Abstract
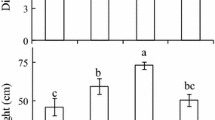
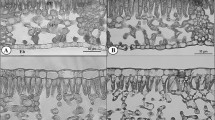
Stomata are small epidermal pores responsible for the strict control of the amount of CO2 that diffuses into the leaves while controlling the amount of water vapor lost to the atmosphere. The time required for the stomatal valve opening and closing is coordinated with an optimized hydraulic supply and strongly responds to the surrounding environment. We demonstrate that intense shading conditions promote high levels of plasticity in the woody species of Podocarpus macrophyllus, Eucalyptus urophylla, and Capsicum chinense, in a series of hydraulic, anatomical, and gas exchange traits—parameters that have been associated with optimized stomatal kinetics. The high levels of plasticity expressed in these species, however, did not translate into alterations in the time to reach 90% of the maximum stomatal conductance (gs) changes (t90) when plants were exposed to dynamic changes in irradiance. In some cases, the growth light affected the maximum slope of gs. This approach, however, was demonstrated not to be ideal for assessing stomatal speed in sun- and shade-acclimated plants as this method was largely dependent on maximum gs. Our findings suggest that stomatal speed, as demonstrated by t90, has low phenotypic plasticity and is most likely under a stronger genetic regulation than other leaf and stomatal anatomical traits.




Similar content being viewed by others
References
Baroli I, Price GD, Badger MR, Von Caemmerer S (2008) The contribution of photosynthesis to the red light response of stomatal conductance. Plant Physiol 146(2):737–747. https://doi.org/10.1104/pp.107.110924
Baruah J, Lal M (2020) Capsicum chinense Jacq.: ethnobotany, bioactivity and future prospects. In: Singh B (ed) Botanical leads for drug discovery. Springer, Singapore. https://doi.org/10.1007/978-981-15-5917-4_1
Brodribb TJ, Holbrook NM (2006) Declining hydraulic efficiency as transpiring leaves desiccate: two types of response. Plant Cell Environ 29(12):2205–2215. https://doi.org/10.1111/j.1365-3040.2006.01594.x
Brodribb TJ, McAdam SAM (2017) Evolution of the stomatal regulation of plant water content. Plant Physiol 174(2):639–649. https://doi.org/10.1104/pp.17.00078
Brodribb TJ, Sussmilch F, McAdam SAM (2020) From reproduction to production, stomata are the master regulators. Plant J 101(4):756–767. https://doi.org/10.1111/tpj.14561
Carins Murphy MR, Jordan GJ, Brodribb TJ (2012) Differential leaf expansion can enable hydraulic acclimation to sun and shade. Plant Cell Environ 35(8):1407–1418. https://doi.org/10.1111/j.1365-3040.2012.02498.x
Carins Murphy MR, Jordan GJ, Brodribb TJ (2016) Cell expansion not cell differentiation predominantly co-ordinates veins and stomata within and among herbs and woody angiosperms grown under sun and shade. Ann Bot 118(6):1127–1138. https://doi.org/10.1093/aob/mcw167
Carins Murphy MR, Jordan GJ, Brodribb TJ (2017) Ferns are less dependent on passive dilution by cell expansion to coordinate leaf vein and stomatal spacing than angiosperms. PLoS ONE 12(9):e0185648. https://doi.org/10.1371/journal.pone.0185648
Chen ZH, Eisenach C, Xu XQ, Hills A, Blatt MR (2012) Protocol: optimised electrophyiological analysis of intact guard cells from Arabidopsis. Plant Methods 8:1–11. https://doi.org/10.1186/1746-4811-8-15
Deans RM, Brodribb TJ, Busch FA, Farquhar GD (2018) Plant water-use strategy mediates stomatal effects on the light induction of photosynthesis. New Phytol 222(1):382–395. https://doi.org/10.1111/nph.15572
Drake PL, Froend RH, Franks PJ (2013) Smaller, faster stomata: scaling of stomatal size, rate of response, and stomatal conductance. J Exp Bot 64(2):495–505. https://doi.org/10.1093/jxb/ers347
Eisenach C, Angeli A (2017) Ion transport at the vacuole during stomatal movements. Plant Physiol 174(2):520–530. https://doi.org/10.1104/pp.17.00130
Eisenach C, Chen ZH, Grefen C, Blatt MR (2012) The trafficking protein SYP121 of Arabidopsis connects programmed stomatal closure and K + channel activity with vegetative growth. Plant J 69(2):241–251. https://doi.org/10.1111/j.1365-313X.2011.04786.x
Eisenach C, Papanatsiou M, Hillert EK, Blatt MR (2014) Clustering of the K+channel GORK of Arabidopsis parallels its gating by extracellular K+. Plant J 78(2):203–214. https://doi.org/10.1111/tpj.12471
Elliott-Kingston C, Haworth M, Yearsley JM, Batke SP, Lawson T, McElwain JC (2016) Does size matter? Atmospheric CO2 may be a stronger driver of stomatal closing rate than stomatal size in taxa that diversified under low CO2. Front Plant Sci 7:1253. https://doi.org/10.3389/fpls.2016.01253
Eyland D, Wesemael JV, Lawson T, Carpentier S (2021) The impact of slow stomatal kinetics on photosynthesis and water use efficiency under fluctuating light. Plant Physiol 186(2):998–1012. https://doi.org/10.1093/plphys/kiab114
Faralli M, Cockram J, Ober E, Wall S, Galle A, Rie JV, Raines C, Lawson T (2019) Genotypic, developmental and environmental effects on the rapidity of gs in wheat: impacts on carbon gain and water-use efficiency. Front Plant Sci 10:492. https://doi.org/10.3389/fpls.2019.00492
Franks PJ, Beerling DJ (2009) Maximum leaf conductance driven by CO>2 effects on stomatal size and density over geologic time. Proc Natl Acad Sci USA 106(25):10343–10347. https://doi.org/10.1073/pnas.090420910
Franks PJ, Farquhar GD (2007) The mechanical diversity of stomata and its significance in gas-exchange control. Plant Physiol 143(1):78–87. https://doi.org/10.1104/pp.106.089367
Franks PJ, Drake PL, Beerling DJ (2009) Plasticity in maximum stomatal conductance constrained by negative correlation between stomatal size and density: an analysis using Eucalyptus globulus. Plant Cell Environ 32(12):1737–1748. https://doi.org/10.1111/j.1365-3040.2009.002031.x
Gerardin T, Douthe C, Flexas J, Brendel O (2018) Shade and drought growth conditions strongly impact dynamic responses of stomata to variations in irradiance in Nicotiana tabacum. Environ Exp Bot 153:188–197. https://doi.org/10.1016/j.envexpbot.2018.05.019
Hetherington AM, Woodward FI (2003) The role of stomata in sensing and driving environmental change. Nature 424:901–908. https://doi.org/10.1038/nature01843
Hronková M, Wiesnerová D, Šimková M, Skůpa P, Dewitte W, Vráblová M, Zažímalová E, Šantrůček J (2015) Light-induced STOMAGEN-mediated stomatal development in Arabidopsis leaves. J Exp Bot 66(15):4621–4630. https://doi.org/10.1093/jxb/erv233
Inoue SI, Kinoshita T (2017) Blue light regulation of stomatal opening and the plasma membrane H+-ATPase. Plant Physiol 174(2):531–538. https://doi.org/10.1104/pp.17.00166
Kardiman R, Ræbild A (2018) Relationship between stomatal density, size and speed of opening in Sumatran rainforest species. Tree Physiol 38(5):696–705. https://doi.org/10.1093/treephys/tpx149
Knapp AK, Smith WK (1987) Stomatal and photosynthetic responses during sun/shade transitions in subalpine plants: influence on water use efficiency. Oecologia 74(1):62–67. https://doi.org/10.1007/BF00377346
Korson L, Drost-Hansen W, Millero FJ (1969) Viscosity of water at various temperatures. J Phys Chem 73(1):34–39. https://doi.org/10.1021/j100721a006
Lawson T, Blatt MR (2014) Stomatal size, speed, and responsiveness impact on photosynthesis and water use efficiency. Plant Physiol 164(4):1556–1570. https://doi.org/10.1104/pp.114.237107
Lawson T, Matthews J (2020) Guard cell metabolism and stomatal function. Annu Rev Plant Biol 29(71):273–302. https://doi.org/10.1146/annurev-arplant-050718-100251
Lawson T, von Caemmerer S, Baroli I (2010) Photosynthesis and stomatal behaviour. In: Lüttge UE, Beyschlag W, Büdel B, Francis D (eds) Progress in botany. Springer, Berlin, pp 265–304. https://doi.org/10.1007/978-3-642-13145-5_11
Lawson T, Kramer DM, Raines CA (2012) Improving yield by exploiting mechanisms underlying natural variation of photosynthesis. Curr Opin Biotechnol 23(2):215–220. https://doi.org/10.1016/j.copbio.2011.12.012
Lee JH, Jung JH, Park CM (2017) Light inhibits COP1-mediated degradation of ICE transcription factors to induce stomatal development in Arabidopsis. Plant Cell 29(11):2817–2830. https://doi.org/10.1105/tpc.17.00371
Martins SCV, Galmés J, Cavatte PC, Pereira LF, Ventrella MC, DaMatta FM (2014) Understanding the low photosynthetic rates of sun and shade coffee leaves: bridging the gap on the relative roles of hydraulic, diffusive and biochemical constraints to photosynthesis. PLoS ONE. https://doi.org/10.1371/journal.pone.0095571
Matthews JSA, Lawson T (2019) Climate change and stomatal physiology. Annu Plant Rev 2(3):1–39. https://doi.org/10.1002/9781119312994.apr0667
Matthews JSA, Vialet-Chabrand S, Lawson T (2018) Acclimation to fluctuating light impacts the rapidity of response and diurnal rhythm of stomatal conductance. Plant Physiol 176(3):1939–1951. https://doi.org/10.1104/pp.17.01809
McAdam SAM, Brodribb TJ (2015) The evolution of mechanisms driving the stomatal response to vapor pressure deficit. Plant Physiol 167(3):833–843. https://doi.org/10.1104/pp.114.252940
McAusland L, VialetChabrand S, Davey P, Baker NR, Brendel O, Lawson T (2016) Effects of kinetics of light-induced stomatal responses on photosynthesis and water-use efficiency. New Phytol 211(4):1209–1220. https://doi.org/10.1111/nph.14000
Medeiros DB, Martins SCV, Cavalcanti JHF, Daloso DM, Martinoia E, Nunes-Nesi A, DaMatta FM, Fernie AR, Araújo WL (2016) Enhanced photosynthesis and growth in atquac1 knockout mutants are due to altered organic acid accumulation and an increase in both stomatal and mesophyll conductance. Plant Physiol 170(1):86–101. https://doi.org/10.1104/pp.15.01053
Medeiros DB, Luz LM, Oliveira HO, Araújo WL, Daloso DM, Fernie AR (2019) Metabolomics for understanding stomatal movements. Theor Exp Plant Physiol 31:91–102. https://doi.org/10.1007/s40626-019-00139-9
Rodríguez-López NF, Martins SCV, Cavatte PC, Silva PEM, Morais LE, Pereira LF, Reis JV, Ávila RT, Godoy AG, Lavinski AO, DaMatta FM (2014) Morphological and physiological acclimations of coffee seedlings to growth over a range of fixed or changing light supplies. Environ Exp Bot 102:1–10. https://doi.org/10.1016/j.envexpbot.2014.01.008
Sack L, Melcher PJ, Zwieniecki MA, Holbrook NM (2002) The hydraulic conductance of the angiosperm leaf lamina: a comparison of three measurement methods. J Exp Botany 53(378):2177–2184. https://doi.org/10.1093/jxb/erf069
Schoch PG, Zinsou C, Sibi M (1980) Dependence of the stomatal index on environmental factors during stomatal differentiation in leaves of Vigna sinensis L. J Exp Botany 31(5):1211–1216. https://doi.org/10.1093/jxb/31.5.1211
Scoffoni C, Pou A, Aasamaa K, Sack L (2008) The rapid light response of leaf hydraulic conductance: new evidence from two experimental methods. Plant Cell Environ 31(12):1803–1812. https://doi.org/10.1111/j.1365-3040.2008.01884.x
Shimazaki KI, Doi M, Assmann SM, Kinoshita T (2007) Light regulation of stomatal movement. Annu Rev Plant Biol 58:219–247. https://doi.org/10.1146/annurev.arplant.57.032905.105434
Strittmatter CGD (1973) Nueva técnica de diafanización. Bot Soc Argent Bot 15(1):126–129
Vialet-Chabrand S, Matthews JS, Lawson T (2021) Light, power, action! Interaction of respiratory energy and blue light-induced stomatal movements. New Phytol 231(6):2231–2246
Vico G, Manzoni S, Palmroth S, Katul G (2011) Effects of stomatal delays on the economics of leaf gas exchange under intermittent light regimes. New Phytol 192(3):640–652. https://doi.org/10.1111/j.1469-8137.2011.03847.x
Wall S, Vialet-Chabrand S, Davey P, Van Rie J, Galle A, Cockram J, Lawson T (2022) Stomata on the abaxial and adaxial leaf surfaces contribute differently to leaf gas exchange and photosynthesis in wheat. New Phytol 235(5):1743–1756. https://doi.org/10.1111/nph.18257
Wei H, Kong D, Yang J, Wang H (2020) Light regulation of stomatal development and patterning: shifting the paradigm from Arabidopsis to grasses. Plant Commun 1(9):100030. https://doi.org/10.1016/j.xplc.2020.100030
Wong SC, Cowan IR, Farquhar GD (1979) Stomatal conductance correlates with photosynthetic capacity. Nature 282(5737):424–426
Woods DB, Turner NC (1971) Stomatal response to changing light by four tree species of varying shade tolerance. New Phytol 70(1):77–84. https://doi.org/10.1111/j.1469-8137.1971.tb02512.x
Yang J, Li C, Kong D, Guo F, Wei H (2020) Light-mediated signaling and metabolic changes coordinate stomatal opening and closure. Front Plant Sci 11:601478. https://doi.org/10.3389/fpls.2020.601478
Zwieniecki MA, Boyce CK, Holbrook NM (2004) Hydraulic limitations imposed by crown placement determine final size and shape of Quercus rubra L. leaves. Plant Cell Environ 27(3):357–365. https://doi.org/10.1111/j.1365-3040.2003.01153.x
Funding
This study was supported by the USDA National Institute of Food and Agriculture, Hatch Project 7003279 (AAC). FMD acknowledges research fellowships granted by the National Council for Scientific and Technological Development, Brazil (CNPq, Grant 305327/2019-4) and the Foundation for Research Assistance of Minas Gerais State, Brazil (FAPEMIG, Project CRA-RED-00053-16; APQ01512-18).
Author information
Authors and Affiliations
Contributions
AAC and TL designed the study; RSF, LAO, and AAC carried out the experiments; RSF and AAC analyzed the data and wrote the manuscript with revisions from FMD, SAMM, and TL; all authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Freitas, R.S., Oliveira, L.A., McAdam, S.A.M. et al. Woody species grown under sun and shade present similar stomatal speed. Theor. Exp. Plant Physiol. 35, 275–286 (2023). https://doi.org/10.1007/s40626-023-00283-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40626-023-00283-3