Abstract
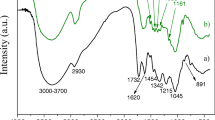
The present study aimed primarily to demonstrate the potential use of mastic tree leaves from which the essential oils have been extracted to remove rhodamine B and ethyl violet from aqueous solutions through biosorption. Pistacia lentiscus (the mastic tree) is widespread in the Mediterranean region. Chemical characterization of the biosorbent was conducted by determining the pH at the point of zero charge and the surface functional groups present through the use of Boehm titration and Fourier-transform infrared spectroscopy. Physical characterization of the material was carried out by scanning electron microscopy, laser diffraction, and BET analysis. The results obtained showed that this material, which has good porosity, can be used as a biosorbent. The findings revealed that the biosorbent surface was slightly acid, with a predominance of phenolic groups. The effects of different parameters such as the contact time, initial dye concentration, biosorbent dosage, initial pH value, temperature, and ionic strength were all optimized. Both dyes (rhodamine B and ethyl violet) exhibited similar isotherm profiles that were type L according to the Brunauer–Emmett–Teller classification and type II according to the International Union of Pure and Applied Chemistry classification. The multilayer adsorption of both dyes was best described by the Freundlich isotherm model. In addition, the biosorption process was found to follow pseudo-second-order kinetics, and both reactions were thermodynamically spontaneous, with the biosorbent showing a remarkable affinity for the dyes. For ethyl violet, the adsorption yield was 88% and the adsorption capacity was 17 mg g−1 at equilibrium; however, for rhodamine B, the adsorption yield was 90% and the adsorption capacity was 9 mg g−1 at equilibrium.
















Similar content being viewed by others
References
Afef D, Henda S, Wahida B, Abderrahman B, Nabil C (2013) Antiinflammatory and antiulcerogenic activities of Pistacialentiscus L. leaves extracts. Ind Crops Prod 49:879–882. https://doi.org/10.1016/j.indcrop.2013.07.010
Alencar WS, Acayanka E, Lima EC, Royer B, de Souza FE, Lameira J, Alves CN (2012) Application of Mangiferaindica (mango) seeds as a biosorbent for removal of Victazol Orange 3R dye from aqueous solution and study of the biosorption mechanism. Chem Eng J 209:577–588. https://doi.org/10.1016/j.cej.2012.08.053
Al-Jobouri IS, Dhahir SA, Al-Saade KA (2013) Adsorption study of rhodamin B dye on Iraqi bentonite and modified bentonite by nanocompounds TiO2, Zno, Al2O3 and sodium dodecyl sulfate. Am J Environ Sci 9(3):269–279. https://doi.org/10.3844/ajessp.2013.269.279
Anantha RK, Kota S (2018) Bio-composites for the sorption of copper from aqueous solution: a comparative study. Groundw Sustain Dev 7:265–276. https://doi.org/10.1016/j.gsd.2018.06.007
Arfi RB, Karoui S, Mougin K, Ghorbal A (2017) Adsorptive removal of cationic and anionic dyes from aqueous solution by utilizing almond shell as bioadsorbent. Euro-Mediterr J Environ Integr. https://doi.org/10.1007/s41207-017-0032-y
Asim O, Necip A (2009) Equilibrium and kinetic adsorption study of Basic Yellow 28 and Basic Red 46 by a boron industry waste. J Hazard Mater 161:148–156. https://doi.org/10.1016/j.jhazmat.2008.03.064
Azharul Islam Md, Ahmed MJ, Khanday WA, Asif M, Hameed BH (2017) Mesoporous activated coconut shell-derived hydrochar prepared via hydrothermal carbonization-NaOH activation for methylene blue adsorption. J Environ Manage 203:237–244. https://doi.org/10.1016/j.jenvman.2017.07.029
Bachrouch O, Jemâa J-B, Wissem AW, Talou T, Marzouk B, Abderraba M (2010) Composition and insecticidal activity of essential oil from Pistacialentiscus L. against Ectomyeloisceratoniae Zeller and Ephestiakuehniella Zeller (Lepidoptera: Pyralidae). J Stored Prod Res 46:242–247. https://doi.org/10.1016/j.jspr.2010.07.001
Banerjee M, Basu RK, Das SK (2018) Cr(VI) adsorption by a green adsorbent walnut shell: adsorption studies, regeneration studies, scale-up design and economic feasibility. Process Saf Environ Prot. https://doi.org/10.1016/j.psep.2018.03.037
Barka N, Assabbane A, Nounah A, Laanab L, Ichoua YA (2009) Removal of textile dyes from aqueous solutions by natural phosphate as a new adsorbent. Desalination 2009:264–275. https://doi.org/10.1016/j.desal.2008.01.015
Bozbas SK, Boz Y (2016) Low-cost biosorbent: Anadarainaequivalvis shells for removal of Pb(II) and Cu(II) from aqueous solution. Process Saf Environ Prot 103:144–152. https://doi.org/10.1016/j.psep.2016.07.007
Chen H, Dou J, Hongbin Xu (2017) Removal of Cr(VI) ions by sewage sludge compost biomass from aqueous solutions: reduction to Cr(III) and biosorption. Appl Surf Sci 245:728–735. https://doi.org/10.1016/j.apsusc.2017.07.053
Chen Y, Wang B, Xin J, Sun P, Dan Wu (2018) Adsorption behavior and mechanism of Cr(VI) by modified biochar derived from Enteromorphaprolifera. Ecotoxicol Environ Saf 164:440–447. https://doi.org/10.1016/j.ecoenv.2018.08.024
Das SK, Ghosh P, Ghosh I, Guha AK (2008) Adsorption of rhodamine B on Rhizopus oryzae: role of functional groups and cellwall components. Colloids Surf B 65:30–34. https://doi.org/10.1016/j.colsurfb.2008.02.020
Divine AG, Sumalinog SC, Capareda MD, de Luna G (2018) Evaluation of the effectiveness and mechanisms of acetaminophen and methylene blue dye adsorption on activated biochar derived from municipal solid wastes. J Environ Manage. https://doi.org/10.1016/j.jenvman.2018.01.010
Djenane D, Yanguela J, Montañés L, Djerbal M, Roncalés P (2011) Antimicrobial activity of Pistacialentiscus and Saturejamontana essential oils against Listeria monocytogens CECT 935 using laboratory media: efficacy and synergistic potential in minced beef. Food Control 22:1046–1053. https://doi.org/10.1016/j.foodcont.2010.12.015
el Alouani M, Alehyen S, EL Achouri M, Taibi M (2018) Removal of cationic dye—methylene blue—from aqueous solution by adsorption on fly ash-based geopolymer. J Mater Environ Sci 9(1):32–46. https://doi.org/10.26872/jmes.2018.9.1.5
El Haddad M, Mamouni R, Saffaj N, Lazar S (2012) Adsorptive removal of basic dye rhodamine B from aquoeus media onto animal bone meal as new low cost adsorbent. Glob J Hum Soc Sci Geogr Environ Geosci 12:19–29
El Haddad M, Slimani R, Mamouni R, Laamari MR, Rafqah S, Lazar S (2013) Evaluation of potential capability of calcined bones on the biosorption removal efficiency of safranin as cationic dye from aqueous solutions. J Taiwan Inst Chem Eng 44:13–18. https://doi.org/10.1016/j.jtice.2012.10.003
El Haddad M, Regti A, Slimani R, Lazar S (2014) Assessment of the biosorption kinetic and thermodynamic for the removal of safranin dye from aqueous solutions using calcined mussel shells. J Ind Eng Chem 20:717–724. https://doi.org/10.1016/j.jiec.2013.05.038
El-Bayaa AA, Badawy NA, Abd AlKhalik E (2009) Effect of ionic strength on the adsorption of copper and chromium ions by vermiculite pure clay mineral. J Hazard Mater 170:1204–1209. https://doi.org/10.1016/j.jhazmat.2009.05.100
Fatih D, Abdulkerim K (2017) Biosorption of heavy metal ions by chemically modified biomass of coastal seaweed community: studies on phycoremediation system modeling and design. Ecol Eng 106:101–108. https://doi.org/10.1016/j.ecoleng.2017.05.024
Gaurav Sharm Mu, Naushad AK, Rana S, Sharma S, Bhatnagar A, Stadler FJ, Ghfar AA, Khan MR (2017) Efficient removal of Coomassie Brilliant Blue R-250 dye using starch/poly(alginic acid-cl-acrylamide) nanohydrogel. Process Saf Environ Prot 109:301–310. https://doi.org/10.1016/j.psep.2017.04.011
Ghasemi J, Asadpour S (2007) Thermodynamics’ study of the adsorption process of methylene blue on activated carbon at different ionic strengths. J Chem Thermodyn 39:967–971. https://doi.org/10.1016/j.jct.2006.10.018
Glossman-Mitnik D (2013) Computational study of the chemical reactivity properties of the rhodamine B molecule. Procedia Comput Sci 18:816–825. https://doi.org/10.1016/j.procs.2013.05.246
Herrera-González AM, Caldera-Villalobos M, Peláez-Cid A-A (2019) Adsorption of textile dyes using an activated carbon and crosslinked polyvinyl phosphonic acid composite. J Environ Manage 234:237–244. https://doi.org/10.1016/j.jenvman.2019.01.012
Huang JH, Huang KL, Su-Qin Liu AT, Wang CY (2008) Adsorption of rhodamine B and methyl orange on a hypercrosslinked polymeric adsorbent in aqueous solution. Colloids Surf A Physicochem Eng Aspects 330:55–61. https://doi.org/10.1016/j.colsurfa.2008.07.050
Humelnicu I, Baiceanu A, Ignat ME, Dulman V (2016) The removal of Basic Blue 41 textile dye from aqueous solution by adsorption onto natural zeolitic tuff: kinetics and thermodynamics. Process Saf Environ Protect. https://doi.org/10.1016/j.psep.2016.11.016
Ibrahim WM, Hassan AF, Azab YA (2016) Biosorption of toxic heavy metals from aqueous solution by Ulva lactuca activated carbon. Egypt J Basic Appl Sci 3:241–249. https://doi.org/10.1016/j.ejbas.2016.07.005
Jiaxing L, Jun H, Guodong S, Guixia Z, Qing H (2009) Effect of pH, ionic strength, foreign ions and temperature on the adsorption of Cu(II) from aqueous solution to GMZ bentonite. Colloids Surf A Physicochem Eng Aspects 349:195–201. https://doi.org/10.1016/j.colsurfa.2009.08.018
Kebir M, Trari M, Maachi R, Nasrallah N, Bellal B, Amrane A (2015) Relevance of a hybrid process coupling adsorption and visible light photocatalysis involving a new hetero-system CuCo2O4/TiO2 for the removal of hexavalent chromium. J Environ Chem Eng 3:548–559. https://doi.org/10.1016/j.jece.2014.12.024
Khan TA, Dahiya S, Ali I (2012) Use of kaolinite as adsorbent: equilibrium, dynamics and thermodynamic studies on the adsorption of rhodamine B from aqueous solution. Appl Clay Sci 69:58–66. https://doi.org/10.1016/j.clay.2012.09.001
Landau S, Azaizehb H, Muklada H, Glasser T, Ungar ED, Baram H, Abbas N, Markovics A (2010) Anthelmintic activity of Pistacialentiscus foliage in two Middle Eastern breeds of goats differing in their propensity to consume tannin-rich browse. Vet Parasitol 173(280–286):280. https://doi.org/10.1016/j.vetpar.2010.07.006
Lim LBL, Namal Priyantha DTB, Tennakoon HI, Chieng MK, Dahri MS (2013) Breadnut peel as a highly effective low-cost biosorbent for methylene blue: equilibrium, thermodynamic and kinetic studies. Arab J Chem. https://doi.org/10.1016/j.arabjc.2013.12.018
Lishuang H, Guang C, Liu Y, Zengqiang Su, Gong S, Yao Y, Wang Y (2020) Adsorption behavior of dyes from an aqueous solution onto composite magnetic lignin adsorbent. Chemosphere 246:125757. https://doi.org/10.1016/j.chemosphere.2019.125757
Liu Z, Zhang F, Liu T, Peng N, Gai C (2016) Removal of azo dye by a highly graphitized and heteroatom doped carbon derived from fish waste: adsorption equilibrium and kinetics. J Environ Manage 182:446–454. https://doi.org/10.1016/j.jenvman.2016.08.008
Mall ID, Srivastava VC, Agarwal NK (2006) Removal of Orange-G and Methyl Violet dyes by adsorption onto bagasse fly ash—kinetic study and equilibrium isotherm analyses. Dyes Pigm 69:210–223. https://doi.org/10.1016/j.dyepig.2005.03.013
Mohapatra RK, Parhi PK, Pandey S, Bindhani BK, Thatoi H, Panda CR (2019) Active and passive biosorption of Pb(II) using live and dead biomass of marine bacterium Bacillusxiamenensis PbRPSD202: kinetics and isotherm studies. J Environ Manage 247:121–134. https://doi.org/10.1016/j.jenvman.2019.06.073
Nguyen TA, Chun-Chieh Fu, Juang R-S (2016) Biosorption and biodegradation of a sulfur dye in high-strength dyeing wastewater by Acidithiobacillusthiooxidans. J Environ Manage 182:265–271. https://doi.org/10.1016/j.jenvman.2016.07.083
Nidheesh PV, Gandhimathi R (2013) Degradation of dyes from aqueous solution by Fenton processes: a review. Environ Sci Pollut Res 20:2099–2132. https://doi.org/10.1007/s11356-012-1385-z
Ogundipe KD, Babarinde A (2017) Comparative study on batch equilibrium biosorption of Cd(II), Pb(II) and Zn(II) using plantain (Musaparadisiaca) flower: kinetics, isotherm, and thermodynamics. Dipe Babarinde Chem Int 3(2):135–149
Othmani A, Kesraoui A, Seffen M (2017) The alternating and direct current effect on the elimination of cationic and anionic dye from aqueous solutions by electrocoagulation and coagulation flocculation. Euro-Mediterr J Environ Integr. https://doi.org/10.1007/s41207-017-0016-y
Paraschos S, Magiatis P, Gousia P, Economou V, Sakkas H, Papadopoulou C, Skaltsounis A-L (2011) Chemical investigation and antimicrobial properties of mastic water and its major constituents. Food Chem 129:907–911. https://doi.org/10.1016/j.foodchem.2011.05.043
Predrag L, Hui S, Uri C, Hassan A, Arieh B (2005) The effects of aqueous extracts prepared from the leaves of Pistacialentiscus in experimental liver disease. J Ethnopharmacol 100:198–204. https://doi.org/10.1016/j.jep.2005.03.006
Rabah H, Khaldi K, Choukchou-Braham A, Lerari-Zinai D, Bachari K (2018) Comparative study of natural and synthetic clays used as supported catalysts in dyes degradation by advanced oxidation processes. Euro-Mediterr J Environ Integr. https://doi.org/10.1007/978-3-319-70548-4_71
Rauf MA, Bukallah SB, Hamour FA, Nasir AS (2008) Adsorption of dyes from aqueous solutions onto sand and their kinetic behavior. Chem Eng J 137:238–243. https://doi.org/10.1016/j.cej.2007.04.025
Saranya K, Sundaramanickam A, Shekhar S, Moorthy M, Rengasamy Subramaniyan S, Thangavel B (2018) Biosorption of multi-heavy metals by coral associated phosphate solubilising bacteria Cronobactermuytjensii KSCAS2. J Environ Manag 222:396–401. https://doi.org/10.1016/j.jenvman.2018.05.083
Saulo LC, Stefanne C, Costa D, Emily N, Gurgel M, da Silva C, Gurgel M, Vieira A (2017) Biosorption of toxic metals using the alginate extraction residue from the brown algae Sargassumfilipendula as a natural ion-exchanger. J Clean Prod 165:491–499. https://doi.org/10.1016/j.jclepro.2017.07.114
Shen Yi, Zhang Z, Xiao K (2015) Evaluation of cobalt oxide, copper oxide and their solid solutions as heterogeneous catalysts for Fenton-degradation of dye pollutants. RSC Adv. https://doi.org/10.1039/C5RA18923C
Tsai WT, Chang YM, Lai CW, Lo CC (2005) Adsorption of ethyl violet dye in aqueous solution by regenerated spent bleaching earth. J Colloid Interface Sci 289:333–338. https://doi.org/10.1016/j.jcis.2005.03.087
Tsechansky L, Graber ER (2014) Methodological limitations to determining acidic groups at biochar surfaces via the Boehm titration. Carbon 66(73):70–733. https://doi.org/10.1016/j.carbon.2013.09.044
Varlikli C, Bekiarb V, Kus M, Boduroglu N, Oner I, Lianos P, Lyberatos G, Icl S (2009) Adsorption of dyes on Sahara desert sand. J Hazard Mater 170:27–34. https://doi.org/10.1016/j.jhazmat.2009.05.030
Wahl V, Saurugger E, Khinast J, Laggner P (2015) Specific surface, crystallinity, and dissolution of lyophilized fibrinogen. Eur J Pharm Biopharm 89:374–382. https://doi.org/10.1016/j.ejpb.2014.12.018
Wencai C, Congcong D, Xiangxue W, Zhenyu Wu, Yubing S, Shuhong Yu et al (2016) Competitive sorption of As(V) and Cr(VI) on carbonaceous nanofibers. Chem Eng J 293:311–318. https://doi.org/10.1016/j.cej.2016.02.073
Xin-jiang H, Liu Y, Wang H, Zeng G, Xi H, Guo Y, Li T, Chen A, Jiang L, Guo F (2015) Adsorption of copper by magnetic graphene oxide-supported B-cyclodextrin: effects of pH, ionic strength, background electrolytes, and citric acid. Chem Eng Res Des 93:675–683. https://doi.org/10.1016/j.cherd.2014.06.002
Yıldırım H (2012) Micropropagation of Pistacialentiscus L. from axenic seedling-derived explants. Sci Hortic 137:29–35. https://doi.org/10.1016/j.scienta.2012.01.020
Yip A-K, Lam F-Y, Xijun Hu (2005) Chemical-vapor-deposited copper on acid-activated bentonite clay as an applicable heterogeneous catalyst for the photo-Fenton-like oxidation of textile organic pollutants. Ind Eng Chem Res 44:7983–7990
Youyuan C, Baoying W, Xin J, Ping S, Dan Wu (2018) Adsorption behavior and mechanism of Cr(VI) by modified biochar derived from Enteromorphaprolifera. Ecotoxicol Environ Saf 164:440–447. https://doi.org/10.1016/j.ecoenv.2018.08.024
Zanin E, Scapinello J, de Oliveira M, Rambo CL, Franscescon F, Freitas L, Muneron JM, de Melloa M, Antonio Fioria J, Oliveira V, Dal Magro J (2014) Adsorption of heavy metals from wastewater graphic industry using clinoptilolite zeolite as adsorbent. Process Saf Environ Prot 105:154–200. https://doi.org/10.1016/j.psep.2016.11.008
Zouhir D (2014) Anti-hypercholesterolemic effect of Pistacialentiscus fatty oil in egg yolk-fed rabbits: a comparative study with simvastatin. Chin J Nat Med 12(8):0561–0566
Funding
No sources, because it is a state university.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all the authors, the corresponding author states that there is no conflict of interest.
Additional information
Responsible Editor: Mongi Seffen.
Rights and permissions
About this article
Cite this article
Bensenane Dhina, F.K., Nassrallah, N., Choukchou-Braham, A. et al. Use of Pistacia lentiscus leaves, after extraction of their oil, as a new biosorbent for the removal of dyes from water. Euro-Mediterr J Environ Integr 6, 55 (2021). https://doi.org/10.1007/s41207-021-00255-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41207-021-00255-6