Abstract
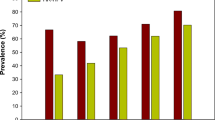
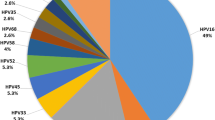
Cervical cancer has emerged as a major threat among the tribal communities of Mizoram where literatures on the prevalence of HPV genotypes are scarce. This study aimed to determine the prevalence of HPV in different cytological grades of cervical samples in relation to lifestyle and socio-demographic factors. Sanger sequencing was performed to confirm the seven high-risk HPV genotypes and logistic regression analysis was performed. The most prevalent genotypes were HPV 16 (45%), HPV 31 (18%), HPV 18 (13%), HPV 33 (11%), HPV 35 (9%), HPV 52 and 58 (2%). Cytological reports showed three Cx-Ca, two HSIL, one LSIL and 29 NILM. Multiple infections of HPV 16, 18, 31 and 33 were present in the Cx-Ca samples, HPV 16, 31 and 33 in HSIL samples, HPV 16, 33 and 35 in the LSIL sample, HPV 16, 18, 31, 35 52 and 58 in 8 NILM and single infection of HPV 16 or 33 in 9 NILM samples. This is the first report of epidemiology and genotype correlation from the Mizo-Mongoloid population showing prevalence of HPV infections in different cytological grades. There is a need to screen HPV status in parallel to PAP smear for better management of timely interventions.


Similar content being viewed by others
Data Availability
The datasets generated and/or analyzed during the current study are not publicly available due to the fact that we do not wish to disclose our dataset since they are part of patients’ medical history. However, they are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660.
Report of National Cancer Registry Programme (ICMR-NCDIR), Bengaluru, India 2020. https://ncdirindia.org/All_Reports/PBCR_Annexures/Default.aspx
Muñoz N, Castellsagué X, Berrington de González A, Gissmann L. Chapter 1: HPV in the etiology of human cancer. Vaccine. 2006; https://doi.org/10.1016/j.vaccine.2006.05.115
Burd EM. Human papillomavirus and cervical cancer. Clin Microbiol Rev. 2003;16(1):1–17. https://doi.org/10.1128/CMR.16.1.1-17.2003.
Ghatak S, Muthukumaran RB, Nachimuthu SK. A simple method of genomic DNA extraction from human samples for PCR-RFLP analysis. J Biomol Technol. 2013;24(4):224–31. https://doi.org/10.7171/jbt.13-2404-001.
Baay MF, Quint WG, Koudstaal J, et al. Comprehensive study of several general and type-specific primer pairs for detection of human papillomavirus DNA by PCR in paraffin-embedded cervical carcinomas. J Clin Microbiol. 1996;34(3):745–7. https://doi.org/10.1128/jcm.34.3.745-747.1996.
Evans MF, Adamson CS, Simmons-Arnold L, et al. Touchdown General Primer (GP5+/GP6+) PCR and optimized sample DNA concentration support the sensitive detection of human papillomavirus. BMC Clin Pathol. 2005;5:10. https://doi.org/10.1186/1472-6890-5-10
Suffys P, Palomino JC, Cardoso Leão S, et al. Evaluation of the polymerase chain reaction for the detection of Mycobacterium tuberculosis. Int J Tuberc Lung Dis. 2000;4(2):179–83.
Van Doorslaer K, Li Z, Xirasagar S, et al. The Papillomavirus Episteme: a major update to the papillomavirus sequence database. Nucleic Acids Res. 2017;45(D1):D499–506. https://doi.org/10.1093/nar/gkw879.
Xia C, Li S, Long T, et al. Current updates on cancer-causing types of human papillomaviruses (HPVs) in East, Southeast, and South Asia. Cancers (Basel). 2021;13(11):2691. https://doi.org/10.3390/cancers13112691.
Muwonge R, Basu P, Gheit T, et al. Acquisition, prevalence and clearance of type-specific human papillomavirus infections in young sexually active Indian women: a community-based multicentric cohort study. PLoS ONE. 2020;15(12): e0244242. https://doi.org/10.1371/journal.pone.0244242.
Mu-Mu-Shwe, Harano T, Okada S, et al. Prevalence of high-risk human papillomavirus (HR-HPV) infection among women with normal and abnormal cervical cytology in Myanmar. Acta Med Okayama. 2014;68(2):79–87. https://doi.org/10.18926/AMO/52404
Bergqvist L, Kalliala I, Aro K, et al. Distribution of HPV genotypes differs depending on behavioural factors among young women. Microorganisms. 2021;9(4):750. https://doi.org/10.3390/microorganisms9040750.
Schmitt M, Depuydt C, Benoy I, et al. Multiple human papillomavirus infections with high viral loads are associated with cervical lesions but do not differentiate grades of cervical abnormalities. J Clin Microbiol. 2013;51(5):1458–64. https://doi.org/10.1128/JCM.00087-13.
Srivastava S, Shahi UP, Dibya A, et al. Distribution of HPV genotypes and involvement of risk factors in cervical lesions and invasive cervical cancer: a study in an Indian population. Int J Mol Cell Med. 2014;3(2):61–73.
Bruni L, Diaz M, Castellsagué X, et al. Cervical human papillomavirus prevalence in 5 continents: meta-analysis of 1 million women with normal cytological findings. J Infect Dis. 2010;202(12):1789–99. https://doi.org/10.1086/657321.
Garrett LR, Perez-Reyes N, Smith PP, et al. Interaction of HPV-18 and nitrosomethylurea in the induction of squamous cell carcinoma. Carcinogenesis. 1993;14(2):329–32. https://doi.org/10.1093/carcin/14.2.329.
Vaccarella S, Herrero R, Snijders PJ, et al. Smoking and human papillomavirus infection: pooled analysis of the international agency for research on cancer HPV prevalence surveys. Int J Epidemiol. 2008;37(3):536–46. https://doi.org/10.1093/ije/dyn033.
Syrjänen K, Shabalova I, Petrovichev N, et al. Smoking is an independent risk factor for oncogenic human papillomavirus (HPV) infections but not for high-grade CIN. Eur J Epidemiol. 2007;22(10):723–35. https://doi.org/10.1007/s10654-007-9180-8.
Minkoff H, Feldman JG, Strickler HD, et al. Relationship between smoking and human papillomavirus infections in HIV-infected and -uninfected women. J Infect Dis. 2004;189(10):1821–8. https://doi.org/10.1086/383479.
Bosch FX, de Sanjosé S. The epidemiology of human papillomavirus infection and cervical cancer. Dis Markers. 2007;23(4):213–27. https://doi.org/10.1155/2007/914823.
Kalra R, Singh SP, Savage SM, et al. Effects of cigarette smoke on immune response: chronic exposure to cigarette smoke impairs antigen-mediated signaling in T cells and depletes IP3-sensitive Ca(2+) stores. J Pharmacol Exp Ther. 2000;293(1):166–71.
Zeidel A, Beilin B, Yardeni I, et al. Immune response in asymptomatic smokers. Acta Anaesthesiol Scand. 2002;46(8):959–64. https://doi.org/10.1034/j.1399-6576.2002.460806.x.
Harris TG, Kulasingam SL, Kiviat NB, et al. Cigarette smoking, oncogenic human papillomavirus, Ki-67 antigen, and cervical intraepithelial neoplasia. Am J Epidemiol. 2004;159(9):834–42. https://doi.org/10.1093/aje/kwh115.
Nagelhout G, Ebisch RM, Van Der Hel O, et al. Is smoking an independent risk factor for developing cervical intra-epithelial neoplasia and cervical cancer? A systematic review and meta-analysis. Expert Rev Anticancer Ther. 2021;21(7):781–94. https://doi.org/10.1080/14737140.2021.1888719.
Global Adult Tobacco Survey Fact Sheet Mizoram 2016 – 17. https://tmc.gov.in/images/act/Mizoram-GATS2-India-StateFactsheet-Countryspecific-latest-v13.pdf.
Kapoor CS, Sharma M. Prevalence of HPV infection in reproductive aged female in Delhi NCR region. Clin Epidemiol Glob Health. 2020;8(2):312–5. https://doi.org/10.1016/j.cegh.2019.12.012.
Sauvaget C, Nene BM, Jayant K, et al. Prevalence and determinants of high-risk human papillomavirus infection in middle-aged Indian women. Sex Transm Dis. 2011;38(10):902–6. https://doi.org/10.1097/OLQ.0b013e318223be5f.
Louie KS, de Sanjose S, Diaz M, et al. Early age at first sexual intercourse and early pregnancy are risk factors for cervical cancer in developing countries. Br J Cancer. 2009;100(7):1191–7. https://doi.org/10.1038/sj.bjc.6604974.
Plummer M, Peto J, Franceschi S; International Collaboration of Epidemiological Studies of Cervical Cancer. Time since first sexual intercourse and the risk of cervical cancer. Int J Cancer. 2012;130(11): 2638–2644. https://doi.org/10.1002/ijc.26250
Basonidis A, Liberis A, Daniilidis A, et al. Human papilloma virus infection and miscarriage: is there an association? Taiwan J Obstet Gynecol. 2020;59(5):656–9. https://doi.org/10.1016/j.tjog.2020.07.005.
Manhart LE, Koutsky LA. Do condoms prevent genital HPV infection, external genital warts, or cervical neoplasia? A meta-analysis Sex Transm Dis. 2002;29(11):725–35. https://doi.org/10.1097/00007435-200211000-00018.
Sabatini ME, Chiocca S. Human papillomavirus as a driver of head and neck cancers. Br J Cancer. 2020;122(3):306–14. https://doi.org/10.1038/s41416-019-0602-7.
Chan CK, Aimagambetova G, Ukybassova T, et al. Human Papillomavirus Infection and Cervical Cancer: Epidemiology, Screening, and Vaccination-Review of Current Perspectives. J Oncol. 2019;2019:3257939. https://doi.org/10.1155/2019/3257939.
Hu Z, Ma D. The precision prevention and therapy of HPV-related cervical cancer: new concepts and clinical implications. Cancer Med. 2018;7(10):5217–36. https://doi.org/10.1002/cam4.1501.
Acknowledgements
The authors wish to thank the Department of Biotechnology (DBT), New Delhi, Govt. of India, for the infrastructural support through Advanced State Biotech Hub. We are thankful to Dr. Lalthakimi Darngawn (OBS and Gynecologist), Synod Hospital Aizawl, Mizoram, India, as well as to Dr. Lalhmingliana (OBS and Gynecologist), Ebenezer Hospital, Aizawl, for their help in sample collection and clinical interpretation.
Funding
The authors declare that no separate funding has been received to carry out this work.
Author information
Authors and Affiliations
Contributions
CVS, LN, LP, KL, EZ and NSK contributed to formulating the questionnaire and study; LN, CVS, LP and KL contributed to the collection of samples and data; CVS, SG and ZZ performed the experiments; CVS, SG and ZS analyzed the data; CVS, SG, ZZ, ZS and NSK contributed to inferring the data and writing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethics Statement
Ethical approval was obtained from Institutional Review Board (IRB), Civil Hospital, Aizawl (B.12018/1/13-CH(A)/IEC/41) and Mizoram University (MZU/IHEC/2015/007), India. All the experiments were performed in accordance with relevant guidelines and regulations. Attested informed consent for study participation was obtained for this study. All authors read and approved the final manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sailo, C.V., Zami, Z., Ghatak, S. et al. Prevalence of High-Risk HPV Types in Women with Negative Cervical Cytology in a State of Northeast India with a High Burden of Cervical Cancer. Indian J Gynecol Oncolog 20, 8 (2022). https://doi.org/10.1007/s40944-022-00610-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40944-022-00610-7