the Office of National Statistics (ONS; https://www.ons.gov.uk/).
Patients entered the model in one of four therapy groups according to their baseline background therapy:
-
No background therapy: no long-term immunosuppressive therapy.
-
Background prednisolone therapy: long-term maintenance prednisolone therapy only.
-
‘Combination background therapy’: long-term maintenance prednisolone therapy in conjunction with other immunosuppressive therapies.
-
Background immunosuppressive (immuno/ve) therapy: patients receiving long-term immunosuppressive therapy only .
After initial allocation, the patients moved into an ‘SSNS relapse’ state either having had a URTI or not, or they remained ‘relapse-free’. From the ‘SSNS relapse’ state, patients could move to ‘hospitalisation after relapse’ or ‘no hospitalisation after relapse’. From either of these three states (i.e. ‘hospitalisation after relapse’, ‘no hospitalisation after relapse’, or ‘relapse-free’), patients either continued with their current background therapy or changed to a new therapy following assessment every 3 months (six cycles). An assessment of background therapy was performed every 3 months in the trial; therefore, in line with trial procedures, the model was designed to only allow patients to move between background therapy groups every 3 months (six cycles). The transition probabilities between therapy groups were therefore dependent on clinical assessment of the rate of relapse in the previous 3 months. Transitions to the ‘No background therapy’ group from all other groups were only allowed if the patient was ‘relapse-free’ during the previous 3 months. For the groups ‘Background prednisolone therapy’ and ‘Combination background therapy’ that included maintenance prednisolone, the average dose administered at recruitment was 0.3 mg/kg.
A 15-day time cycle was deemed clinically meaningful based on the definition of a URTI-related relapse, which can occur within 14 days from a URTI episode. In the primary analysis, treatment costs and QALYs were estimated for 24 model-cycles (12 months). In the secondary analysis, the time horizon was extended beyond 12 months up to age 18 years to observe the long-term treatment costs and effects. For the extrapolated model, a starting age of 2 years was assumed and the model was run for 16 years (384 model-cycles). Due to the low rates of SSNS relapse among adults, a lifetime horizon was not considered relevant. A half-cycle correction was applied. The electronic supplementary Appendix describes all model assumptions in full.
2.4 Model Parameters
All model parameters including mean costs, HRQoL (utility) weights and transition probabilities were derived from the PREDNOS2 trial data and are summarised in Tables 1 and 2 and Appendix Tables A1 and A2. A discount rate of 3.5% was applied to both costs and outcomes for the secondary analysis [20]. All costs are reported in 2019 UK sterling (£).
Table 1 presents the unit costs. Resource use data collected at 3, 6, 9 and 12 months included primary care, hospital admissions, outpatient care, emergency visits and any prescribed medication. Staff costs were calculated using NHS reference costs [21] and the unit costs of the hospital-related services were derived from the National Schedule of NHS costs 2018/2019 [22]. For hospital admissions, a 5-day admission episode, which is the maximum expected length of stay for nephrotic syndrome in the UK, was used as a ‘trim point’ to differentiate between unit costs of episodes within and beyond the trim point. When the patient was discharged within 24 h, a regular day-rate tariff was applied. For outpatient care, different unit costs were applied for consultant- and non-consultant-led visits. Although resource use data of both renal and non-renal aetiology was collected in the clinical trial, only renal-related costs were considered for the economic analysis. The main justification was that renal-related episodes, mainly relapses, were expected to be the only cost event that would drive the cost difference between the intervention and the control arm. Missing information on the aetiology of visits or admissions was assumed to be non-renal related and were thus excluded. Adverse events were not costed separately to avoid double counting, since they were related to either outpatient or hospital events and were captured within the hospital data.
Treatment costs included immunosuppressants, prednisolone (both as the study drug and as a maintenance background treatment, or for relapse treatment) and any other prescribed medication. Over-the-counter medicines were not included in the analysis. A microcosting approach was used that categorised prescribed medication into 36 groups, and a mean unit cost was applied to each group with two or more drugs. All drug unit costs were estimated using the British National Formulary for Children [23].
In the primary analysis, HRQoL utility values were estimated using a combination of a crosswalk/mapping technique [24] to derive CHU-9D scores from the PedsQL responses for patients aged 2–4 years, the direct CHU-9D responses for patients between 5 and 17 years of age, and the direct EQ-5D-3L data for older patients. For more details about the mapping algorithm used for patients aged 2–4 years, please see the study by Lambe et al. [24].
Mean utility values were estimated for each therapy group from patients who were relapse-free at each trial follow-up point. Where relevant, a disutility associated with a relapse was applied regardless of whether a URTI had preceded or not. This disutility was estimated as the average difference in utility between patients who had experienced a relapse and patients who were relapse-free, as follows: patient utility data were collected every 3 months in the trial, at each time point these data were used to categorise patients into two groups based on whether they had experienced a relapse (or not) during the previous 3 months, and then the average difference between the utility values was used as a measure of disutility associated with a relapse. To calculate quality-adjusted life-years within the model, the area under the curve approach was applied combining the utility estimates with time [25]. Table 2 describes both the cost and utility data used in the model. Annual costs were divided by 24 to represent per-cycle costs. The detailed resource use costs per therapy group are presented in Appendix Table A3.
Transition probabilities for moving between the health states for each therapy group were calculated from the trial data and are described in full in the electronic supplementary Appendix (Model assumptions and Appendix Table A1). Following clinical expert advice, the model assumed that background therapy could only be altered after every 3 months, therefore this transition probability was time-dependent and was applied every sixth cycle. Transitions to the ‘No background therapy’ group from all other groups only happened if the patient was clinically assessed and relapse-free (relapse-free probabilities).
2.5 Sensitivity Analysis
Uncertainty was assessed by conducting probabilistic (PSA) and deterministic sensitivity analysis (DSA). In the PSA, 10,000 Monte Carlo simulations were used to plot a cost-effectiveness acceptability curve (CEAC) for different willingness to pay (WTP) thresholds for a QALY. Methodological uncertainty around the model parameters and model structure was assessed in three DSAs. In DSA1, we varied the method for deriving utility values for each age group. Compared with the base-case analysis, the CHU-9D data were used to obtain utility values for ages 5–11 years (ages 5–17 years in the base-case) and EQ-5D-3L data for ages 12–18 years (ages older than 17 years in the base-case). This age cut-off was chosen because of the guidelines for the use of CHU-9D when it was first developed [26]. In DSA2, monthly transitions between background therapy groups were applied to the model, and in DSA3, we tested the uncertainty associated with the disutility value by applying a higher value (0.03) of disutility associated with a relapse and an extreme scenario of negative disutility or a utility gain after relapse (− 0.04) [Table 3]. The upper and lower (extreme scenario of utility gain) bounds correspond to the 95% confidence interval (CI) of disutility.
A subgroup analysis was also performed to explore the uncertainty from using a mapping technique to derive CHU-9D scores from PedsQL responses. The base-case model at 1 year was run again after excluding patients aged between 2 and 4 years to observe if there were any differences in the cost-effectiveness results.
3 Results
3.1 Primary Analysis
The cost-effectiveness results for the primary analysis (at 1 year) are summarised in Table 3. These show that low-dose prednisolone is dominant as it accrued a lower mean cost and generated 0.003 more QALYs when compared with placebo.
Background therapy was a key driver of costs and effects, and it was noted that after 1 year, proportionally more patients had moved to the ‘No background therapy’ group in the prednisolone arm compared with the standard arm (43% vs. 39%) and there was a higher proportion of patients in the ‘Combination background therapy’ group within the standard arm relative to the prednisolone arm (32% vs. 30%) (Appendix Table A4). Although the model only ‘allowed’ patients to change background therapy every 3 months, even after 1 year these small differences influenced the incremental cost and effect differences.
The findings of all three DSAs show low-dose prednisolone to remain the dominant option throughout. As shown in Table 3, the one-way sensitivity analysis (DSA3) confirmed the base-case results, therefore no further reporting was necessary. In the subgroup analysis, by removing the cohort for which the HRQOL values are mapped, the low-dose prednisolone arm still generated more QALYs when compared with placebo and therefore mapping from PEDSQL to CHU-9D for children aged 2–5 years did not alter the overall treatment recommendation (Table 4).
3.2 Secondary Analysis
Table 5 summarises the results of the extrapolated analysis. Over 16 years, low-dose prednisolone generated an average cost saving of £176 and 0.01 more QALYs when compared with placebo and therefore remains the dominant treatment option.
3.3 Probabilistic Sensitivity Analysis
The primary PSA showed that the majority of cost and effect distributions lie in the southeast quadrant of the cost-effectiveness plane (Fig. 2) and therefore low-dose prednisolone is less costly and more effective than placebo. When plotted on a CEAC, the probability of prednisolone being cost effective at a threshold WTP value of £20,000 is approximately 80% (Fig. 3).
Similarly, the PSA results for the secondary analysis illustrate that the majority of points in the cost-effectiveness plane lie in the southeast quadrant (Fig. 2) and there is a 90% probability of low-dose prednisolone being cost effective at a WTP threshold of £20,000 (Fig. 3). Low-dose prednisolone is therefore considered the most cost-effective option at all WTP thresholds.
4 Discussion
Administering a 6-day course of low-dose prednisolone at the time of a URTI for the management of children with SSNS leads to reduced costs and increased QALYs when compared with placebo. This finding was maintained in the extrapolated analysis up to age 18 years and was robust to all sensitivity analyses. Therefore, based on a 16-year time horizon, placebo is dominated by a 6-day course of low-dose prednisolone.
There are two main reasons for this result. First, the cost differences were mainly driven by the difference in costs of background therapy and the costs associated with having a relapse. After 1 year, proportionally more patients in the prednisolone arm had discontinued background therapy and moved to the ‘No background therapy’ group compared with placebo, and there were fewer cases of hospitalisation after relapse (Appendix Table A1). When these differences were sustained and extrapolated over 16 years, this led to a difference in incremental cost. Second, when the utility decrement associated with having a relapse was accounted for, this led to low-dose prednisolone gaining more QALYs compared with placebo.
These economic findings are particularly interesting as the clinical-effectiveness results from the PREDNOS2 trial concluded that using prednisolone at the time of a URTI does not lead to a statistically significant reduction in SSNS relapses and therefore should not be routinely recommended as a strategy. It therefore appears that the clinical and cost-effectiveness results from the PREDNOS2 trial are producing different recommendations. The explanation for this stems from the underlying differences in the theoretical approaches, as explained clearly by Claxton [27] and more recently Raftery et al. [28]. Unlike the clinical-effectiveness analysis, the economic analysis is focused on the ratio of costs and outcomes and assessing value, based on what society is willing to pay for a unit gain in outcome (QALYs). With this in mind, although the PREDNOS2 trial showed that the difference in SSNS relapse between the treatment arms did not reach statistical significance, there was a difference in relapse rate favouring the prednisolone arm. The number of relapses reported within the statistical analysis by therapy group were equivalent to the number of relapses captured within the model. As these relapse episodes are frequently associated with high hospital costs and a corresponding reduction in HRQoL, this small difference in relapse rate, albeit not statistically significant for the clinical effectiveness, combined with the difference in costs associated with background therapy and the relative cheap costs of the intervention (prednisolone), led to the low-dose prednisolone strategy being the ‘dominant’ treatment option. This apparent variation in findings between the clinical and cost effectiveness of this trial emphasises the importance of the morbidity and economic consequences of SSNS relapses as well as the salient underlying costs associated with background therapy. It also highlights our previous point that economic evaluation is not about hypothesis testing but about decision analysis based on the combined ratio of cost and effect, relative to an established threshold cited by the National Institute for Health and Care Excellence (NICE).
The robustness of findings was tested in a PSA by plotting 10,000 paired cost and QALY estimates on the cost-effectiveness plane and CEAC. The incremental net benefit was used to construct the CEAC due to well-established limitations when ICERs from bootstrap replicates are spread over the four quadrants of the cost-effectiveness plane. According to published recommendations on dealing with uncertainty in cost-effectiveness analyses, CEACs provide a visual representation of the joint uncertainty of cost, as well as QALY estimates, and thus offer a simple interpretation of the results. Given that 95% CIs for cost-effectiveness ratios are not always defined, CEACs are considered an effective tool for removing reliance on such ratio statistics [29].
To our knowledge, this is the first model-based economic evaluation to explore the cost effectiveness of 6-day low-dose prednisolone at the time of a URTI in a UK population. A strength of the study is that it used data from the largest ever clinical trial of an investigational medicinal product in children with nephrotic syndrome [14]. The study used a Markov model to capture disease progression and perform the cost-utility analysis; Markov models account for the repetitive nature of events while handling concomitant treatment consequences on both costs and health outcomes [30]. Both methodological and structural uncertainty were explored in separate sensitivity analyses. A strength of the model is that all parameters were derived from the PREDNOS2 trial data collected over 12 months, which was a large, methodologically robust, double-blinded, multicentre randomised controlled trial. For the secondary analysis, the time horizon was set at 16 years as once patients reach adulthood, SSNS tends to be better controlled and the rate of relapse substantially falls.
In terms of limitations, the economic model used a crosswalk/mapping technique to generate utility values for children under 5 years of age and this may have introduced additional uncertainty. Although utility values should ideally be derived using direct HRQoL elicitation, no validated utility-based instrument exists for patients as young as 2–4 years of age. By excluding this cohort from the sample in a subgroup analysis, this did not impact on the ICER.
Another possible limitation was that patients in the model could change their background therapy every 3 months but not more frequently. The decision to alter background therapy is usually assessed on a patient-by-patient basis by a clinician who has in-depth insight into the patient history, and restricting it to every 3 months in the model could potentially have reduced this ‘real-life’ variation and biased the results. However, we relaxed this assumption within a sensitivity analysis to a potential change every month and found it to have no impact on the ICER. Furthermore, a natural decreasing rate of relapse by age was not included to avoid adding further uncertainty in the model, since the reduction in relapse rate depends on heterogeneous epidemiological factors [31]. Additionally, dividing each treatment arm into four background therapy groups in the model resulted in a small number of patients (about 34) per group, which is acknowledged as a limitation of this study. Additional data collection would be helpful to further validate the model inputs.
The results from the model may have limited generalisability to other populations outside the UK since the model only used the PREDNOS2 (and therefore UK) trial data. Geographic or other epidemiological factors may affect relapse and URTI rates, resulting in differences in disease course and treatment therapy, and further research will be necessary to assess the cost effectiveness within these country settings.
5 Conclusion
This study assessed the cost effectiveness of using low-dose prednisolone in patients with SSNS who experience a URTI and found it to offer good value for money. It raises important questions about treatment recommendations when the clinical and economic analysis of trial data produce different results. Given the high costs and the HRQOL impacts from a relapse, as well as the costs of background therapy, prednisolone is a relatively cheap intervention that, from an economics perspective, was shown to be a cost-effective use of public resources. How this economics evidence is weighted against the clinical evidence from the PREDNOS2 trial is open to debate.
References
McKinney PA, et al. Time trends and ethnic patterns of childhood nephrotic syndrome in Yorkshire, UK. Pediatr Nephrol. 2001;16(12):1040–4.
Rees L, et al. Paediatric Nephrology (Oxford Specialist Handbooks in Paediatrics). Oxford: Oxford University Press; 2012.
Webb NJA, et al. Long term tapering versus standard prednisolone treatment for first episode of childhood nephrotic syndrome: phase III randomised controlled trial and economic evaluation. BMJ. 2019;365: l1800.
Larkins N, et al. Steroid-sensitive nephrotic syndrome: an evidence-based update of immunosuppressive treatment in children. Arch Dis Child. 2016;101(4):404–8.
Noone DG, Iijima K, Parekh R. Idiopathic nephrotic syndrome in children. Lancet. 2018;392(10141):61–74.
McCaffrey J, Lennon R, Webb NJ. The non-immunosuppressive management of childhood nephrotic syndrome. Pediatr Nephrol. 2016;31(9):1383–402.
Alwadhi RK, Mathew JL, Rath B. Clinical profile of children with nephrotic syndrome not on glucorticoid therapy, but presenting with infection. J Paediatr Child Health. 2004;40(1–2):28–32.
Arun S, et al. Efficacy of zinc supplements in reducing relapses in steroid-sensitive nephrotic syndrome. Pediatr Nephrol. 2009;24(8):1583–6.
MacDonald NE, et al. Role of respiratory viruses in exacerbations of primary nephrotic syndrome. J Pediatr. 1986;108(3):378–82.
Mattoo TK, Mahmoud MA. Increased maintenance corticosteroids during upper respiratory infection decrease the risk of relapse in nephrotic syndrome. Nephron. 2000;85(4):343–5.
Abeyagunawardena AS, Trompeter RS. Increasing the dose of prednisolone during viral infections reduces the risk of relapse in nephrotic syndrome: a randomised controlled trial. Arch Dis Child. 2008;93(3):226–8.
Gulati A, et al. Daily corticosteroids reduce infection-associated relapses in frequently relapsing nephrotic syndrome: a randomized controlled trial. Clin J Am Soc Nephrol. 2011;6(1):63–9.
Abeyagunawardena AS, et al. Short courses of daily prednisolone during upper respiratory tract infections reduce relapse frequency in childhood nephrotic syndrome. Pediatr Nephrol. 2017;32(8):1377–82.
Christian MT, et al. Evaluation of daily low-dose prednisolone during upper respiratory tract infection to prevent relapse in children with relapsing steroid-sensitive nephrotic syndrome: the PREDNOS 2 Randomized Clinical Trial. JAMA Pediatr. 2022;176(3):236–43.
Webb NJ, et al. Short course daily prednisolone therapy during an upper respiratory tract infection in children with relapsing steroid-sensitive nephrotic syndrome (PREDNOS 2): protocol for a randomised controlled trial. Trials. 2014;15:147.
Ramsey SD, et al. Cost-effectiveness analysis alongside clinical trials II-An ISPOR Good Research Practices Task Force report. Value Health. 2015;18(2):161–72.
Stevens K. Valuation of the Child Health Utility 9D Index. Pharmacoeconomics. 2012;30(8):729–47.
Varni JW, et al. The PedsQL 4.0 as a pediatric population health measure: feasibility, reliability, and validity. Ambul Pediatr. 2003;3(6):329-341.
Devlin N, Parkin D, Janssen B. Methods for Analysing and Reporting EQ-5D Data. Springer International Publishing; 2020.
National Institute for Health Care Excellence. NICE process and methods guides, in guide to the methods of technology appraisal 2013. London: National Institute for Health and Care Excellence; 2013.
Curtis L, Burns A. Unit Costs of Health and Social Care 2019. Personal Social Services Research Unit. https://www.pssru.ac.uk/project-pages/unit-costs/unit-costs-2019/. Accessed Feb 2020.
National Health Service. National Schedule of NHS costs. 2019. https://www.england.nhs.uk/wp-content/uploads/2020/08/1_-_NCC_Report_FINAL_002.pdf. Accessed Feb 2020.
Paediatric Formulary Committee. BNF for Children (online). BMJ Group (ed). Pharmaceutical Press and RCPCH Publications; 2020. www.medicinescomplete.com. Accessed Feb 2020.
Lambe T, et al. Mapping The Paediatric Quality Of Life Inventory (PedsQL™) Generic Core Scales onto the Child Health Utility Index-9 Dimension (CHU-9D) score for economic evaluation in children. Pharmacoeconomics. 2018;36(4):451–65.
Manca A, Hawkins N, Sculpher MJ. Estimating mean QALYs in trial-based cost-effectiveness analysis: the importance of controlling for baseline utility. Health Econ. 2005;14(5):487–96.
Canaway AG, Frew EJ. Measuring preference-based quality of life in children aged 6–7 years: a comparison of the performance of the CHU-9D and EQ-5D-Y–the WAVES pilot study. Qual Life Res. 2013;22(1):173–83.
Claxton K. The irrelevance of inference: a decision-making approach to the stochastic evaluation of health care technologies. J Health Econ. 1999;18(3):341–64.
Raftery J, et al. ’Not clinically effective but cost-effective’—paradoxical conclusions in randomised controlled trials with “doubly null” results: a cross-sectional study. BMJ Open. 2020;10(1): e029596.
O’Brien BJ, Briggs AH. Analysis of uncertainty in health care cost-effectiveness studies: an introduction to statistical issues and methods. Stat Methods Med Res. 2002;11(6):455–68.
Briggs A, Sculpher M. An introduction to Markov modelling for economic evaluation. Pharmacoeconomics. 1998;13(4):397–409.
Sinha A, et al. Disease course in steroid sensitive nephrotic syndrome. Indian Pediatr. 2012;49(11):881–7.
Acknowledgements
1. Kindly provide the members list for the PREDNOS Collaborative Group, NIHR Medicines for Children Research Network Nephrology Clinical Studies Group. The PREDNOS Collaborative Group comprises local research teams as listed below:. PREDNOS 2 TRIAL MANAGEMENT GROUP. Professor Nicholas Webb (Royal Manchester Children’s Hospital, Chief Investigator to May2018). Dr Martin Christian (Nottingham Children’s Hospital, Chief Investigator from June 2018). Miss Natalie Ives (Reader in Clinical Trials, Senior Statistician, BCTU). Professor Emma Frew (Professor in Health Economics, University of Birmingham). Mrs Elizabeth Brettell (Renal Trials Manager, BCTU). PREDNOS 2 TRIAL MANAGEMENT AND DAY-TO-DAY CO-ORDINATION. BIRMINGHAM CLINICAL TRIALS UNIT, UNIVERSITY OF BIRMINGHAM. Mrs Elizabeth Brettell, Team Leader. Emma Barsoum, Trial Manager. Ms Helen Bodenham-Chilton, Trial Manager. Mr Adam Khan, Trial Manager. Ms Noreen Akhtar, Senior Data Manager. Mrs Charmaine Hunt, Senior Data Manager. Ms Carla Galinha, Data Manager. Ms Dominique Smith, Data Manager. Mr Neil Winkles, Senior Systems Developer. PREDNOS 2 TRIAL STATISTICS, BCTU. Miss Natalie Ives, Reader in Clinical Trials, Senior Statistician. Ms Rebecca Woolley, Senior Medical Statistician. Mr Samir Mehta, Senior Medical Statistician. Mrs Catherine Moakes, Senior Medical Statistician. PREDNOS 2 TRIAL HEALTH ECONOMICS. Professor Emma Frew, Professor in Health Economics. Ms Nafsika Afentou, Health Economics Research Associate. PATIENT AND PUBLIC REPRESENTATIVES. Mrs Wendy Cook, Director, Nephrotic Syndrome Trust (NeST). Mrs Sandra Cope. TRIAL STEERING COMMITTEE. Dr Megan Thomas (Consultant Community Paediatrician, Blackpool Teaching Hospitals NHS Trust, Independent Chair). Dr Nigel Coad (Consultant Paediatrician, University Hospitals of Coventry and Warwickshire NHS Trust, Independent Member). Mrs Sandra Cope (Consumer Representative, Independent Member). Dr Andrew Duncan (Consultant Paediatrician, Borders General Hospital, Melrose. Independent Member). Darren Green (Consultant Acute Physician and Nephrologist, Salford Royal NHS FoundationTrust, Independent Member). Dr Kate Hillman (Consultant Nephrologist, Manchester Royal Infirmary, Independent Member). Dr Zala Ibrahim (Consultant Paediatrician, Russells Hall Hospital, Dudley, Independent Member). Dr Ly-Mee Yu (Senior Medical Statistician, Nuffield Department of Primary Care Health Sciences, University of Oxford, Independent Member). Dr Martin Christian (Chief Investigator, Non-independent Member). Natalie Ives (Co-Investigator, Senior Statistician, Non-independent Member). Professor Nicholas Webb, (Chief Investigator, Non-independent Member). PREDNOS 2 DATA MONITORING COMMITTEE. Professor Philip Kalra (Consultant Nephrologist and Honorary Professor, Salford Royal Hospital NHS Trust, Chair). Professor Saul Faust (Reader in Paediatric Immunology and Infectious Diseases, Southampton General Hospital). Dr Andrea Marshall (Senior Research Fellow in Medical Statistics, Warwick Clinical Trials Unit). PARTICIPATING CENTERS AND PREDNOS 2 COLLABORATIVE GROUP MEMBERS. Cambridge University Hospitals NHS Foundation Trust. Addenbrooke's Hospital:. Dr Birgit Ulbrich, Principal Investigator, Dr Peter Heinz, Sara Foster, Airedale NHS Foundation Trust. Airedale General Hospital:. Dr Pronab Bala, Principal Investigator. Western Health and Social Care Trust. Altnagelvin Area Hospital:. Dr Damien Armstrong, Principal Investigator, Julie Brown & Team. Wirral University Teaching Hospital NHS Foundation Trust. Arrowe Park Hospital:. Dr Elizabeth Breen, Principal Investigator, Sharon Hughes & Team. Barnsley Hospital NHS Foundation Trust. Barnsley Hospital:. Dr Rajeev Gupta, Principal Investigator, Miranda Murray,. Doncaster and Bassetlaw Hospitals NHS Foundation Trust. Doncaster Royal Infirmary & Bassetlaw Hospital:. Dr Mathew Kurian, Principal Investigator, Dr Sunitha Sampath, Dr Sarah Didier, Sarah Shortland, Sharon Allen, Lisa Warren & Team,. Birmingham Women's and Children's NHS Foundation Trust. Birmingham Children's Hospital:. Dr David Milford, Principal Investigator, Dr Mordi Muorah, Alison Watson, Fatima Bibi & Team,. Bradford Teaching Hospitals NHS Foundation Trust. Bradford Royal Infirmary:. Dr Eduardo Moya, Principal Investigator,. University Hospitals Bristol NHS Foundation Trust. Bristol Royal Hospital for Children:. Prof Moin Saleem, Principal Investigator, Dr Alison Kelly, Michelle Ross, Natalie Fineman & Team,. Calderdale and Huddersfield NHS Foundation Trust. Calderdale Royal Hospital & Huddersfield Royal Infirmary:. Dr Eilean Crosbie, Principal Investigator, Rachel Swingler, Susan Kilroy & Team. Chesterfield Royal Hospital NHS Foundation Trust. Chesterfield Royal Hospital:. Dr Oyekunle Ayonrinde, Principal Investigator, Amanda Smith, Colchester Hospital University NHS Foundation Trust. Colchester General Hospital:. Dr Andrea Turner, Principal Investigator, Dr Jonathan Campbell, Aine Turner, Countess of Chester Hospital NHS Foundation Trust. Countess of Chester Hospital:. Dr Stephen Brearey, Principal Investigator, Caroline Burchett & Sarah De-Beger, Croydon Health Services NHS Trust. Croydon University Hospital:. Dr Theo Fenton, Principal Investigator, North Cumbria University Hospitals NHS Trust. Cumberland Infirmary:. Dr Glyn Jones, Principal Investigator, Nicci Kelsall, Dartford and Gravesham NHS Trust. Darent Valley Hospital:. Dr Selwyn D'Costa, Principal Investigator, County Durham and Darlington NHS Foundation Trust. Darlington Memorial Hospital, University Hospital of North Durham:. Dr Dinakaran Jayachandran, Principal Investigator, Dr Asha Nair Catherine Tarn Nozedar, Dawn Egginton, Plymouth Hospitals NHS Trust. Derriford Hospital:. Dr Oliver Cuthell, Principal Investigator, Dr Catherine Derry, Dr Kathiresan Natesan, Sarah-Jane Sharman, The Mid Yorkshire Hospitals NHS Trust. Dewsbury and District Hospital, Pinderfields Hospital:. Dr Rajeeva Singh, Principal Investigator, Dr Kathryn Deakin, Gail Castle & Team, Northern Lincolnshire and Goole Hospitals NHS Foundation Trust, Diana, Princess of Wales Hospital (Grimsby). Dr Bukar Wobi, Principal Investigator, Dr Bemigho Etuwewe, Caroline Burnett & Team, Surrey and Sussex Healthcare NHS Trust, East Surrey Hospital:. Dr Kamal Khoobarry, Principal Investigator. East Sussex Healthcare NHS Trust, Eastbourne District General Hospital, Conquest Hospital:. Dr Graham Whincup, Principal Investigator, Anne Cowley, Guy's and St Thomas' NHS Foundation Trust. Evelina London Children's Hospital:. Dr Ania Koziell, Principal Investigator, Dr Manish Sinha, Dr Christopher Reid, Mohammad Ahmad & Team, The Pennine Acute Hospitals NHS Trust. Fairfield General Hospital, North Manchester General Hospital, The Royal Oldham Hospital, Rochdale Infirmary:. Dr Beena Padmakumar, Principal Investigator, Dr Talaivirichan Magadevan, Grainne O'Connor, Louise Woodhead, University Hospitals of Morecambe Bay NHS Foundation Trust Royal Lancaster Infirmary & Furness General Hospital. Dr Mireille Formosa, Principal Investigator, Dr Nayan Peepah Nardeosingh, Kathryn Allison, Betsi Cadwaladr University Health Board, Glan Clwyd Hospital:. Dr Markus Hesseling, Principal Investigator, Annette Bolger, Lucie Hobson & Team, Gloucestershire Hospitals NHS Foundation Trust, Gloucestershire Royal Hospital:. Dr Adamu Sambo, Principal Investigator, Dr Lyda Jadresic, Susan Beames, Great Ormond Street Hospital for Children NHS Foundation Trust, Great Ormond Street Hospital:. Dr Detlef Bockenhauer, Principal Investigator, Dr Daljit Hothi, Elizabeth Vella, Corinne Linton, Shaima Yussuf, Tendai Bazaya, Mahmoud Abou-Rayyah & Team. Great Western Hospitals NHS Foundation Trust, Great Western Hospital:. Dr Nick West, Principal Investigator. Homerton University Hospital NHS Foundation Trust, Homerton University Hospital:. Dr Rajiv Sood, Principal Investigator, Hilarious De Jesus & Team Hull and East Yorkshire Hospitals NHS Trust, Hull Royal Infirmary:. Dr Vikas Gupta, Principal Investigator, Dr Verghese Mathew. James Paget University Hospitals NHS Foundation Trust, James Paget University Hospital:. Dr Esi Bentsi-Enchill , Principal Investigator, Allyson Davison. Oxford University Hospitals NHS Trust, John Radcliffe Hospital:. Dr Janet Craze, Principal Investigator. East Kent Hospitals University NHS Foundation Trust, Kent and Canterbury Hospital, Queen Elizbaeth the Queen Mother Hospital, William Harvey Hospital:. Dr Elhussein Rfidah, Principal Investigator, Janine Musselwhite, Angela Moon & Team. Kettering General Hospital NHS Foundation Trust, Kettering General Hospital:. Dr Harsha Bilolikar, Principal Investigator, Sonia White & Team Sherwood Forest Hospitals NHS Foundation Trust, King's Mill Hospital:. Dr Simon Rhodes, Principal Investigator, Caroline Moulds. Leeds Teaching Hospitals NHS Trust, Leeds General Infirmary:. Dr Hitesh Prajapati, Principal Investigator, Dr Eric Finlay, Dr Pallavi Yadav, Dr Amanda Newnham, Dr Kay Tyerman, Majorie Allen, Lucy Wellings & Team, University Hospitals of Leicester NHS Trust, Leicester Royal Infirmary:. Dr Angela Hall, Principal Investigator, Jackie Philps & Team. Mid Cheshire Hospitals NHS Foundation Trust, Leighton Hospital:. Dr Subajini Kaviethasan, Principal Investigator, Sally Smith & Team. United Lincolnshire Hospitals NHS Trust, Pilgrim Hospital & Lincoln County Hospital:. Dr David Broodbank, Principal Investigator, Dr Sourabh Mukhopadhyay, Dr Ruchika Gupta,Amanda Roper, Susie Butler & Team. Luton and Dunstable Hospital NHS Foundation Trust Luton and Dunstable University Hospital:. Dr Tomasz Rajkowski, Principal Investigator, Dr Michael Eisenhut, Karen Duncan, Karen Samm, Samantha Clough & Team. East Cheshire NHS Trust, Macclesfield District General Hospital:. Dr David Wright, Principal Investigator, Dr Krishnakumar Thattakkat, Dr Ignatius Losa, Natalie Keenan & Team. Maidstone and Tunbridge Wells NHS Trust, Maidstone Hospital, Tunbridge Wells Hospital:. Dr Krishnan Balasubramanian, Principal Investigator. Walsall Healthcare NHS Trust, Manor Hospital, Walsall:. Dr Muhammad Javed, Principal Investigator, Sharon Kempson, Marie Phipps & Team. Medway NHS Foundation Trust, Medway Maritime Hospital:. Dr Janette Cansick, Principal Investigator, Maines Msiska. Milton Keynes University Hospital NHS Foundation Trust, Milton Keynes Hospital:. Dr Lazarus Anguvaa, Principal Investigator, Dr Mya Aye, Sally Conway, Natalie Beer, Francesca Wright & Team. Taunton and Somerset NHS Foundation Trust, Musgrove Park Hospital:. Dr Jennifer Langlands, Principal Investigator, Kirsty O'Brien, Nicola Thorne & Team. The Royal Wolverhampton Hospitals NHS Trust, New Cross Hospital:. Dr Karen Davies, Principal Investigator, Sharon Kempson, Marie Phipps & Team. Barts Health NHS Trust, Newham University Hospital, The Royal London Hospital, Whipps Cross Hospital:. Dr Ami Parikh, Principal Investigator, Dr Nimze Gadong, Dr Bahadur Anjum, Nicolene Plaatjies, Ivone Lancoma-Malcolm, Hilarious De Jesus & Team. Norfolk and Norwich University Hospitals NHS Foundation Trust, Norfolk and Norwich University Hospital:. Dr Vipan Datta, Principal Investigator, Dr Chris Upton, Louisa Fear, Louise Coke & Team. Northern Devon Healthcare NHS Trust, North Devon District Hospital:. Dr Dermot Dalton, Principal Investigator, Becky Holbrook & Team. Northampton General Hospital NHS Trust, Northampton General Hospital:. Dr Imogen Norton, Principal Investigator,. Nottingham University Hospitals NHS Trust, Nottingham Children's Hospital (Queens Medical Centre):. Dr Martin Christian, Principal Investigator, Dr Andrew Lunn, Olivia Vincent, Helen Navarra, Neelam Khan & Team. Peterborough and Stamford Hospitals NHS Foundation Trust, Peterborough City Hospital:. Dr Mona Aslam, Principal Investigator, Paula Goodyear. Poole Hospital NHS Foundation Trust, Poole Hospital:. Dr Steve Wadams, Principal Investigator, Susan Power, Amy Roff & Team. The Shrewsbury and Telford Hospital NHS Trust, Princess Royal Hospital, Royal Shrewsbury Hospital:. Dr Manish Gupta, Principal Investigator, Dr Naeem Ayub, Charlotte Owen & Team. Portsmouth Hospitals NHS Trust, Queen Alexandra Hospital (Portsmouth):. Dr Judith Scanlan, Principal Investigator, Sharon McCready, Andrew Gribbin. Burton Hospitals NHS Foundation Trust, Queen's Hospital, Burton:. Dr Mansoor Ahmed, Principal Investigator, Dr Dominic Muogbo, Dr Heather Carswell, Stephanie Boswell, Claire Backhouse & Team. Barking, Havering and Redbridge University Hospitals NHS Trust, Queen's Hospital, Romford:. Dr Junaid Solebo, Principal Investigator, Helen Smith. NHS Highland, Raigmore Hospital:. Dr Alan Webb, Principal Investigator, Ing-Marie Logie, Sandra Dekker. The Rotherham NHS Foundation Trust, Rotherham Hospital:. Dr Sanjay Suri, Principal Investigator, Janet Shackleton & Team. NHS Grampian, Royal Aberdeen Children's Hospital:. Dr Craig Oxley, Principal Investigator, Margaret Connon & Team,. Wrightington, Wigan and Leigh NHS Foundation Trust, Royal Albert Edward Infirmary:. Dr Vineeta Joshi, Principal Investigator, Nicola Pemberton & Team. NHS Greater Glasgow and Clyde, Royal Alexandra Hospital:. Dr Heather Maxwell, Principal Investigator, Dr Amita Sharma, Elizabeth Waxman & Team. Belfast Health and Social Care Trust, Royal Belfast Hospital for Sick Children:. Dr Karl McKeever, Principal Investigator, Muriel Millar & Team. Royal Berkshire NHS Foundation Trust, Royal Berkshire Hospital:. Dr Ann Gordon, Principal Investigator, Dr Susan Edees, Susan Hallett & Team. East Lancashire Hospitals NHS Trust, Royal Blackburn Hospital:. Dr Javed Iqbal, Principal Investigator, Dr Beate von Bremen, Heather Collier, Andrew Lancaster & Team. Bolton NHS Foundation Trust, Royal Bolton Hospital:. Dr Fiona Watson, Principal Investigator, Joanne Henry & Team. Derby Hospitals NHS Foundation Trust, Royal Derby Hospital:. Dr Richard Bowker, Principal Investigator, Coral Smith. Royal Devon & Exeter NHS Foundation Trust, Royal Devon & Exeter Hospital (Wonford):. Dr Hannah Cottis, Principal Investigator, Dr Rebecca Samuel, Caroline Harrill, Suzanne Wilkins & Team. NHS Greater Glasgow and Clyde, Royal Hospital for Children (Glasgow):. Dr Heather Maxwell, Principal Investigator, Dr Ben Reynolds, Dr David Hughes, Elizabeth, Waxman & Team. NHS Lothian Royal Hospital for Sick Children (Edinburgh):. Dr Ben Reynolds, Principal Investigator, Dr David Hughes, Tracey McGregor, Maxine Ramsay, Julie Baggott, Naomi Matos & Team. Alder Hey Children's NHS Foundation Trust, Royal Liverpool Children's Hospital (Alder Hey):. Dr Caroline Jones, Principal Investigator, Dr Henry Morgan, Dr Richard Holt, Dr Louise Oni,Theresa Moorcroft, Joanne Shakeshaft & Team,. Central Manchester University Hospitals NHS Foundation Trust, Royal Manchester Children's Hospital:. Dr Mohan Shenoy, Prof Nicholas Webb (Principal Investigator), Dr Amrit Kaur, Dr Dean Wallace, Dr Nicholas Plant, Dr Shaila Sukthankar, Angela Branson, Helen Blackburn, Jane Howell, Jess Nichols & Team. University Hospitals of North Midlands NHS Trust, Royal Stoke University Hospital, The County Hospital (Stafford Hospital):. Dr Furqan Basharat, Principal Investigator, Dr Saeeda Raja, Marie Phipps, Helen Parker, Joanne Tomlinson, Eric Roe & Team. Royal United Hospital Bath NHS Foundation Trust, Royal United Hospital (Bath):. Dr Lynn Diskin, Principal Investigator, Alison Barratt. The Dudley Group NHS Foundation Trust, Russells Hall Hospital:. Dr Subramanian Mahadevan-Bava, Principal Investigator, Abigail Weston, Daljit Kaur. York Teaching Hospital NHS Foundation Trust, Scarborough Hospital:. Dr Udupa Venkatesh, Principal Investigator, Emma Temlett, Simon Dyer, Kerry Elliott, Rosie Furness & Team. Sheffield Children's NHS Foundation Trust, Sheffield Children's Hospital:. Dr Andrew Lunn, Principal Investigator, Janet Shackleton, Sarah Shortland, Miranda Murray & Team. University Hospital Southampton NHS Foundation Trust, Southampton Children's Hospital:. Dr Rodney Gilbert, Principal Investigator, Dr Matthew Harmer, Dr Shuman Haq, Lisa Fairhead, Louise Haskell, Victoria Bingham & Team. Southend University Hospital NHS Foundation Trust, Southend Hospital:. Dr Anupam Shrivastava, Principal Investigator, Onie Hove, Bernard Hadebe. Isle of Wight NHS Trust, St Mary's Hospital:. Dr Christopher Magier, Principal Investigator, Bettina Harms, Sian Butterworth. Ashford and St Peter's Hospitals NHS Foundation Trust, St Peter's Hospital:. Dr Tariq Bhatti, Principal Investigator, Dr Aisling Parker, Lorna Walding & Team. Western Sussex Hospitals NHS Foundation Trust, St Richard's Hospital, Worthing Hospital:. Dr Nicholas Brennan, Principal Investigator. Stockport NHS Foundation Trust, Stepping Hill Hospital:. Dr Chris Cooper, Principal Investigator, Sara Bennett & Team. Tameside Hospital NHS Foundation Trust, Tameside Hospital:. Dr Anjali Date, Principal Investigator, Dr Anjali Petkar, Wendy Hulse & Team. Worcestershire Acute Hospitals NHS Trust, Worcestershire Royal Hospital, The Alexandra Hospital (Redditch):. Dr Munir Ahmed, Principal Investigator, Dr Tom Dawson, Connie Rowlands & Stephanie Chamberlain. Wye Valley NHS Trust, The County Hospital, Hereford:. Dr Simon Meyrick, Principal Investigator, Dr Iain Darwood, Emma Collins. South Tees NHS Foundation Trust, The Friarage Hospital, The James Cook University Hospital:. Dr Rajesh Lall, Principal Investigator, Dr Elizabeth Onifade, Joanna Green & Team. The Newcastle Upon Tyne Hospitals NHS Foundation Trust, The Great North Children's Hospital:. Dr Sally Johnson, Principal Investigator, Dr Heather Lambert, Dr Yincent Tse, Dr Michal Malina, Dr Vijaya Sathyanarayan, Jenny Booth, Kathryn Bell, Stephen Crulley & Team. The Ipswich Hospital NHS Trust, The Ipswich Hospital:. Dr Jackie Buck, Principal Investigator, Deborah Beeby, Louise Hunt & Team. York Teaching Hospital NHS Foundation Trust, The York Hospital. Dr Sundeep Sandhu, Principal Investigator, Dr Gur Millman, Dr Murray Wheeler, Anna Clayton, David Thompson & Team. NHS Ayrshire and Arran, University Hospital Crosshouse:. Dr Bridget Oates, Principal Investigator, Claire Bell, Joanna Wardrop & Team. North Tees and Hartlepool NHS Foundation Trust, University Hospital of North Tees:. Dr Vijay Tandle, Principal Investigator, Carolyn Campbell, Dawn Egginton & Team. Cardiff and Vale University Health Board, University Hospital of Wales:. Dr Shivaram Hegde, Principal Investigator, Dr Rajesh Krishnan, Zoe Morrison, Jennifer Muller, Louise Yendle & Team,. Warrington and Halton Hospitals NHS Foundation Trust, Warrington Hospital:. Dr Delyth Webb, Principal Investigator, Natalie Rogers,. Chelsea and Westminster Hospital NHS Foundation Trust, West Middlesex University Hospital:. Dr Nour Elhadi, Principal Investigator, Dr Dipali Shah, Amrinder Sayan & Team,West Suffolk NHS Foundation Trust, West Suffolk Hospital:. Dr Karine Cesar, Principal Investigator, Dr Raman Lakshman, Helen Cockerill. Frimley Health NHS Foundation Trust, Wexham Park Hospital:. Dr Zilla Huma, Principal Investigator,NHS Lanarkshire, Wishaw General Hospital:. Dr Thin Thin Saing, Principal Investigator, Angela Brown, Karen Leitch & Team,University Hospital of South Manchester NHS Foundation Trust, Wythenshawe Hospital:. Dr Gopi Vemuri, Principal Investigator, Claire Holliday, Jessica Carey, Louise Woodhead & Team. Betsi Cadwaladr University Health Board, Ysbyty Gwynedd:. Dr Madalitso Kubwalo, Principal Investigator, Annette Bolger & Team.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
Martin T. Christian reported serving as a member of the Clinical Practice Guideline Group for Steroid Sensitive Nephrotic Syndrome of the International Pediatric Nephrology Association during the conduct of this study. Nicholas J.A. Webb reported being an employee of Novartis since August 2018 outside the submitted work. Nafsika Afentou reported receiving grants from the National Institute for Health Research (NIHR) outside the submitted work. Emma Frew reported receiving grants from the NIHR and Zhejiang Yongning Pharmaceutical Co. Ltd outside the submitted work. Elizabeth A. Brettell reported receiving grants from the NIHR during the conduct of this study. David V. Milford reported receiving grants from the NIHR during the conduct of this study. Natalie J. Ives reported receiving grants from the NIHR during the conduct of this study. Samir Mehta, Rebecca L. Woolley, Adam R. Khan, Detlef Bockenhauer, Moin A. Saleem, Angela S. Hall, Ania Koziell, Heather Maxwell, Shivaram Hegde, Hitesh Prajapati, Rodney D. Gilbert, Caroline Jones, Karl McKeever, and Wendy Cook have no conflicts of interest to declare.
Funding
This project was funded by the NIHR Health Technology Assessment programme.
Author contributions
Nafsika Afentou undertook the health economic evaluation supervised by Emma Frew. Samir Mehta and Rebecca Woolley undertook statistical analyses, supervised by Natalie Ives, and provided statistical input into the economic evaluation and the trial design. Elizabeth Brettell and Adam Khan supervised the central team at Birmingham Clinical Trials Unit and co-ordinated governance of the trial. David Milford, Detlef Bockenhauer, Moin Saleem, Angela Hall, Ania Koziell, Heather Maxwell, Shivaram Hegde, Eric Finlay, Rodney Gilbert, Caroline Jones and Karl McKeever were co-applicants to the NIHR HTA grant and were principal investigators to the trial in their own institutions. Wendy Cook, the director of the Nephrotic Syndrome Trust (NeST), contributed to the trial design and provided input into parent/children-facing documents. Nicholas Webb conceived the idea for the trial, led the design of the protocol, was principal applicant for the NIHR HTA grant and was chief investigator until 2018. Martin Christian was a co-investigator within the grant application and chief investigator to the trial from 2018. Nafsika Afentou and Emma Frew led on writing the manuscript and all authors edited the manuscript.
Ethics approval
Ethics approval for the PREDNOS2 trial was granted by the National Research Ethics Service Committee North West—Greater Manchester Central (reference 12/NW/0766) on 4 December 2012.
Consent to participate
Not applicable.
Consent for publication (from patients/participants)
Not applicable.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Afentou, N., Frew, E., Mehta, S. et al. Economic Evaluation of Using Daily Prednisolone versus Placebo at the Time of an Upper Respiratory Tract Infection for the Management of Children with Steroid-Sensitive Nephrotic Syndrome: A Model-Based Analysis. PharmacoEconomics Open 6, 605–617 (2022). https://doi.org/10.1007/s41669-022-00334-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-022-00334-6