Abstract
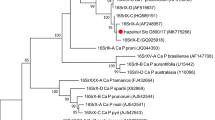
In summer 2018, extensive phytosanitary checks occurred in Tufanbeyli, Adana, Turkey, in a vast forest. These were prompted by significant juniper tree losses with unusual shoot growth and "witches' broom" appearance. To investigate the presence of phytoplasma in the affected juniper trees, nested-PCR assays were employed. Universal phytoplasma-specific primer pairs were used for amplification of the 16S rRNA gene. A total of 300 samples were collected, comprising 242 samples from the witches' brooms displaying symptoms and 58 samples from apparently healthy trees. PCR results showed 90.5% of symptomatic juniper trees tested positive for phytoplasma. All asymptomatic junipers, except three trees, tested negative for phytoplasma in PCR analysis. The nucleotide sequences of 107 PCR amplicons derived from the infected samples were analyzed, revealing a high degree of similarity, ranging from 99.1 to 99.3%, to the 16S ribosomal DNA (rDNA) sequence of Ligustrum witches' broom phytoplasma (LiWBP). It is worth noting that this phytoplasma has been previously documented to infect California privet (Ligustrum ovalifolium) and maize (Zea mays L.) plants in Turkey. Phylogenetic analysis of 16S rDNA identified separate LiWBP strains from Ligustrum, maize, and juniper, forming a distinct clade. This underscores their unique evolutionary lineage and genetic divergence from other known phytoplasma groups. BLAST and in-silico RFLP analysis suggest the presence of a new subgroup or lineage specific to juniper within the LiWBP-related strains. This study unveiled an emerging witches' broom disease in juniper, attributing it to the Ligustrum witches' broom phytoplasma (LiWBP) and a newly discovered subgroup lineage.



Similar content being viewed by others
References
Ahrens U, Seemüller E (1992) Detection of DNA of plant pathogenic mycoplasmalike organisms by a polymerase chain reaction that amplifies a sequence of the 16S rRNA gene. Phytopathology 82:828–832. https://doi.org/10.1094/Phyto-82-828
Bertaccini A, Duduk B, Paltrinieri S, Contaldo N (2014) Phytoplasmas and phytoplasma diseases: a severe threat to agriculture. Am J Plant Sci 5:1763–1788. https://doi.org/10.4236/ajps.2014.512191
Çağlar BK, Elbeaino T (2013) A novel phytoplasma associated with witches’ broom disease of Ligustrum ovalifolium in Turkey. Eur J Plant Pathol 137:113–117. https://doi.org/10.1007/s10658-013-0222-7
Çağlar BK, Pehlivan S, Atakan E, Elbeaino T (2021a) Maize (Zea mays L.): a new host for Ligustrum witches’ broom phytoplasma. Pathogens 10:723. https://doi.org/10.3390/pathogens10060723
Çağlar BK, Şimşek E, Dikilitas M, Bertaccini A (2021b) Characterization of ‘Candidatus Phytoplasma solani’ associated with a maize leaf reddening disease in Turkey. J Phytopathol 169:658–666. https://doi.org/10.1111/JPH.13036
Davis RE, Dally EL, Zhao Y et al (2010) First report of a new subgroup 16SrIX-E (‘Candidatus Phytoplasma phoenicium’-related) phytoplasma associated with juniper witches’ broom disease in Oregon, USA. Plant Pathol 59:1161–1161. https://doi.org/10.1111/j.1365-3059.2010.02294.x
Deng S, Hiruki C (1991) Amplification of 16S rRNA genes from culturable and nonculturable mollicutes. J Microbiol Methods 14:53–61. https://doi.org/10.1016/0167-7012(91)90007-D
Dijkstra J, Lee PE (1972) Transmission by dodder of sandal spike disease and the accompanying mycoplasma-like organisms via Vinca rosea. Neth J Plant Pathol 78:218–224. https://doi.org/10.1007/BF01977322
Duduk B, Paltrinieri S, Lee I-M, Bertaccini A (2013) Nested PCR and RFLP analysis based on the 16S rRNA gene. In: Dickinson M, Hodgetts J (eds) Phytoplasma methods and protocols. Humana Press, Totowa, pp 159–171. https://doi.org/10.1007/978-1-62703-089-2_14
Gülcü S, Demir S, Dirlik S (2019) Boylu ardıç’ta (Juniperus excelsa Bieb.) bazı kozalak ve tohum özellikleri bakımından populasyonlar arası farklılıklar. Turk J for 20(3):187–194. https://doi.org/10.18182/tjf.561242
Gundersen D, Lee I (1996) Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopathol Mediter 35(3):144–151. http://www.jstor.org/stable/42685262
Hennon PE, Frankel SJ, Woods AJ et al (2020) A framework to evaluate climate effects on forest tree diseases. For Pathol 50:e12649. https://doi.org/10.1111/efp.12649
Krishnareddy M (2013) Impact of climate change on ınsect vectors and vector-borne plant viruses and phytoplasma. In: Singh HCP, Rao NKS, Shivashankar KS (eds) Climate-Resilient horticulture: adaptation and mitigation strategies. Springer, India, pp 255–277. https://doi.org/10.1007/978-81-322-0974-4_23
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Lee IM, Hammond RW, Davis RE, Gundersen DE (1993) Universal amplification and analysis of pathogen 16S rDNA for classification and identification of mycoplasmalike organisms. Phytopathology 83:834–842
Lehtijärvi A, Oskay F, Aday Kaya AG, et al (2017) Emerging tree diseases in Turkey. In: International symposium of new horizons in forestry, 18–20 October 2017, Isparta Turkey, p 377. http://ormanweb.isparta.edu.tr/isfor2017/documents/pdf/377.pdf. Accessed 03 Aug 2023
Marcone C (2017) Elm yellows: a phytoplasma disease of concern in forest and landscape ecosystems. For Pathol 47:e12324. https://doi.org/10.1111/efp.12324
Marcone C, Franco-Lara L, Toševski I (2018) Major phytoplasma diseases of forest and urban trees. In: Rao GP, Bertaccini A, Fiore N, Liefting LW (eds) Phytoplasmas: plant pathogenic bacteria—I: characterisation and epidemiology of phytoplasma—associated diseases. Springer, Singapore, pp 287–312. https://doi.org/10.1007/978-981-13-0119-3_10
Marcone C, Valiunas D, Mondal S, Sundararaj R (2021) On some significant phytoplasma diseases of forest trees: an update. Forests 12(4):408. https://doi.org/10.3390/f12040408
Ozturk M, Gucel S, Kucuk M, Sakcali S (2010) Forest diversity, climate change and forest fires in the Mediterranean region of Turkey. J Environ Biol 31(1–2): 1–9. http://jeb.co.in/journal_issues/201001_jan10/paper_01.pdf. Accessed 03 Aug 2023
Stenlid J, Oliva J, Boberg JB, Hopkins AJM (2011) Emerging diseases in European forest ecosystems and responses in society. Forests 2:486–504. https://doi.org/10.3390/f2020486
Wei W, Zhao Y (2022) Phytoplasma taxonomy: nomenclature, classification, and identification. Biology 11:1119. https://doi.org/10.3390/biology11081119
Yücedağ C, Gailing O (2013) Genetic variation and differentiation in Juniperus excelsa M. Bieb. populations in Turkey. Trees 27:547–554. https://doi.org/10.1007/s00468-012-0807-3
Zamharir MG, Razavi M, Rabbaninasab H (2019) First report of ‘candidatus phytoplasma trifolii’-related strain presence in juniperus procubens witches’ broom in Iran. Phytopathogen. Moll. 9:310–313. https://doi.org/10.5958/2249-4677.2019.00130.0
Acknowledgements
We would like to express our gratitude to forest engineers for their contribution in identifying tree species.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval of human or animal rights
All authors are fully aware of this submission and have declared that they have no competing interests. This article does not contain any studies with human or animal subjects performed by any of the authors.
Informed consent
All authors have reviewed the manuscript and approved its submission to Journal of Plant Disease and Protection.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Çağlar, B.K., Şimşek, E. & Elbeaino, T. Ligustrum witches broom phytoplasma caused witches’ broom disease for Juniperus excelsa Bieb. in Turkey. J Plant Dis Prot 131, 515–522 (2024). https://doi.org/10.1007/s41348-023-00822-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-023-00822-9