Abstract
Purpose
Early-stage bulky cancers of the cervix and locally advanced cancer (defined as FIGO 2009 Stages IB2, IIA2 and IIB) form a distinctive subset of “curable” cervical cancers with good 5-year survival rates. The use of neoadjuvant chemotherapy followed by radical hysterectomy has been considered an attractive approach to improve disease control and reduce toxicity associated with radiation. In view of economic implications, availability of radiotherapy centres and radiotherapy induced morbidity, the advantages of downsizing the disease without the use of radiation are tempting and open a completely new philosophy of radical treatment for locally advanced cervical cancer. This study was conducted to establish non-inferiority of neoadjuvant chemotherapy in carcinoma cervix with concurrent chemoradiation in terms of disease-free survival and overall survival and to assess the pathological response and chances of avoiding radiation and its associated morbidity.
Methods
Our study retrospectively reviewed clinical results of patients with cervical cancer staged IB2, IIA2, and IIB according to the 2009 FIGO classification treated with neoadjuvant chemotherapy followed by radical hysterectomy.
Results
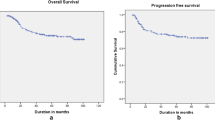
Survival following neoadjuvant chemotherapy and radical surgery was found to be less in comparison with standard survival rates in IB2, IIA2, and IIB (FIGO 2009) cervical cancer.
Conclusion
Therapeutic efficacy of NACT should be viewed with caution since more than one-third of patients were treated with adjuvant brachytherapy. However, in developing countries with higher incidence of cervical cancers wherein brachytherapy resources are scarce, it is still open for debate and discussion.


Similar content being viewed by others
References
Sardi JE, Giaroli A, Sananes C, Ferreira M, Soderini A, Bermudez A, Snaidas L, Vighi S, Gomez Rueda N, di Paola G. Long-term follow-up of the first randomized trial using neoadjuvant chemotherapy in stage Ib squamous carcinoma of the cervix: the final results. Gynecol Oncol. 1997;67(1):61–9. https://doi.org/10.1006/gyno.1997.4812. (PMID: 9345358).
Keys HM, Bundy BN, Stehman FB, et al. Cisplatin, radiation, and adjuvant hysterectomy compared with radiation and adjuvant hysterectomy for bulky stage IB cervical carcinoma. N Engl J Med. 1999;340:1154–61.
Souhami L, Gil RA, Allan SE, et al. A randomized trial of chemotherapy followed by pelvic radiation therapy in stage IIIB carcinoma of the cervix. J Clin Oncol. 1991;9:970–7.
Rotman M, Sedlis A, Piedmonte MR, et al. A phase III randomized trial of postoperative pelvic irradiation in stage IB cervical carcinoma with poor prognostic features: follow-up of a gynecologic oncology group study. Int J Radiat Oncol Biol Phys. 2006;65:169–76.
Linghu H, Xu XR, Mei YY, Tang JY, Tang LD, Sun T. Response of early-stage bulky cervical squamous carcinoma to preoperative adjuvant chemotherapy. Chin Med Sci J. 2004;19(2):116–9.
He L, Wu L, Su G, Wei W, Liang L, Han L, Kebria M, Liu P, Chen C, Yu Y, Zhong M, Wang W. The efficacy of neoadjuvant chemotherapy in different histological types of cervical cancer. Gynecol Oncol. 2014;134(2):419–25. https://doi.org/10.1016/j.ygyno.2014.06.001. (Epub 2014 Jun 8 PMID: 24918866).
Mossa B, Mossa S, Corosu L, Marziani R. Follow-up in a long-term randomized trial with neoadjuvant chemotherapy for squamous cell cervical carcinoma. Eur J Gynaecol Oncol. 2010;31(5):497–503 (PMID: 21061788).
Minig L, Colombo N, Zanagnolo V, et al. Platinum-based neoadjuvant chemotherapy followed by radical surgery for cervical carcinoma international federation of gynecology and obstetrics stage IB2-IIB. Int J Gynecol Cancer. 2013;23:1647–54.
Kitagawa R, Katsumata N, Shibata T, et al. Paclitaxel plus carboplatin versus paclitaxel plus cisplatin in metastatic or recurrent cervical cancer: The open-label randomized phase III trial JCOG0505. J Clin Oncol. 2015;33:2129–35.
Moioli M, Papadia A, Mammoliti S, Pacella E, Menoni S, Menada MV, Ragni N. Chemotherapy with cisplatin and paclitaxel in locally advanced cervical cancer: has this regimen still a role as neoadjuvant setting? Minerva Ginecol. 2012;64(2):95–107 (PMID: 22481620).
Park DC, Kim JH, Lew YO, Kim DH, Namkoong SE. Phase II trial of neoadjuvant paclitaxel and cisplatin in uterine cervical cancer. Gynecol Oncol. 2004;92(1):59–63. https://doi.org/10.1016/j.ygyno.2003.09.015. (PMID: 14751139).
Park DC, Suh MJ, Yeo SG. Neoadjuvant paclitaxel and cisplatin in uterine cervical cancer: long-term results. Int J Gynecol Cancer. 2009;19(5):943–7. https://doi.org/10.1111/IGC.0b013e3181a23c2e. (PMID: 19574789).
Modarress M, Maghami FQ, Golnavaz M, Behtash N, Mousavi A, Khalili GR. Comparative study of chemoradiation and neoadjuvant chemotherapy effects before radical hysterectomy in stage IB–IIB bulky cervical cancer and with tumor diameter greater than 4 cm. Int J Gynecol Cancer. 2005;15(3):483–8.
Gupta S, Maheshwari A, Parab P, Mahantshetty U, Hawaldar R, Sastri S, et al. Neoadjuvant chemotherapy followed by radical surgery versus concomitant chemotherapy and radiotherapy in patients with stage IB2, IIA, or IIB squamous cervical cancer: a randomized controlled trial. J Clin Oncol. 2018;36(16):1548–615.
Zeng J, Sun P, Ping Q, Jiang S, Hu Y. Clinical outcome of FIGO 2018 stage IB3/IIA2 cervical cancer treated by neoadjuvant chemotherapy followed by radical surgery due to lack of radiotherapy equipment A retrospective comparison with concurrent chemoradiotherapy. PLoS ONE. 2022;17(3):e0266001. https://doi.org/10.1371/journal.pone.0266001. (PMID: 35324998; PMCID: PMC8947074).
Buda A, Fossati R, Colombo N, Fei F, Floriani I, Alletti DG, Katsaros D, Landoni F, Lissoni A, Malzoni C, Sartori E, Scollo P, Torri V, Zola P, Mangioni C. Randomized trial of neoadjuvant chemotherapy comparing paclitaxel, ifosfamide, and cisplatin with ifosfamide and cisplatin followed by radical surgery in patients with locally advanced squamous cell cervical carcinoma: the SNAP01 (studio neo-adjuvante portio) Italian collaborative study. J Clin Oncol. 2005;23(18):4137–45.
Chuang L, Kanis MJ, Miller B, Wright J, Small W Jr, Creasman W. Treating locally advanced cervical cancer with concurrent chemoradiation without brachytherapy in low-resource countries. Am J Clin Oncol. 2016;39(1):92–7. https://doi.org/10.1097/COC.0000000000000222. (PMID: 26398064).
Li W, Zhang W, Sun L, Wang L, Cui Z, Zhao H, Wang D, Zhang Y, Guo J, Yang Y, Wang W, Bin X, Lang J, Liu P, Chen C. Neoadjuvant chemotherapy followed by surgery versus abdominal radical hysterectomy alone for oncological outcomes of stage IB3 cervical cancer-a propensity score matching analysis. Front Oncol. 2021;13(11):730753. https://doi.org/10.3389/fonc.2021.730753. (PMID: 34589433 PMCID: PMC8473821).
Çakır C, Kılıç F, Dur R, Yüksel D, Ünsal M, Korkmaz V, Kılıç Ç, KimyonCömert G, Boran N, Türkmen O, Koç S, Turan T. Neoadjuvant chemotherapy for locally advanced stage (IB2-IIA2-IIB) cervical carcinoma: experience of a tertiary center and comprehensive review of the literature. Turk J Obstet Gynecol. 2021;18(3):190–202. https://doi.org/10.4274/tjod.galenos.2021.70493.PMID:34580469;PMCID:PMC8480215.
Chen H, Liang C, Zhang L, Huang S, Wu X. Clinical efficacy of modified preoperative neoadjuvant chemotherapy in the treatment of locally advanced (stage IB2 to IIB) cervical cancer randomized study. Gynecol Oncol. 2008;110(3):308–15.
Kim HS, Sardi JE, Katsumata N, Ryu HS, Nam JH, Chung HH, Park NH, Song YS, Behtash N, Kamura T, Cai HB, Kim JW. Efficacy of neoadjuvant chemotherapy in patients with FIGO stage IB1 to IIA cervical cancer: an international collaborative meta-analysis. Eur J Surg Oncol. 2013;39(2):115–24. https://doi.org/10.1016/j.ejso.2012.09.003. (Epub 2012 Oct 18 PMID: 23084091).
Kenter G, Greggi S, Vergote I, et al. Results from neoadjuvant chemotherapy followed by surgery compared to chemoradiation for stage Ib2-IIb cervical cancer, EORTC 55994. J Clin Oncol. 2019;37(15 suppl):5503–5503.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shanmugam, S., Susikar, S. & Pravenkumar, R.R. Neoadjuvant Chemotherapy Followed by Surgery in Figo Stage IB2, IIA2, and IIB Cervical Cancer—Survival Analysis. Indian J Gynecol Oncolog 21, 56 (2023). https://doi.org/10.1007/s40944-023-00738-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40944-023-00738-0