Abstract
Purpose
As per GLOBACON 2012, the incidence of cervical cancer is 22.9% with a mortality rate of 54.9% (http://globocan.iarc.fr, 2010). As the disease affects relatively younger population, high fatality rate impacts family and society in a disastrous way. Cervical carcinoma has a relatively poor prognosis due to advanced stage at presentation. We retrospectively analysed the treated cases of carcinoma cervix to know the impact of various prognostic factors on survival and patterns of failure in our hospital.
Methods
All cases treated from 2010 to 2015 (total 920 pts) who completed curative treatment in a tertiary care hospital were analysed to evaluate overall survival, disease-free survival, factors impacting survival, patterns of failure, factors affecting failure, mortality, and treatment-related toxicity. OS and DFS were calculated with Kaplan–Meier method, and log-rank test was used to identify the difference between groups. Failure factors were correlated with logistic regression, and toxicity was expressed in percentage.
Results
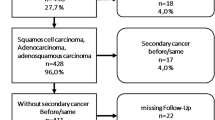
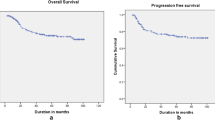
Univariate and multivariate analyses were performed. Pre treatment staging, lymphadenopathy, imaging used for diagnosis, hemoglobin levels extent of vagina involved and during treatment usage of concurrent chemotherapy, dose of radiation used, development of neutropenia and aftercompletion of treatment type of response had shown significant impact on DFS and OS. The presence of lymphadenopathy, increasing involvement of vagina, low pretreatment haemoglobin levels, and not receiving chemotherapy had increased risk of failure, while pretreatment evaluation with CT or MRI decreased the risk of failure. Para-aortic nodes (28%) were the commonest among distant failures. Acute bowel toxicity and radiation proctitis were commonly seen.
Conclusions
Presentation in locally advanced stage decreases survival; hence, detection in early stages by population-based screening programmes is needed. Pretreatment imaging with MRI/CT improves staging and thereby improves survival. There is no impact of 2D versus 3D CRT planning; hence, in high-case-load centres, 2D planning may be used. Follow-up at regular intervals with imaging may aid in earlier identification of failures with better salvage and improved survival.


Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. GLOBOCAN 2008 v1.2, cancer incidence and mortality worldwide: IARC CancerBase No. 10 [Internet]. International Agency for Research on Cancer, Lyon. 2010. http://globocan.iarc.fr. Accessed on 26–27 Mar 2017.
Indian Council of Medical Research. Consensus document for the management of cervical carcinoma. Division of Publication and Information on behalf of the Secretary DHR & DG, ICMR, New Delhi; 2016.
Sakurai H, Mitsuhashi N, Takahashi M. Analysis of recurrence of squamous cell carcinoma of the uterine cervix after definitive radiation therapy alone: patterns of recurrence, latent period, and prognosis. Int J Radiat Oncol Biol Phys. 2001;50:1136–44.
Lim K, Small W Jr, Portelance L, Creutzberg C, Jurgenliemk-Schulz I, Mundt A. Consensus guidelines for delineation of clinical target volume for Intensity-modulated pelvic radiotherapy for the definitive treatment of cervix cancer. Int J Radiat Oncol Biol Phys. 2011;79:348–55.
American Brachytherapy Society (ABS). Brachytherapy Guidelines-Microsoft (®) Excel spread sheet “LQ spread sheet_v2.1 [Internet]. Americanbrachytherapy.org. 2016 [cited 27 March 2017]. https://www.americanbrachytherapy.org/guidelines.
National Cancer Institute Cancer Therapy Evaluation Program (CTEP). Common Toxicity Criteria for Adverse Events v4.03 (CTCAE). 2009. [cited 27 March 2017]. https://ctep.cancer.gov.
Petereit DG, Sarkaria JN, Chappell R, Fowler JF, Hartmann TJ, et al. The adverse effect of treatment prolongation in cervical carcinoma. Int J Radiat Oncol Biol Phys 1995;32(5):1301–7.
Meanwell CA, Kelly KA, Wilson S, Roginski C, Woodman C, Griffiths R, et al. Young age as a prognostic factor in carcinoma cervix: analysis of population based data from 10,022 cases. Br Med J. 1988;296:386–91.
Khatun S, Ferdous J. Menopause and gynecological malignancy. J South Asian Fed Menopause Soc. 2013;1(2):75–9.
Bourgioti C, Chatoupis K, Moulopoulos LA. Current imaging strategies for the evaluation of uterine carcinoma cervix. World J Radiol. 2016;8(4):342–54.
Fyles AW, Pintilie M, Kirkbride P, Levin W, Manchul LA, Rawlings GA. Prognostic factors in patients with cervix cancer treated by radiation therapy: results of a multiple regression analysis. Radiother Oncol. 1995;35(2):107–17.
Coia L, Won M, Lanciano R, Marcial VA, Martz K, Hank G. The patterns of care outcome study for cancer of the uterine cervix results of the second national practice survey. Cancer. 1990;66:2452–6.
Sinistrero G, Sismondi P, Zola P. Results of treatment of uterine cervix cancer by radiotherapy. Radiother Oncol. 1988;13:257–65.
Endo D, Todo Y, Okamoto K, Minobe S, Kato H, Nishiyama N. Prognostic factors for patients with carcinoma cervix treated with concurrent chemoradiotherapy: a retrospective analysis in a Japanese cohort. J Gynecol Oncol. 2015;26(1):12–8.
Viswanathan AN, Thomadsen B. American Brachytherapy Society consensus guidelines for locally advanced carcinoma of the cervix. Part I: general principles. Brachytherapy. 2012;11(1):33–46.
Perez CA, Breaux S, Madoc-Jones H. Radiation therapy alone in the treatment of carcinoma of the uterine cervix: I. Analysis of tumor recurrence. Cancer. 1983;51:1293–402.
Petereit DG, Pearcey R. Literature analysis of high dose rate brachytherapy fractionation schedules in the treatment of carcinoma cervix: Is there an optimal fractionation schedule? Int J Radiat Oncol Biol Phys. 1999;43:359–66.
NCI Clinical Announcement. Concurrent Chemoirradiation for carcinoma cervix. NCI Press Officer: Chemotherapy plus Radiation Improves Survival. http://www.nih.gov/news/pr/feb99/nci-22.htm. Accessed on 20 June 2008.
Green JA. Survival and recurrence after concomitant chemotherapy and radiotherapy for cancer of the uterine cervix; a systematic review and meta-analysis. Lancet. 2001;358:781–6.
Obermair A, Cheuk R, Horwood K, Neudorfer M, Janda M, Giannis G, et al. Anemia before and during concurrent chemoradiotherapy in patients with carcinoma cervix: effect on progression-free survival. Int J Gynecol Cancer. 2003;13:633–9.
Gulia A, Patel F, Rai B, Sharma SC. Conventional four field radiotherapy versus computed tomography-based treatment planning in cervical carcinoma: a dosimetric study. South Asian J Cancer. 2013;2(3):132–5.
Pendlebury SC, Cahill S, Crandon AJ, Bull CA. Role of bipedal lymphangiogram in radiation treatment planning for cervix cancer. Int J Radiat Oncol Biol Phys. 1993;27:959–62.
Fenkell L, Assenholt M, Nielsen SK, Haie-Meder C, Pötter R, Lindegaard J, Tanderup K. Parametrial boost using midline shielding results in an unpredictable dose to tumor and organs at risk in combined external beam radiotherapy and brachytherapy for locally advanced carcinoma cervix. Int J Radiat Oncol Biol Phys. 2011;79(5):1572–9.
Tewari KS, Eskander RN. Impact of chemotherapy-induced neutropenia on survival in patients with breast, ovarian and carcinoma cervix: a systematic review. J Hematol Malig. 2012;2(3):38–44.
Lapuz C, Kondalsamy-Chennakesavan S, Bernshaw D, Khaw P, Narayan K. Stage IB cervix cancer with nodal involvement treated with primary surgery or primary radiotherapy: patterns of failure and outcomes in a contemporary population. J Med Imaging Radiat Oncol. 2016;60:274–82.
Tiltman AJ. The pathology of cervical tumours. Best Pract Res Clin Obstet Gynaecol. 2005;19(4):485–500.
Schmid MP, Franckena M, Kirchheiner K, Sturdza A, Georg P, Dörr W, Pötter R. Distant metastasis in patients with carcinoma cervix after primary radiotherapy with or without chemotherapy and image guided adaptive brachytherapy. Gynecol Oncol. 2014;133(2):256–62.
Fagundes H, Perez CA, Grigsby PW, Lockett MA. Distant metastases after irradiation alone in carcinoma of the uterine cervix. Int J Radiat Oncol Biol Phys. 1992;24(2):197–204.
Endo D, Todo Y, Okamoto K, Minobe S, Kato H, Nishiyama N. Prognostic factors for patients with carcinoma cervix treated with concurrent chemoradiotherapy: a retrospective analysis in a Japanese cohort. J Gynecol Oncol. 2015;26(1):12–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
Retrospective study, adding ethical certificate as separate file.
Informed Consent
Informed consent was obtained from all individual participants included in the study before treatment in our hospital, and no individual identity was disclosed.
Rights and permissions
About this article
Cite this article
Prathipati, A., Jilla, S., Subramanian, B.V. et al. Impact of Various Prognostic Factors on Overall Survival, Disease-Free Survival and Patterns of Failure in Carcinoma Cervix: A Tertiary Care Centre Experience from South India. Indian J Gynecol Oncolog 16, 11 (2018). https://doi.org/10.1007/s40944-018-0179-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40944-018-0179-8