Abstract
Aims and Objectives
To identify risk factors associated with delay in adjuvant chemotherapy in ovarian cancer patients.
Materials and Methods
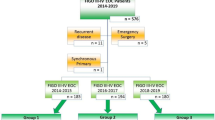
This is single institute retrospective study. All patients of highgrade epithelial ovarian, fallopian tube, and primary peritoneal carcinoma, FIGO Stage II–IV, who underwent PDS at our institution from January 2018 to December 2019 were included. Patients were excluded if they had non-epithelial ovarian histology, borderline tumors, or if they were declared unresectable or R-2 (> 2 cm residual tumor) resection at the end of surgery, synchronous tumors or if they received chemotherapy from outside our institution. The demographic data, biochemical parameters and surgical complexity score was recorded and analyzed statistically.
Results
We identified a total of 102 patients who met the inclusion criteria with a mean age of 60.1 years, SD 11.4. Of these, 22 patients (21.6%) experienced a delay in chemotherapy and 80 patients (78.4%) started chemotherapy without any delay. Our study highlighted that age > 65 years, low albumin preoperatively < 3.5 gm/dl, higher Clavin-Dindo scores (3–5) in the postoperative period was significantly associated with delay in chemotherapy after multivariate analysis.
Conclusion
This study enables the oncologist to understand factors that delay chemotherapy in advanced ovarian cancers post-surgery and would help in customizing the treatment for patients for better outcomes, target the delay group for high compliance and work towards improving the timeliness of chemotherapy initiation, enhancing patient outcomes to optimize the overall management of advanced ovarian cancer.
Similar content being viewed by others
References
Chandramohan K, Thomas B. Cancer trends and burden in India. Lancet Oncol. 2018;19(12):e663.
Berek JS, Kehoe ST, Kumar L, Friedlander M. Cancer of the ovary, fallopian tube, and peritoneum. Int J Gynecol Obstet. 2018;143:59–78.
Bristow RE, Tomacruz RS, Armstrong DK, Trimble EL, Montz FJ. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J Clin Oncol. 2002;20(5):1248–59.
Sioulas VD, Schiavone MB, Kadouri D, Zivanovic O, Roche KL, O’Cearbhaill R, et al. Optimal primary management of bulky stage IIIC ovarian, fallopian tube and peritoneal carcinoma: are the only options complete gross resection at primary debulking surgery or neoadjuvant chemotherapy? Gynecol Oncol. 2017;145(1):15–20.
Sinno AK, Li X, Thompson RE, Tanner EJ, Levinson KL, Stone RL, et al. Trends and factors associated with radical cytoreductive surgery in the United States: a case for centralized care. Gynecol Oncol. 2017;145(3):493–9.
Schorge JO, Bregar AJ, Durfee J, Berkowitz RS. Meigs to modern times: the evolution of debulking surgery in advanced ovarian cancer. Gynecol Oncol. 2018;149(3):447–54.
Fotopoulou C, Jones BP, Savvatis K, Campbell J, Kyrgiou M, Farthing A, et al. Maximal effort cytoreductive surgery for disseminated ovarian cancer in a UK setting: challenges and possibilities. Arch Gynecol Obstet. 2016;294(3):607–14.
Feng Z, Wen H, Bi R, Yang W, Wu X. Prognostic impact of the time interval from primary surgery to intravenous chemotherapy in high grade serous ovarian cancer. Gynecol Oncol. 2016;141(3):466–70.
Alvarez RD. We need a new paradigm in gynecologic cancer care: SGO proposes solutions for delivery, quality and reimbursement policies. Gynecol Oncol. 2013;129(1):3–4.
Bonte AS, Luyckx A, Wyckmans L, Trinh XB, Van Dam PA. Quality indicators for the management of endometrial, cervical and ovarian cancer. Eur J Surg Oncol. 2019;45(4):528–37.
Liu Y, Zhang T, Wu Q, Jiao Y, Gong T, Ma X, et al. Relationship between initiation time of adjuvant chemotherapy and survival in ovarian cancer patients: a dose-response meta-analysis of cohort studies. Sci Rep. 2017;7(1):9461.
Aletti GD, Dowdy SC, Podratz KC, Cliby WA. Relationship among surgical complexity, short-term morbidity, and overall survival in primary surgery for advanced ovarian cancer. Am J Obstet Gynecol. 2007;197(6):676.e1-676.e7.
Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–51.
Sugarbaker PH. Successful management of microscopic residual disease in large bowel cancer. Cancer Chemother Pharmacol. 1999;43(7):S15-25.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–13.
Mahner S, Eulenburg C, Staehle A, Wegscheider K, Reuss A, Pujade-Lauraine E, et al. Prognostic impact of the time interval between surgery and chemotherapy in advanced ovarian cancer: analysis of prospective randomised phase III trials. Eur J Cancer. 2013;49(1):142–9.
Alexander M, Blum R, Burbury K, Coutsouvelis J, Dooley M, Fazil O, et al. Timely initiation of chemotherapy: a systematic literature review of six priority cancers—results and recommendations for clinical practice: time-to-chemotherapy and survival. Intern Med J. 2017;47(1):16–34.
Bun S, Yunokawa M, Ebata T, Kobayashi Kato M, Shimoi T, Kato T, et al. Feasibility of initial treatment in elderly patients with ovarian cancer in Japan: a retrospective study. Int J Clin Oncol. 2019;24(9):1111–8.
Seagle BLL, Butler SK, Strohl AE, Nieves-Neira W, Shahabi S. Chemotherapy delay after primary debulking surgery for ovarian cancer. Gynecol Oncol. 2017;144(2):260–5.
Singh S, Guetzko M, Resnick K. Preoperative predictors of delay in initiation of adjuvant chemotherapy in patients undergoing primary debulking surgery for ovarian cancer. Gynecol Oncol. 2016;143(2):241–5.
Baldewpersad Tewarie NMS, Van Driel WJ, Van Ham M, Wouters MW, Kruitwagen R, Kruse AJ, et al. Postoperative outcomes of primary and interval cytoreductive surgery for advanced ovarian cancer registered in the Dutch Gynecological Oncology Audit (DGOA). Gynecol Oncol. 2021;162(2):331–8.
Kengsakul M, Nieuwenhuyzen-de Boer GM, Udomkarnjananun S, Kerr SJ, Niehot CD, Van Beekhuizen HJ. Factors predicting postoperative morbidity after cytoreductive surgery for ovarian cancer: a systematic review and meta-analysis. J Gynecol Oncol. 2022;33(4):e53.
Kumar A, Janco JM, Mariani A, Bakkum-Gamez JN, Langstraat CL, Weaver AL, et al. Risk-prediction model of severe postoperative complications after primary debulking surgery for advanced ovarian cancer. Gynecol Oncol. 2016;140(1):15–21.
Wright JD, Ananth CV, Tsui J, Glied SA, Burke WM, Lu YS, et al. Comparative effectiveness of upfront treatment strategies in elderly women with ovarian cancer: Upfront Treatment for Ovarian Cancer. Cancer. 2014;120(8):1246–54.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Batra Modi, K., Batra, S., Sardana, S. et al. An Audit on Factors Delaying Time to Chemotherapy in Stage IIB–IV Ovarian Cancer Patients After Primary Debulking Surgery. Indian J Gynecol Oncolog 22, 79 (2024). https://doi.org/10.1007/s40944-024-00843-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40944-024-00843-8