Abstract
The present text discussed about the recent developments and applications of the Ullmann reaction in respect of the C–C bond formation. The major drawback of the Ullmann reaction, i.e., harsh reaction condition has recently been overcome with the development of various synthetic methodologies. Green synthetic methodologies such as metal-, ligand-, and additive free conditions, recyclable heterogenous catalysts, microwave- and ultrasound-assisted synthesis, etc. have got special importance in this field. This text lucidly describes all these recently developed synthetic methodologies which make the century old Ullmann reaction much efficient, effective, economical and attractive in the field of synthetic organic chemistry. On-surface Ullmann coupling for the C–C bond formation, i.e., invoking C–C coupling reactions on a reactive surface has been discussed. All such developments of the Ullmann C–C coupling facilitate its application in the synthesis of bio-active heterocycles, drug-like molecules and natural products which has also been discussed in this text.
Similar content being viewed by others
Introduction
In 1901, Ullmann reported a useful technique for the formation of a new C–C bond between two aryls by the condensation of two molecules of aromatic halide in the presence of finely divided copper which is known as Ullmann reaction (Scheme 1) [1]. This is the first transition metal mediated coupling reaction for the formation of aryl–aryl bond. The reaction is most often been done on aryl iodides, but bromides and chlorides have also been used. Aryl bromides and chlorides do not usually react unless there is an electron withdrawing group like nitro, ester, etc. at ortho and/or para to the halogen atom. Hence the order of reactivity is ArI > ArBr > ArCl. For the unsymmetrical coupling (two different aryl iodides), the best yields are observed when one aryl halide is very reactive and the other is relatively unreactive. Reactive aryl halides mean have electron withdrawing group at ortho to the halogen. Several excellent reviews have already been published on the historical development of this classical transformation [2–8].
Although, during the first 70 years of the 20th century, Ullmann reaction was one of the main reactions for the formation of C–C bond (aryl–aryl bond), but due to some limitations like harsh reaction conditions (generally >200 °C), high copper catalyst loading, poor functional group tolerance and generally the low yield of the products had limited the applications of Ullmann reaction that time. But with the increasing understanding of the mechanism of the Ullmann reaction and development of novel ligands, modifications of catalytic system and application of the green technologies allowed the Ullmann reaction to be conducted under mild conditions with desirable yields with excellent functional group tolerance. This facilitates the wide applications of the Ullmann reaction to the synthesis of heterocycles, drug-like molecules, natural products, chiral auxiliaries, etc. So a text covering recently reported advanced catalytic systems and other green technologies and their applications to the synthesis of compounds of interest is really sought.
Mechanism of Ullmann coupling reaction
The mechanism for the Ullmann reaction is not known with certainty but there are two proposed popular mechanisms [1, 9]:
-
1.
Free radical mechanism.
-
2.
Ionic mechanism.
Free radical mechanism
According to free radical mechanism, the copper generates an aryl radical (2.2) by a single electron transfer (SET) from copper metal to the alkyl halide (2.1). Two aryl radicals then react to form the final biaryl products (2.3) (Scheme 2).
Ionic mechanism
According to the ionic mechanism, an aryl copper intermediate is formed. This mechanism begins with the oxidative addition of the copper to the aryl halide (2.1) followed by a single electron transfer to form an organocuprate reagent (3.2). This organocuprate (3.2) performs another oxidative addition on an aryl halide forms biaryl copper halide complex (3.3) which after reductive elimination results in the final biaryl product (2.3) (Scheme 3).
Recent developments of synthetic methodologies of Ullmann-type reaction
Generally the harsh reaction conditions (>200 °C), high copper catalyst loading and poor functional group tolerance had limited the applications of Ullmann-type coupling reactions since they have been discovered. But with the developments of sustainable, environmentally benign methodologies, reusable catalyst, etc. make the reaction possible to be performed in mild conditions and thereby widely accepted and applied in organic synthesis, medicinal chemistry and natural product synthesis. For clear presentation, the recently developed synthetic methodologies have been classified into two classes—new or advanced catalytic systems and green technologies. All the recent developments of the Ullmann-type coupling (focusing mainly on the publications during the last 10 years, i.e., from 2006 to 2015) have been discussed in respect of C–C bond formation.
New or advanced catalytic systems
In 2011, Karimi et al. developed that Au nanoparticles supported on a bifunctional periodic mesoporous organosilica (Au@PMO) is an effective and reusable heterogeneous catalyst for Ullmann coupling reaction of aryl iodides in good to excellent yields (Scheme 4) [10]. Most interestingly, when iodobenzene (1.1) was treated with phenol (4.3), the C–C homocoupling product (1.2) was obtained in 52 % yield instead of C–O coupling product (4.4).
In a subsequent report, Maheswaran and co-workers described that the gold nanoparticles stabilized on a nanocrystalline magnesium oxide support, NAP-Mg–Au(0), is an efficient, reusable heterogeneous catalyst for the Ullmann homocoupling of aryl iodides (Scheme 5) [11].
Dabiri, Bazgir and co-workers also reported a Au nanoparticle grapheme oxide nanocomposite (Au NPs-RGO) as an effective and reusable heterogeneous catalyst for the Ullmann homocoupling of aryl iodides. The extra advantage of this catalyst over the other two Au-catalysts discussed above was that aryl bromides bearing electron-withdrawing groups were also coupled in the presence of this catalyst (Scheme 6) [12].
Rothenberg et al. presented a room temperature catalytic alternative to the Ullmann reaction based on electroreductive homocoupling of haloarenes in ionic liquids catalyzed by palladium nanoparticles (Scheme 7) [13]. The attractiveness of this system lies in the fact that the nanoparticles are generated in situ in an electrochemical cell and only electrons and water are necessary for closing the catalytic cycle.
In 2009, Nacci et al. developed a simple and efficient method for the Ullmann homocoupling of aryl bromides 8.1a–e to afford the products 8.2a–e under very mild reaction conditions using Pd colloids as the catalyst, an aldehyde as the reducing agent in an ionic liquid (tetrabutylammonium bromide, TBAB) as the reaction medium (Scheme 8) [14]. The role of TBAB is very crucial for this process because it behaves simultaneously as the base, ligand and reaction medium. Moreover, the efficiency of Pd-catalysts has also been investigated in Ullmann coupling of bromopyridine and iodopyridine in presence of co-reductants [15, 16]. Here it is important to note that, Cai et al. developed Pd(OAc)2 catalyzed Ullmann reaction in microemulsion without other reducing agent [17]. In 2010, Cheng et al. also described a graphite-supported palladium nanoparticles-catalyzed reductive Ullmann reaction of aryl chloride in an ionic liquid/supercritical carbon dioxide system [18].
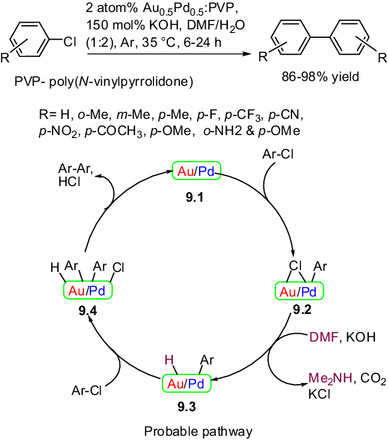
In the above discussions, the gold or palladium nanoparticles were used as the alternative to copper catalyst for Ullmann homocoupling under mild reaction conditions. Recently, Sakurai and co-workers achieved an Ullmann coupling of chloroarene under ambient conditions using bimetallic gold–palladium alloy nanoclusters as an effective catalyst and DMF as both a co-solvent and reducing agent [19]. The reaction does not occur with either gold or palladium single-metal clusters alone, nor with a physical mixture of the two metals. The unusual character of the bimetallic Au/Pd alloy NCs, which display a higher catalytic activity towards chloroarenes than for bromoarenes, is interesting. Possible catalytic pathway for Ullmann coupling of chloroarenes catalyzed by bimetallic Au0.5Pd0.5:PVP is depicted in Scheme 9. An intermediate 9.2 might be formed via oxidative addition of chloroarenes with the bimetallic gold–palladium alloy nanoclusters 9.1. Then DMF donates the hydrogen to form Cl free intermediate 9.3, followed by the oxidative addition of ArCl to form intermediate 9.4. Subsequent reductive elimination of Ar–Ar and HCl would lead to regenerate the catalyst 9.1. In a subsequent report, the same group also described the Ullmann coupling of chloropyridines catalyzed by poly(N-vinylpyrrolidone) (PVP)-stabilized bimetallic gold–palladium alloy nanoclusters under ambient conditions [20].
Moreover, on-surface Ullmann coupling for the C–C bond formation, i.e., invoking C–C coupling reactions on a reactive surface has came into light recently [21–24]. In this method, the halogenated precursor molecules are deposited onto metal surfaces and depending on the type of metal surface, coupling proceeds either directly [25] or via a metastable organic intermediate [26].
Green technologies
In 2006, Li et al. described the Ullmann coupling reaction of iodobenzene in aqueous medium over the Ph-MCM-41 supported Pd-catalyst [27]. The advantage of this catalyst was that with the modification of MCM-41 with Ph-groups could enhance the activity and selectivity of the catalyst owing to the increase of hydrophobicity of the catalyst.
In 2007, the same group develops a new supported Pd catalyst using the mesophorous Ph–Al-MCM-41 as the support, which exhibit high activity and selectivity, as well as strong durability during a water-medium iodobenzene Ullmann reaction [28].
In a subsequent report in 2007, the same group also reported a supported Pd catalyst on the phenyl-functionalized SBA-15 (Pd/Ph-SBA-15) with a high distribution of Pd active sites, ordered mesopores, and strong surface hydrophobicity, leading to enhanced activity and selectivity in a water-medium iodobenzene Ullmann coupling reaction [29].
In 2009, Wang and Wan reported the Ullmann coupling reaction of chlorobenzene in aqueous medium over the ordered mesophorous carbons supported Pd-catalyst (Pd/OMC) [30].
In 2010, Jiang and co-workers developed the Ullmann coupling reaction of aryl chlorides in aqueous medium using porous MOF-supported palladium NPs as the catalyst [31].
In 2011, Cheng et al. described an economical and green pathway for the reductive Ullmann coupling reactions of aryl chlorides using recyclable graphene oxide supported palladium nanoparticle catalyst in ionic liquid-supercritical carbon dioxide system (Scheme 10) [32]. The application of naturally abundant “greenhouse” gas CO2 makes this methodology quite attractive.
In 2012, Kamal et al. demonstrated an efficient heterogeneous catalyst system for the water mediated Ullmann coupling reactions of aryl halides using amphiphilic carbon spheres supported Pd NPs(Pd@CSP) [33]. The effect of surface polarity of the CSPs has a pronounced influence on the catalyst activity.
In 2012, Monopoli and Nacci et al. also described an Ullmann process promoted by support-free gold nanoparticles under green conditions, i.e., in water or ionic liquids as the reaction media and glucose as a renewable reductant [34].
In 2014, Bazgir et al. demonstrated an efficient catalyst system for the water mediated Ullmann coupling reactions of aryl halides using Pd NPs supported on high nitrogen doped graphene (Pd NPs-HNG) [35].
In 2014, Karimi and co-workers reported that Pd NPs supported on ionic liquid derived nano-fibrillated mesoporous carbon (Pd@IFMC) are highly active in catalyzing the Ullmann homocoupling of a range of aryl chlorides, bromides and iodides in aqueous medium [36].
In 2014, Dabiri and co-workers demonstrated that a gold nanoparticle supported on a magnetic/s-graphene nanocomposite (Au/Fe3O4/s-G) is a highly water-dispersible/magnetically separable semi heterogeneous catalyst for Ullmann homocoupling of aryl iodides in water [37].
In 2014, Wang and Zhang et al. reported a new ion exchange resin supported Au alloyed Pd single-atom catalyst for the Ullmann coupling reaction of aryl chlorides in water [38]. The catalyst exhibited exceptional activity and excellent durability for Ullmann reactions of aryl chlorides with broad substrate scope and could be reused eight times with no change in yield.
Moreover, recently icosahedral (Ih) nanocrystals have shown excellent catalytic activity due to their unique exposed surface, bounded by {111} facets and built by twin boundary defects towards different chemical reactions. Very recently, Tan and Tang et al. successfully prepared icosahedral gold–platinum alloy nanocrystals into hollow silica nanospheres (Ih Au–Pt@SiO2) which is highly active and stable catalyst for Ullmann homocoupling of aryl iodides in water and ethanol mixture solvent [39].
Another aspect of green technologies implied for the Ullmann reaction is the microwave/ultrasound-assisted organic synthesis includes faster and easier heating of the reaction, shorter reaction time, and high-throughput chemistry. Sometimes, the reaction yields and selectivity can also be greatly improved under microwave irradiation. Although microwave-assisted Ullmann C–O [40, 41], C–N [42, 43] and C–S [44, 45] bond formation has been explored to some extent but there are very few reports on Ullmann C–C bond formation. Recently, Narasimharao and Mokhtar et al. developed a microwave-assisted efficient protocol for the classic Ullmann homocoupling reaction using Cu–Mg–Al hydrotalcite catalysts without using any base or reducing agent [46]. Among four hydrotalcite catalysts containing different amounts of Cu and Mg cations, maximum activity was found for the catalyst that contained an atomic ratio of Cu/Al = 2 and Mg/Al = 0. In 1991, Adolph and Nelson investigated that ultrasonic irradiation can promote the Ullmann coupling reaction of picryl bromide at or below room temperature [47].
Synthetic applications of Ullmann-type coupling reactions
In the recent years, Ullmann-type coupling reactions have been applied tremendously in the formation of C–C bonds toward synthesis of many important heterocycles, drug-like molecules natural products, etc. [48]. The text covers the synthetic applications of Ullmann-type C–C coupling reactions mainly during the past 20 years, i.e., from 1996 to 2015. For the sake of clear presentation, they have been divided into two parts—synthesis of non-natural products and natural products.
Synthesis of non-natural products
In 2007, Wang and Zhu et al. developed a highly efficient one-pot domino process for the formation of three new C–C bonds induced by the corporation of Ullmann-type coupling and C–H transformation together with cleavage of four C–Cl bonds toward synthesis of bay-region fused di perylene-3,4:9,10-tetracarboxylic acid bisimides (diPBIs) (11.2) from easily available tetrachloro-PBI (11.1) (Scheme 11) [49]. This triply linked fused diPBIs is very promising for use as functional components of photovoltaic devices. Very recently, Xu et al. have illustrated on-surface formation of polyphenyl chains through a hierarchical reaction pathway involving Ullmann coupling and cross-dehydrogenative coupling together [50].
Recently, Lee et al. developed an unprecedented tandem Pd-catalyzed Ullmann-type homocoupling reaction of the Blaise reaction intermediate generated from o-bromo aryl nitriles (12.1) for the one pot synthesis of enamino ester-functionalized biaryls (12.2) in good yields (Scheme 12) [51].
Keay and co-workers designed and synthesized a chiral auxiliary (13.1) which provides only one diastereomer (13.2) in the intramolecular Ullmann coupling toward the synthesis of BIPHEP derivatives (13.3) (Scheme 13) [52].
Cai and co-workers developed a tandem reaction for the synthesis of 1H-[1,2,3]triazolo[4,5-c]quinolin-4(5H)-ones (14.3) by the combination of copper catalyzed azide-alkyne cycloaddition (CuAAc) and intramolecular Ullmann C–C coupling (Scheme 14) [53]. In this reaction, the intramolecular trapping of the C–Cu intermediate produced in CuAAC led to further formation of an aryl C–C bond through Ullmann C–C coupling.
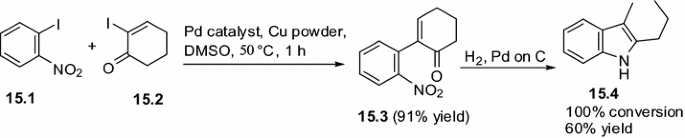
Moreover, Banwell and co-workers developed the synthesis of indoles (15.4) via Pd(0)-mediated Ullmann cross-coupling of o-halonitrobenzene (15.1) and with arrange of α-halo-enones (15.2) followed by reductive cyclization (Scheme 15) [54].
Synthesis of natural products
Hauser et al. reported the first total synthesis of the 7,7′-linked bianthraquinone biphyscion using Ullmann reaction as the key step [55]. The key step was the synthesis of symmetrical biphenyl through traditional Ullmann coupling of a protected iodoresorcinol (Scheme 16).
Meyers and Willemsen developed the first asymmetric synthesis of (S)-(+)-gossypol via a traditional Ullmann coupling [56]. Heating a 40 % solution of bromonaphthyl oxazoline derivative 17.1 in freshly distilled DMF and activated copper at reflux for 1 h gave the binaphthyl derivative 17.2 by Ullmann coupling in 80 % yield as a 17:1 diastereoisomeric mixture. This is the key step for the total synthesis of (S)-(+)-gossypol (Scheme 17).
Meyers et al. also reported the total synthesis of (−)-mastigophorene A (18.3) and (−)-mastigophorene B (18.6) in optically pure form [57] via their developed methodology by oxazoline-mediated asymmetric Ullmann couplings [58–61] of monomeric aryl bromides 18.1 and 18.4, respectively (Scheme 18).
Based on the above methodology developed by Meyers’ group on the asymmetric Ullmann coupling of bromo-oxazoline derivatives, Lin et al. succeed to synthesize both optically pure (+)- and (−)-isokotanin A from (R)-19.1 and (S)-19.1, respectively (Scheme 19) [62].
Banwell and Lupton reported the total synthesis of the alkaloid (±)-aspidospermidine exploiting the Pd(0)-catalyzed Ullmann cross-coupling reaction [63]. This total synthesis involved the pivotal Pd(0)-catalyzed Ullmann cross-coupling of α-iodoenone 20.1 with o-nitrobenzene in DMSO at 70 °C using 5 g atom equiv. of copper powder and Pd2(dba)3 as catalyst (Scheme 20).
Xu and Lin et al. developed an unprecedented nickel-catalyzed asymmetric Ullmann coupling of bis-ortho-substituted aryl halide 21.1 using chiral BINOL-based monodentate phosphoramidite ligand 21.2 to get the enantioenriched compound 21.3 (98 % ee after recrystallization). Starting from compound 21.3, the formal synthesis of (+)-isoschizandrin was accomplished (Scheme 21) [64].
Conclusion
In this text emphasis has been given on two main sections of Ullmann reaction in the C–C bond formation. The first focuses on the recent development of synthetic methodologies related to new or advanced catalytic systems and other green technologies, including ligand-free systems, heterogeneous reusable catalysts, microwave and ultrasound-assisted reactions. Among many exciting developments of the Ullmann coupling reactions, green synthetic methodologies will continue to have a significant impact on this field. The second part emphasizes on the recent applications of Ullmann reactions in the synthesis of heterocycles, drug-like molecules and natural products.
References
Ullmann F, Bielecki J (1901) Ber Dtsch Chem Ges 34:2174–2185
Hassan J, Sévignon M, Gozzi C, Schulz E, Lemaire M (2002) Chem Rev 102:1359–1469
Lin H, Sun D (2013) Org Prep Proced Int 45:341–394
Finet JP, Fedorov AY, Combes S, Boyer G (2002) Curr Org Chem 6:597–626
Zhou F, Cai Q (2015) Beilstein J Org Chem 11:2600–2615
Beletskaya IP, Cheprakov AV (2004) Coord Chem Rev 248:2337–2364
Sambiagio C, Marsden SP, Blacker AJ, McGowan PC (2014) Chem Soc Rev 43:3525–3550
Dhakshinamoorthy A, Asiric AM, Garcia H (2015) Chem Soc Rev 44:1922–1947
Ullmann F (1904) Justus Liebigs Ann Chem 332:38–81
Karimi B, Esfahani FK (2011) Chem Commun 47:10452–10454
Layek K, Maheswaran H, Kantam ML (2013) Catal Sci Technol 3:1147–1150
Movahed SK, Fakharian M, Dabiri M, Bazgir A (2014) RSC Adv 4:5243–5247
Pachón LD, Elsevier CJ, Rothenberg G (2006) Adv Synth Catal 348:1705–1710
Calò V, Nacci A, Monopoli A, Cotugno P (2009) Chem Eur J 15:1272–1279
Nelson TD, Crouch RD (2004) Org React 63:265–570
Lee K, Lee PH (2008) Tetrahedron Lett 49:4302–4305
Jiang JZ, Cai C (2007) Colloid Surf A 305:145–148
Cheng J, Tang L, Xu J (2010) Adv Synth Catal 352:3275–3286
Dhital RN, Kamonsatikul C, Somsook E, Bobuatong K, Ehara M, Karanjit S, Sakurai H (2012) J Am Chem Soc 134:20250–20253
Dhital RN, Kamonsatikul C, Somsook E, Sakurai H (2013) Catal Sci Technol 3:3030–3035
Fan Q, Gottfried JM, Zhu J (2015) Acc Chem Res 48:2484–2494
Wang W, Shi X, Wang S, Van Hove MA, Lin N (2011) J Am Chem Soc 133:13264–13267
Lahrood AR, Björk J, Heckl WM, Lackinger M (2015) Chem Commun 51:13301–13304
Basagni A, Ferrighi L, Cattelan M, Nicolas L, Handrup K, Vaghi L, Papagni A, Sedona F, Valentin CD, Agnolia S, Sambia M (2015) Chem Commun 51:12593–12596
Eichhorn J, Nieckarz D, Ochs O, Samanta D, Schmittel M, Szabelski PJ, Lackinger M (2014) ACS Nano 8:7880–7889
Eichhorn J, Strunskus T, Rastgoo-Lahrood A, Samanta D, Schmittel M, Lackinger M (2014) Chem Commun 50:7680–7682
Wan Y, Chen J, Zhang D, Li H (2006) J Mol Cat A Chem 258:89–94
Li H, Chen J, Wan Y, Chai W, Zhang F, Lu Y (2007) Green Chem 9:273–280
Li H, Chai W, Zhang F, Chen J (2007) Green Chem 9:1223–1228
Wang H, Wan Y (2009) J Mater Sci 44:6553–6562
Yuan B, Pan Y, Li Y, Yin B, Jiang H (2010) Angew Chem Int Ed 49:4054–4058
Cheng J, Zhang G, Du J, Tang L, Xu J, Li J (2011) J Mater Chem 21:3485–3494
Kamal A, Srinivasulu V, Seshadri BN, Markandeya N, Alarifi A, Shankaraiah N (2012) Green Chem 14:2513–2522
Monopoli A, Cotugno P, Palazzo G, Ditaranto N, Mariano B, Cioffi N, Ciminale F, Nacci A (2012) Adv Synth Catal 354:2777–2788
Movahed SK, Dabiri M, Bazgir A (2014) App Cat A Gen 488:265–274
Karimi B, Behzadnia H, Vali H (2014) ChemCatChem 6:745–748
Dabiri M, Shariatipour M, Movahed SK, Bashiribod S (2014) RSC Adv 4:39428–39434
Zhang L, Wang A, Miller JT, Liu X, Yang X, Wang W, Li L, Huang Y, Mou CY, Zhang T (2014) ACS Catal 4:1546–1553
Wu X, Tan L, Chen D, Meng X, Tang F (2014) Chem Commun 50:539–541
Shen L, Simmons CJ, Sun D (2012) Tetrahedron Lett 53:4173–4178
Collins JC, Farley KA, Limberakis C, Liras S, Price D, James K (2012) J Org Chem 77:11079–11090
Baqi Y, Müller CE (2007) Org Lett 9:1271–1274
Liu ZJ, Vors JP, Gesing ERF, Bolm C (2011) Green Chem 13:42–45
Bagley MC, Dix MC, Fusillo V (2009) Tetrahedron Lett 50:3661–3664
Bagley MC, Fusillo V, Hills EGB, Mulholland AT, Newcombe J, Pentecost LJ, Radley EL, Stephens BR, Turrell CC (2012) Arkivoc vii:294–313
Narasimharao K, Al-Sabban E, Saleh TS, Gallastegui AG, Sanfiz AC, Basahel S, Al-Thabaiti S, Alyoubi A, Obaid A, Mokhtar M (2013) J Mol Catal A: Chem 379:152–162
Nelson KA, Adolph HG (1991) Synth Commun 21:293–305
Evano G, Blanchard N, Toumi M (2008) Chem Rev 108:3054–3131
Qian H, Wang Z, Yue W, Zhu D (2007) J Am Chem Soc 129:10664–10665
Zhang C, Sun Q, Chen H, Tan Q, Xu W (2015) Chem Commun 51:495–498
Xuan Z, Kim JH, Lee S (2015) J Org Chem 80:7824–7829
Gorobets E, McDonald R, Keay BA (2006) Org Lett 8:1483–1485
Cai Q, Yan J, Ding K (2012) Org Lett 14:3332–3335
Banwell MG, Kelly BD, Kokas OJ, Lupton DW (2003) Org Lett 5:2497–2500
Hauser FM, Gauuan PJF (1999) Org Lett 1:671–672
Meyers AI, Willemsen JJ (1997) Chem Commun 1573–1574
Degnan AP, Meyers AI (1999) J Am Chem Soc 121:2762–2769
Meyers AI, Willemsen JJ (1998) Tetrahedron 54:10493–10511
Meyers AI, Price A (1998) J Org Chem 63:412–413
Nelson TD, Meyers AI (1994) J Org Chem 59:2655–2658
Meyers AI (1998) J Het Chem 35:991–1002
Lin GQ, Zhong M (1996) Tetrahedron Lett 37:3015–3018
Banwell MG, Lupton DW (2005) Org Biomol Chem 3:213–215
Chen WW, Zhao Q, Xu MH, Lin GQ (2010) Org Lett 12:1072–1075
Acknowledgments
DST, Government of India, is greatly acknowledged for giving a research grant to the author through INSPIRE Faculty Award (No. IFA12/CH/56). The author is also grateful to Syamsundar College, for facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mondal, S. Recent advancement of Ullmann-type coupling reactions in the formation of C–C bond. ChemTexts 2, 17 (2016). https://doi.org/10.1007/s40828-016-0036-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40828-016-0036-2