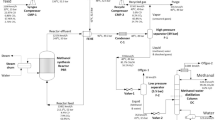
Abstract
The effects of various operating and design parameters in reactive distillation for methanol synthesis using a polar solvent (tetraglyme) and Cu–Zn-Al2O3 catalyst on the process performance are investigated in this simulation work. It is observed that trade-offs exist between the reaction (reactant conversion) and separation (methanol purity) as these parameters are varied. The most sensitive parameters are the solvent flow rate, temperature of feed streams, reflux ratio, and cooling duties on the stages. Following the parametric studies, optimization of the RD column was performed to maximize the methanol production and recovery. The optimum case resulted in a methanol productivity of 2.7 mol/kgcatalyst h which is an increase of 12.5%, and CO and H2 conversions of 44% and 31% vis-à-vis 39% and 28%, respectively, in the base case. The product purity in the distillate for the optimum case also improved to 87% from 82.4% in the base case. The observed catalyst loading in the optimum cases is explained on the basis of the approach to equilibrium for the methanol synthesis reaction. Based on comparison with the conventional packed bed reactor, it is concluded that a more active catalyst for liquid phase methanol synthesis is needed to make RD competitive in terms of methanol productivity.








Similar content being viewed by others
Data Availability
Most of the data generated or analyzed during this study are included in this article and its supplementary information files. It can also be made available from the corresponding author on reasonable request.
Abbreviations
- (r-)WGS:
-
(Reverse) Water gas shift
- DME:
-
Dimethyl ether
- MTBE:
-
Methyl tertiary butyl ether
- PBR:
-
Packed bed (tubular) reactor
- RD:
-
Reactive distillation
- RR:
-
Refluxed rectifier
- RS:
-
Reactive stripper
- TEGDME:
-
Tetraethylene glycol dimethyl ether
- E i :
-
Activation energy for reaction “i”
- F j :
-
Fugacity of species “j”
- k i :
-
Pre-exponential factor of rate expression for reaction “i”
- K eq ,i :
-
Equilibrium constant of reaction “i”
- R :
-
Universal gas constant
- r i :
-
Rate of reaction “i”
- T :
-
Temperature
- T ref :
-
Reference temperature
References
Aspen Plus® v8.4 (2013) AspenTech Inc., Burlington, MA, USA, 2013
Baur R, Krishna R (2004) Distillation column with reactive pump arounds: an alternative to reactive distillation. Chem Eng Process Process Intensif 43:435–445. https://doi.org/10.1016/S0255-2701(03)00128-4
Bozzano G, Manenti F (2016) Efficient methanol synthesis: perspectives, technologies and optimization strategies. Prog Energy Combust Sci 56:71–105. https://doi.org/10.1016/j.pecs.2016.06.001
Bussche KMV, Froment GF (1996) A steady-state kinetic model for methanol synthesis and the water gas shift reaction on a commercial Cu / ZnO / Al 2 O 3 catalyst. J Catal 10:1–10. https://doi.org/10.1006/jcat.1996.0156
Espino RL, Pletzke TS (1975) Methanol production. United States Patent 3(888):896
Ghosh S, Seethamraju S (2019) Feasibility of reactive distillation for methanol synthesis. Chem Eng Process Process Intensif 145:107673. https://doi.org/10.1016/j.cep.2019.107673
Graaf GH, Sijtsema PJJM, Stamhuis EJ, Joosten GEH (1986) Chemical equilibria in methanol synthesis. Chem Eng Sci 41:2883–2890
Graaf GH, Winkelman JGM, Stamhuis EJ, Beenackers AACM (1988) Kinetics of the three phase methanol synthesis. Chem Eng Sci 43:2161–2168. https://doi.org/10.2524/jtappij.17.111
Graaf GH, Smit HJ, Stamhuis EJ, Beenackers AACM (1992) Gas-liquid solubilities of the methanol synthesis components in various solvents. J Chem Eng Data 37:146–158. https://doi.org/10.1021/je00006a004
Hussain A, Chaniago YD, Riaz A, Lee M (2019) Significance of operating pressure on process intensification in a distillation with side-reactor configuration. Sep Purif Technol 213:533–544. https://doi.org/10.1016/j.seppur.2018.12.065
Khosla P, Krishnan C, Elliott R (1991) Binary and multicomponent vapor-liquid equilibria of synthesis gas components, methanol and water with Tetra ethylene glycol dimethyl ether (Tetraglyme). Chem Eng Commun 102:35–46. https://doi.org/10.1080/00986449108910847
Kobl K, Thomas S, Zimmermann Y, Parkhomenko K, Roger AC (2016) Power-law kinetics of methanol synthesis from carbon dioxide and hydrogen on copper-zinc oxide catalysts with alumina or zirconia supports. Catal Today 270:31–42. https://doi.org/10.1016/j.cattod.2015.11.020
Krishnan C, Elliott JR, Berty JM (1991) Continuous operation of the berty reactor for the solvent methanol process. Ind Eng Chem Res 30:1413–1418. https://doi.org/10.1021/ie00055a002
Nemphos, S.P., Groten, W.A., Adams, J.R., 1999. Process for production of methanol. United States Patent 5886055
Nieminen, H., Givirovskiy, G., Laari, A., Koiranen, T., 2018. Alcohol promoted methanol synthesis enhanced by adsorption of water and dual catalysts. J. CO2 Util. 24, 180–189. https://doi.org/https://doi.org/10.1016/j.jcou.2018.01.002
Olah GA, Goeppert A, Surya Prakash GK (2006) Beyond oil and gas: the methanol economy. Wiley-VCH, Weinheim, Germany
Ren S, Shoemaker WR, Wang X, Shang Z, Klinghoffer N, Li S, Yu M, He X, White TA, Liang X (2019) Highly active and selective Cu-ZnO based catalyst for methanol and dimethyl ether synthesis via CO 2 hydrogenation. Fuel 239:1125–1133. https://doi.org/10.1016/j.fuel.2018.11.105
Sage V, Hazewinkel P, Khamphasith M, Young B, Burke N, May EF (2019) High-pressure cryogenic distillation data for improved LNG production. Sep Purif Technol 229:115804
Taylor R, Krishna R (2000) Modelling reactive distillation. Chem Eng Sci 55:5183–5229. https://doi.org/10.1016/S0009-2509(00)00120-2
van der Laan GP, Beenackers AACM, Ding B, Strikwerda JC (1999) Liquid-phase methanol synthesis in apolar (squalane) and polar (tetraethylene glycol dimethylether) solvents. Catal Today 48:93–100. https://doi.org/10.1016/S0920-5861(98)00362-9
Acknowledgements
The authors thank the Industrial Research and Consultancy Centre, Indian Institute of Technology Bombay, and Ministry of Human Resource Development, India, for providing the scholarship to SG during his graduate studentship. We also gratefully acknowledge Aspen Tech for providing the software used in this work. The authors also thank Prof. Ranjan Kumar Malik (Department of Chemical engineering, IIT Bombay) for his suggestions and comments. The authors are also thankful to the anonymous reviewers whose comments helped in increasing the quality of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghosh, S., Seethamraju, S. Process Intensification with a Polar Solvent in Liquid Phase Methanol Synthesis. Process Integr Optim Sustain 5, 827–842 (2021). https://doi.org/10.1007/s41660-021-00180-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41660-021-00180-9