Abstract
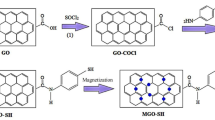
Graphene oxide (GO) was chemically modified (MGO) by reaction with tetraethyl orthosilicate and barbituric acid to obtain its modified form. The chemical structures of GO and its modified form were established using XRD, FTIR, SEM, and BET (N2-adsorption/desorption) analysis. The prepared GO and MGO were evaluated as efficient metal ions adsorbents during the remediation of copper and iron ions in wastewater. The parameters controlling the adsorption process were examined including: the mass of adsorbents, pH of the medium, and concentration of ionic species, temperature, and process time. The results revealed that the maximum adsorption capacities of Cu(II) and Fe(III)metal ions were GO's 19.99 and 21.6 mg/g, compared to MGO at 24.1 mg/g and 25.13 mg/g, receptively. After 105 min in a solution pH = 6 at 25 °C, using 0.35 g/L of the adsorbent. Additional increase in contact time did not show considerable change in equilibrium concentration. That indicates to the adsorption stage reach equilibrium. The adsorption isotherms could be fitted well by the Langmuir model referring to correlation factor around 1, indicating that the adsorption of the metal ion onto GO and MGO is favorable. DFT study described the increasing in the adsorption efficiency of the MGO.









Similar content being viewed by others
Data availability
The datasets generated during and/or analyses during the current study are available from the corresponding author on reasonable request.
References
Kumar PS, Gayathri R, Rathi BS (2021) A review on adsorptive separation of toxic metals from aquatic system using biochar produced from agro-waste. Chemosphere 285:131438. https://doi.org/10.1016/j.chemosphere.2021.131438
Soliman NK, Moustafa AF (2020) Industrial solid waste for heavy metals adsorption features and challenges; a review. J Mater Res Technol 9:10235–10253. https://doi.org/10.1016/j.jmrt.2020.07.045
Sharma P, Pandey AK, Kim SH, Singh SP, Chaturvedi P, Varjani S (2021) Critical review on microbial community during in-situ bioremediation of heavy metals from industrial wastewater. Environ Technol Innov 24:101826. https://doi.org/10.1016/j.eti.2021.101826
Bu X, Song Y, Pan J, Wang X, Qin C, Jia Y, Du Z, Qin JG, Chen L (2022) Toxicity of chronic copper exposure on Chinese mitten crab (Eriocheir sinensis) and mitigation of its adverse impact by myo-inositol. Aquaculture 547:737511. https://doi.org/10.1016/j.aquaculture.2021.737511
Rivella S, Crielaard BJ (2014) Disorders of iron metabolism: iron deficiency and iron overload and anemia of chronic diseases. In: McManus LM, Mitchell RN (eds) Pathobiology of human disease. Academic Press, San Diego, pp 1471–1487
M.M. Galal, (2015) Environmental Impact Assessment of Wastewater Treatment Plants - (Zenien and 6th of October WWTP), 4:953–964.
Mouratib R, Achiou B, El Krati M, Younssi SA, Tahiri S (2020) Low-cost ceramic membrane made from alumina- and silica-rich water treatment sludge and its application to wastewater filtration. J Eur Ceram Soc 40:5942–5950. https://doi.org/10.1016/j.jeurceramsoc.2020.07.050
Pal P, Kumar R (2020) Chapter 2-Recent advances in biological treatment processes for wastewater and water treatment. In: Basile A, Ghasemzadeh K (eds) Current trends and future developments on (bio-) membranes, 1st edn. Elsevier, pp 41–66
Kodispathi T, Jacinth Mispa K (2021) Fabrication, characterization, ion-exchange studies and binary separation of polyaniline/Ti(IV) iodotungstate composite ion-exchanger for the treatment of water pollutants. Environ Nanotechnol Monit Manag 16:100555. https://doi.org/10.1016/j.enmm.2021.100555
Baynosa ML, Mady AH, Nguyen VQ, Kumar DR, Sayed MS, Tuma D, Shim J-J (2020) Eco-friendly synthesis of recyclable mesoporous zinc ferrite@reduced graphene oxide nanocomposite for efficient photocatalytic dye degradation under solar radiation. J Colloid Interface Sci 561:459–469. https://doi.org/10.1016/j.jcis.2019.11.018
Nguyen VQ, Mady AH, Mahadadalkar MA, Baynosa ML, Kumar DR, Rabie AM, Lee J, Kim WK, Shim J-J (2022) Highly active Z-scheme heterojunction photocatalyst of anatase TiO2 octahedra covered with C-MoS2 nanosheets for efficient degradation of organic pollutants under solar light. J Colloid Interface Sci 606:337–352. https://doi.org/10.1016/j.jcis.2021.07.128
Ozbey-Unal B, Omwene PI, Yagcioglu M, Balcik-Canbolat Ç, Karagunduz A, Keskinler B, Dizge N (2020) Treatment of organized industrial zone wastewater by microfiltration/reverse osmosis membrane process for water recovery: from lab to pilot scale. J Water Process Eng 38:101646. https://doi.org/10.1016/j.jwpe.2020.101646
Hafez OA, Mohamed R, Kana MA, Mohamed E, Negm N (2021) Treatment of industrial wastewater containing copper and lead ions using new carboxymethyl chitosan-activated carbon derivatives. Egypt J Chem. https://doi.org/10.21608/ejchem.2021.82163.4050
Ashour RM, Abdelhamid HN, Abdel-Magied AF, Abdel-Khalek AA, Ali MM, Uheida A, Muhammed M, Zou X, Dutta J (2017) Rare earth ions adsorption onto graphene oxide nanosheets. Solvent Extr Ion Exch 35:91–103. https://doi.org/10.1080/07366299.2017.1287509
Abubshait HA, Farag AA, El-Raouf MA, Negm NA, Mohamed EA (2021) Graphene oxide modified thiosemicarbazide nanocomposite as an effective eliminator for heavy metal ions. J Mol Liq 327:114790. https://doi.org/10.1016/j.molliq.2020.114790
Amer A, Sayed GH, Ramadan RM, Rabie AM, Negm NA, Farag AA, Mohammed EA (2021) Assessment of 3-amino-1H-1,2,4-triazole modified layered double hydroxide in effective remediation of heavy metal ions from aqueous environment. J Mol Liq 341:116935. https://doi.org/10.1016/j.molliq.2021.116935
Guo T, Bulin C, Ma Z, Li B, Zhang Y, Zhang B, Xing R, Ge X (2021) Mechanism of Cd(II) and Cu(II) adsorption onto few-layered magnetic graphene oxide as an efficient adsorbent. ACS Omega 6:16535–16545. https://doi.org/10.1021/acsomega.1c01770
Sadeek SA, Negm NA, Hefni HH, Wahab MA (2015) Metal adsorption by agricultural biosorbents: adsorption isotherm, kinetic and biosorbents chemical structures. Int J Biol Macromol 81:400–409. https://doi.org/10.1016/j.ijbiomac.2015.08.031
Aragaw TA, Ayalew AA (2019) Removal of water hardness using zeolite synthesized from Ethiopian kaolin by hydrothermal method. Water Pract Technol 14:145–159. https://doi.org/10.2166/wpt.2018.116
Hussein AA (2019) Adsorption of lead ions from aqueous solution by using sunflower husks. Int Res J Innov Eng Technol 3(8):7–11
Chen H, Li Y, Deng M, Lu J (2019) Study on the absorption effect of hydrolyzed hoof horn powder on cadmium ions. IOP Conf Ser Earth Environ Sci 384:12070. https://doi.org/10.1088/1755-1315/384/1/012070
Negm NA, Abubshait HA, Abubshait SA, Abou Kana MTH, Mohamed EA, Betiha MM (2020) Performance of chitosan polymer as platform during sensors fabrication and sensing applications. Int J Biol Macromol 165:402–435. https://doi.org/10.1016/j.ijbiomac.2020.09.130
Gao J, Du Z, Zhao Q, Guo Y, Cheng F (2021) Enhanced Li+ adsorption by magnetically recyclable iron-doped lithium manganese oxide ion-sieve: synthesis, characterization, adsorption kinetics and isotherm. J Mater Res Technol 13:228–240. https://doi.org/10.1016/j.jmrt.2021.04.073
Argun ME, Dursun S, Ozdemir C, Karatas M (2007) Heavy metal adsorption by modified oak sawdust: thermodynamics and kinetics. J Hazard Mater 141:77–85. https://doi.org/10.1016/j.jhazmat.2006.06.095
Wadhawan S, Jain A, Nayyar J, Mehta SK (2020) Role of nanomaterials as adsorbents in heavy metal ion removal from waste water: a review. J. Water Process Eng 33:101038. https://doi.org/10.1016/j.jwpe.2019.101038
Sadegh H, Ali GAM, Gupta VK, Makhlouf ASH, Shahryari-ghoshekandi R, Nadagouda MN, Sillanpää M, Megiel E (2017) The role of nanomaterials as effective adsorbents and their applications in wastewater treatment. J Nanostruct Chem 7:1–14. https://doi.org/10.1007/s40097-017-0219-4
Khorshidi P, Shirazi RHSM, Miralinaghi M, Moniri E, Saadi S (2020) Adsorptive removal of mercury (II), copper (II), and lead (II) ions from aqueous solutions using glutathione-functionalized NiFe2O4/graphene oxide composite. Res Chem Intermed 46:3607–3627. https://doi.org/10.1007/s11164-020-04164-1
Politaeva N, Yakovlev A, Yakovleva E, Chelysheva V, Tarantseva K, Efremova S, Mukhametova L, Ilyashenko S (2022) Graphene oxide-chitosan composites for water treatment from copper cations. Water 14:1430. https://doi.org/10.3390/w14091430
Kumari P, Tripathi KM, Jangir LK, Gupta R, Awasthi K (2021) Recent advances in application of the graphene-based membrane for water purification. Mater. Today Chem. 22:100597. https://doi.org/10.1016/j.mtchem.2021.100597
Karanjikar SR, Singh Sena A, Manekar P, Mudagi S, Singh Juneja A (2021) Utilization of graphene and its derivatives for air & water filtration: a review. Mater Today Proc 50:2007–2017. https://doi.org/10.1016/j.matpr.2021.09.346
Hou D, Li K, Ma R, Liu Q (2020) Influence of order degree of coaly graphite on its structure change during preparation of graphene oxide. J Mater 6:628–641. https://doi.org/10.1016/j.jmat.2020.04.009
Sahoo P, Shubhadarshinee L, Jali BR, Mohapatra P, Barick AK (2022) Synthesis and characterization of graphene oxide and graphene from coal. Mater Today Proc 56:2421–2427. https://doi.org/10.1016/j.matpr.2021.08.206
Dong Y, Li J, Shi L, Xu J, Wang X, Guo Z, Liu W (2013) Graphene oxide-iron complex: synthesis, characterization and visible-light-driven photocatalysis. J Mater Chem A 1:644–650. https://doi.org/10.1039/c2ta00371f
Zhang C, Fu X, Zhang X, Li J, Fan X, Zhang G (2020) The effects of metal complexes of nano-graphene oxide to thermal decomposition of FOX-7. Nanomaterials 10:144. https://doi.org/10.3390/nano10010144
Fan JP, Mao DY, Zhang XH, Qi GX, Liao DD, Chen HP, Huang K (2020) Preparation and characterization of a novel freestanding flexible reduced graphene oxide composite membrane for adsorption of isoflavone in Radix Puerariae Lobatae. Colloids Surfaces A Physicochem Eng Asp 599:124911. https://doi.org/10.1016/j.colsurfa.2020.124911
Abdelhamid HN, Wu H (2015) Reduced graphene oxide conjugate thymine as a new probe for ultrasensitive and selective fluorometric determination of mercury(II) ions. Microchim Acta 182:1609–1617. https://doi.org/10.1007/s00604-015-1461-4
Paulchamy B, Arthi G, Lignesh BD (2015) A simple approach to stepwise synthesis of graphene oxide nanomaterial. J Nanomed Nanotechnol 06:1–4. https://doi.org/10.4172/2157-7439.1000253
Sheet I, Kabbani A, Holail H (2014) Removal of heavy metals using nanostructured graphite oxide, silica nanoparticles and silica/graphite oxide composite. Energy Proced 50:130–138. https://doi.org/10.1016/j.egypro.2014.06.016
Kausar A, Rafique I, Anwar Z, Muhammad B (2015) Perspectives of epoxy/graphene oxide composite: significant features and technical applications. Polym Plast Technol Eng. https://doi.org/10.1080/03602559.2015.10987700
Bouibed A, Doufnoune R (2019) Synthesis and characterization of hybrid materials based on graphene oxide and silica nanoparticles and their effect on the corrosion protection properties of epoxy resin coatings. J Adhes Sci Technol 33:834–860. https://doi.org/10.1080/01694243.2019.1571660
Pourhashem S, Vaezi M, Rashidi A, Bagherzadeh MR (2017) Distinctive roles of silane coupling agents on the corrosion inhibition performance of graphene oxide in epoxy coatings. Prog Org Coat 111:47–56. https://doi.org/10.1016/j.porgcoat.2017.05.008
Feigao X, Wang C, Li D, Wang M, Fei X, Deng X (2015) Preparation of modified epoxy–SiO2 hybrid materials and their application in the stone protection. Prog Org Coat 81:58–65. https://doi.org/10.1016/j.porgcoat.2014.12.017
Dalagan JQ, Enriquez EP (2014) One-step synthesis of mesoporous silica-graphene composites by simultaneous hydrothermal coupling and reduction of graphene oxide. Bull Mater Sci 37:589–595. https://doi.org/10.1007/s12034-014-0661-6
Kudin KN, Ozbas B, Schniepp HC, Prudhomme RK, Aksay IA, Car R (2008) Raman spectra of graphite oxide and functionalized graphene sheets. Nano Lett 8:36–41. https://doi.org/10.1021/nl071822y
Altalhi AA, Mohamed EA, Morsy SM, Abou Kana MTH, Negm NA (2021) Catalytic manufacture and characteristic valuation of biodiesel-biojet achieved from Jatropha curcas and waste cooking oils over chemically modified montmorillonite clay. J Mol Liq 340:117175. https://doi.org/10.1016/j.molliq.2021.117175
Altalhi AA, Morsy SM, Abou Kana MTH, Negm NA, Mohamed EA (2021) Pyrolytic conversion of waste edible oil into biofuel using sulphonated modified alumina. Alexandria Eng J 61:4847–4861. https://doi.org/10.1016/j.aej.2021.10.038
Negm NA, Sayed GH, Yehia FZ, Dimitry OIH, Rabie AM, Azmy EAM (2016) Production of biodiesel production from castor oil using modified montmorillonite clay. Egypt J Chem 59:1045–1060
Negm NA, Sayed GH, Habib OI, Yehia FZ, Mohamed EA (2017) Heterogeneous catalytic transformation of vegetable oils into biodiesel in one-step reaction using super acidic sulfonated modified mica catalyst. J Mol Liq 237:38–45. https://doi.org/10.1016/j.molliq.2017.04.076
Negm NA, Sayed GH, Yehia FZ, Habib OI, Mohamed EA (2017) Biodiesel production from one-step heterogeneous catalyzed process of Castor oil and Jatropha oil using novel sulphonated phenyl silane montmorillonite catalyst. J Mol Liq 234:157–163. https://doi.org/10.1016/j.molliq.2017.03.043
Farag AA, Mohamed EA, Sayed GH, Anwer KE (2021) Experimental/computational assessments of API steel in 6 M H2SO4 medium containing novel pyridine derivatives as corrosion inhibitors. J Mol Liq 330:115705. https://doi.org/10.1016/j.molliq.2021.115705
Lebron YAR, Moreira VR, Drumond GP, da Silva MM, de Bernardes RO, de Santos LVS, Jacob RS, Viana MM, de Vasconcelos CKB (2020) Graphene oxide for efficient treatment of real contaminated water by mining tailings: metal adsorption studies to Paraopeba river and risk assessment. Chem Eng J Adv 2:100017. https://doi.org/10.1016/j.ceja.2020.100017
Tang J, Wang T, Sun X, Hu Y, Xie Q, Guo Y, Xue H, He J (2013) Novel synthesis of reduced graphene oxide-ordered mesoporous carbon composites and their application in electrocatalysis. Electrochim Acta 90:53–62. https://doi.org/10.1016/j.electacta.2012.11.099
Wang F, Zheng M, Zhu C, Zhang B, Chen W, Ma L, Shen W (2015) Visible light photocatalytic H2-production activity of wide band gap ZnS nanoparticles based on the photosensitization of graphene. Nanotechnology 26:345402. https://doi.org/10.1088/0957-4484/26/34/345402
Viet Quang D, Rayer A, Hadri N, Abu Zahra M (2015) Preparation of polyethylenimine impregnated mesoporous precipitated silica for CO2 capture, https://doi.org/10.1016/B978-0-444-63461-0.50003-1.
Samaraweera H, Pittman CU, Thirumalai RV, Hassan EB, Perez F, Mlsna T (2021) Characterization of graphene/pine wood biochar hybrids: potential to remove aqueous Cu2+. Environ Res 192:110283. https://doi.org/10.1016/j.envres.2020.110283
Li J, Zhang S, Chen C, Zhao G, Yang X, Li J, Wang X (2012) Removal of Cu(II) and fulvic acid by graphene oxide nanosheets decorated with Fe3O4 nanoparticles. ACS Appl Mater Interfaces 4:4991–5000. https://doi.org/10.1021/am301358b
Zhao L, Yang S, Yilihamu A, Ma Q, Shi M, Ouyang B, Zhang Q, Guan X, Yang S-T (2019) Adsorptive decontamination of Cu2+-contaminated water and soil by carboxylated graphene oxide/chitosan/cellulose composite beads. Environ Res 179:108779. https://doi.org/10.1016/j.envres.2019.108779
Huang Q, Chen Y, Yu H, Yan L, Zhang J, Wang B, Du B, Xing L (2018) Magnetic graphene oxide/MgAl-layered double hydroxide nanocomposite: one-pot solvothermal synthesis, adsorption performance and mechanisms for Pb2+, Cd2+, and Cu2+. Chem. Eng. J. 341:1–9. https://doi.org/10.1016/j.cej.2018.01.156
Yan H, Li H, Tao X, Li K, Yang H, Li A, Xiao S, Cheng R (2014) Rapid removal and separation of iron(II) and manganese(II) from micropolluted water using magnetic graphene oxide. ACS Appl Mater Interfaces 6:9871–9880. https://doi.org/10.1021/am502377n
Ali IO, El-Sheikh SM, Salama TM, Abdel-Khalek EK, Thabet MS, Bakr MF, Fodial MH (2021) Novel composites of multifunctional NaP zeolite/graphene oxide for highly efficient removal of Fe(III) from aqueous solution. J Inorg Organomet Polym Mater 31:577–590. https://doi.org/10.1007/s10904-020-01748-7
Hashem HE, Mohamed EA, Farag AA, Negm NA, Azmy EAM (2021) New heterocyclic Schiff base-metal complex: synthesis, characterization, density functional theory study, and antimicrobial evaluation. Appl Organomet Chem 35:e6322. https://doi.org/10.1002/aoc.6322
Farag AA, Abdallah HE, Badr EA, Mohamed EA, Ali AI, El-Etre AY (2021) The inhibition performance of morpholinium derivatives on corrosion behavior of carbon steel in the acidized formation water: theoretical, experimental and biocidal evaluations. J. Mol. Liq. 341:117348. https://doi.org/10.1016/j.molliq.2021.117348
Anwer KE, Farag AA, Mohamed EA, Azmy EM, Sayed GH (2021) Corrosion inhibition performance and computational studies of pyridine and pyran derivatives for API X-65 steel in 6M H2SO4. J Ind Eng Chem. https://doi.org/10.1016/j.jiec.2021.03.016
Farag AA, Eid AM, Shaban MM, Mohamed EA, Raju G (2021) Integrated modeling, surface, electrochemical, and biocidal investigations of novel benzothiazoles as corrosion inhibitors for shale formation well stimulation. J Mol Liq 336:116315. https://doi.org/10.1016/j.molliq.2021.116315
Hashem HE, Farag AA, Mohamed EA, Azmy EM (2022) Experimental and theoretical assessment of benzopyran compounds as inhibitors to steel corrosion in aggressive acid solution. J Mol Struct 1249:131641. https://doi.org/10.1016/j.molstruc.2021.131641
Mohamed EA, Hashem HE, Azmy EM, Negm NA, Farag AA (2021) Synthesis, structural analysis, and inhibition approach of novel eco-friendly chalcone derivatives on API X65 steel corrosion in acidic media assessment with DFT & MD studies. Environ Technol Innov 24:101966. https://doi.org/10.1016/j.eti.2021.101966
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abdel Wahab, M.M., Sayed, G.H., Ramadan, R.M. et al. Synergistic effects of graphene oxide grafted with barbituric acid nanocomposite for removal of heavy metals from aqueous solution. Nanotechnol. Environ. Eng. 8, 347–359 (2023). https://doi.org/10.1007/s41204-022-00274-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41204-022-00274-w