Abstract
Background
Recreational cycling is a popular activity which stimulates and improves cardiovascular fitness. The corresponding benefits for bone are unclear.
Purpose
This study examined the effect of running (high-impact) vs. cycling (low-impact), at the same moderate-to-vigorous exercise intensity, on markers of bone formation (N-terminal propeptide of type I collagen, PINP) and bone resorption (C-telopeptide of type I collagen, CTX-1), a non-collagenous bone remodeling marker (osteocalcin), as well as bone-modulating factors, including parathyroid hormone (PTH), irisin (myokine) and sclerostin (osteokine).
Methods
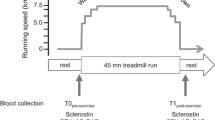
Thirteen healthy men (23.7 ± 1.0 y) performed two progressive exercise tests to exhaustion (peak VO2) on a cycle ergometer (CE) and on a treadmill (TM). On subsequent separate days, in randomized order, participants performed 30-min continuous running or cycling at 70% heart rate reserve (HRR). Blood was drawn before, immediately post- and 1 h into recovery.
Results
PTH transiently increased (CE, 51.7%; TM, 50.6%) immediately after exercise in both exercise modes. Sclerostin levels increased following running only (27.7%). Irisin increased following both running and cycling. In both exercise modes, CTX-1 decreased immediately after exercise, with no significant change in PINP and osteocalcin.
Conclusion
At the same moderate-to-vigorous exercise intensity, running appears to result in a greater transient sclerostin response compared with cycling, while the responses of bone markers, PTH and irisin are similar. The longer-term implications of this differential bone response need to be further examined.


Similar content being viewed by others
References
Nikander R, Sievänen H, Heinonen A et al (2010) Targeted exercise against osteoporosis: A systematic review and meta-analysis for optimising bone strength throughout life. BMC Med 8:47. https://doi.org/10.1186/1741-7015-8-47
Lima F, De Falco V, Baima J et al (2001) Effect of impact load and active load on bone metabolism and body composition of adolescent athletes. Med Sci Sports Exerc 33:1318–1323. https://doi.org/10.1097/00005768-200108000-00012
Dolan E, Varley I, Ackerman KE et al (2020) The bone metabolic response to exercise and nutrition. Exerc Sport Sci Rev 48:49–58. https://doi.org/10.1249/JES.0000000000000215
Rector RS, Rogers R, Ruebel M, Hinton PS (2008) Participation in road cycling vs running is associated with lower bone mineral density in men. Metabolism Clin Experim 57:226–232. https://doi.org/10.1016/j.metabol.2007.09.005
Barry DW, Kohrt WM (2008) BMD decreases over the course of a year in competitive male cyclists. J Bone Miner Res 23:484–491. https://doi.org/10.1359/jbmr.071203
Kohrt WM, Bloomfield SA, Little KD et al (2004) Physical activity and bone health. Med Sci Sports Exerc 36:1985–1996. https://doi.org/10.1249/01.MSS.0000142662.21767.58
Powell KE, King AC, Buchner DM, et al (2019) The scientific foundation for the physical activity guidelines for Americans, 2nd edition. J Phys Activ Health 16:1–11. https://doi.org/10.1123/jpah.2018-0618
Kouvelioti R, Kurgan N, Falk B et al (2019) Cytokine and sclerostin response to high-intensity interval running versus cycling. Med Sci Sports Exerc 51:2458–2464. https://doi.org/10.1249/MSS.0000000000002076
Hadjidakis DJ, Androulakis II (2006) Bone remodeling. In: Annals of the New York Academy of Sciences. Blackwell Publishing Inc., pp 385–396
Banfi G, Lombardi G, Colombini A, Lippi G (2010) Bone metabolism markers in sports medicine. Sports Med 40:697–714. https://doi.org/10.2165/11533090-000000000-00000
Vasikaran SD, Paul Chubb SA (2016) The use of biochemical markers of bone turnover in the clinical management of primary and secondary osteoporosis. Endocrine 52:222–225. https://doi.org/10.1007/s12020-016-0900-2
Bauer D, Krege J, Lane N et al (2012) National Bone Health Alliance Bone Turnover Marker Project: Current practices and the need for US harmonization, standardization, and common reference ranges. Osteoporos Int 23:2425–2433. https://doi.org/10.1007/s00198-012-2049-z
Winkler DG, Sutherland MK, Geoghegan JC et al (2003) Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. Eur Mol Biol Organiz 22:6267–6276
Robling AG, Bellido TM, Harris SE et al (2007) Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/Sclerostin. J Biol Chem 283:5866–5875. https://doi.org/10.1074/jbc.m705092200
Kouvelioti R, LeBlanc P, Falk B et al (2019) Effects of high-intensity interval running versus cycling on sclerostin, and markers of bone turnover and oxidative stress in young men. Calcif Tissue Int 104:582–590. https://doi.org/10.1007/s00223-019-00524-1
Kouvelioti R, Kurgan N, Falk B et al (2018) Response of sclerostin and bone turnover markers to high intensity interval exercise in young women: does impact matter? Biomed Res Int 2018:1–8. https://doi.org/10.1155/2018/4864952
Kouvelioti R, Kurgan N, Falk B et al (2019) Cytokine and sclerostin response to high-intensity interval running versus cycling. Med Sci Sports Exerc. https://doi.org/10.1249/mss.0000000000002076
Kaji H (2016) Effects of myokines on bone. BoneKEy Reports 5:1–6. https://doi.org/10.1038/bonekey.2016.48
Boström P, Wu J, Jedrychowski MP et al (2012) A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 481:463–468. https://doi.org/10.1038/nature10777
Farmer SR, Ph D (2019) Clinical implications of basic research boning up on Irisin. NEJM 1480–1482
Wittich A, Mautalen CA, Oliveri MB et al (1998) Professional football (soccer) players have a markedly greater skeletal mineral content, density and size than age- and BMI-matched controls. Calcif Tissue Int 63:112–117. https://doi.org/10.1007/s002239900499
Kim H, Wrann CD, Jedrychowski M et al (2018) Irisin . Cell 175:1756-1768.e17. https://doi.org/10.1016/j.cell.2018.10.025
Colaianni G, Cuscito C, Mongelli T et al (2015) The myokine irisin increases cortical bone mass. Proc Natl Acad Sci USA 112:12157–12162. https://doi.org/10.1073/pnas.1516622112
American Heart Association Recommendations for Physical Activity in Adults and Kids | American Heart Association. https://www.heart.org/en/healthy-living/fitness/fitness-basics/aha-recs-for-physical-activity-in-adults. Accessed 7 Jul 2020
Physical Activity Guidelines for Americans 2nd edition
Dill DB, Costill DL (1974) Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J Appl Physiol 37:247–248. https://doi.org/10.1152/jappl.1974.37.2.247
Borer KT (2013) Advanced exercise endocrinology. Chapter 10. p 219–228, 1st ed. Human Kinetics
Frings-Meuthen P, Boehme G, Liphardt AM et al (2013) Sclerostin and DKK1 levels during 14 and 21 days of bed rest in healthy young men. J Musculoskelet Neuronal Interactions 13:45–52
Spatz JM, Fields EE, Yu EW et al (2012) Serum sclerostin increases in healthy adult men during bed rest. J Clin Endocrinol Metab 97:1736–1740. https://doi.org/10.1210/jc.2012-1579
Falk B, Haddad F, Klentrou P et al (2016) Differential sclerostin and parathyroid hormone response to exercise in boys and men. Osteoporos Int 27:1245–1249. https://doi.org/10.1007/s00198-015-3310-z
Pickering ME, Simon M, Sornay-Rendu E et al (2017) Serum sclerostin increases after acute physical activity. Calcif Tissue Int 101:170–173. https://doi.org/10.1007/s00223-017-0272-5
Silva BC, Bilezikian JP (2015) Parathyroid hormone: Anabolic and catabolic actions on the skeleton. Curr Opin Pharmacol 22:41–50
Onyia JE, Helvering LM, Gelbert L et al (2005) Molecular profile of catabolic versus anabolic treatment regimens of parathyroid hormone (PTH) in rat bone: An analysis by DNA microarray. J Cell Biochem 95:403–418. https://doi.org/10.1002/jcb.20438
Barry DW, Kohrt WM (2007) Acute effects of 2 hours of moderate-intensity cycling on serum parathyroid hormone and calcium. Calcif Tissue Int 80:359–365. https://doi.org/10.1007/s00223-007-9028-y
Townsend R, Elliott-Sale KJ, Pinto AJ et al (2016) Parathyroid hormone secretion is controlled by both ionized calcium and phosphate during exercise and recovery in men. J Clin Endocrinol Metab 101:3231–3239. https://doi.org/10.1210/jc.2016-1848
Kohrt WM, Wherry SJ, Wolfe P et al (2018) Maintenance of serum ionized calcium during exercise attenuates parathyroid hormone and bone resorption responses. J Bone Miner Res 33:1326–1334. https://doi.org/10.1002/jbmr.3428
Scott JPR, Sale C, Greeves JP et al (2010) The role of exercise intensity in the bone metabolic response to an acute bout of weight-bearing exercise. J Appl Physiol 110:423–432. https://doi.org/10.1152/japplphysiol.00764.2010
Gardinier JD, Mohamed F, Kohn DH (2015) PTH signaling during exercise contributes to bone adaptation. J Bone Miner Res 30:1053–1063. https://doi.org/10.1002/jbmr.2432
Winn NC, Grunewald ZI, Liu Y et al (2017) Plasma irisin modestly increases during moderate and high-intensity afternoon exercise in obese females. PLoS ONE 12:1–12. https://doi.org/10.1371/journal.pone.0170690
Vegge G, Hollan I, Slettaløkken G et al (2015) Irisin in blood increases transiently after single sessions of intense endurance exercise and heavy strength training. PLoS ONE 10:e0121367. https://doi.org/10.1371/journal.pone.0121367
Nygaard H, Slettaløkken G, Vegge G et al (2015) Irisin in blood increases transiently after single sessions of intense endurance exercise and heavy strength training. PLoS ONE 10:e0121367. https://doi.org/10.1371/journal.pone.0121367
Scott JPR, Sale C, Greeves JP, et al (2011) The role of exercise intensity in the bone metabolic response to an acute bout of weight-bearing exercise. J Appl Physiol 110:423–32. https://doi.org/10.1152/japplphysiol.00764.2010
Guillemant J, Accarie C, Peres G, Guillemant S (2004) Acute effects of an oral calcium load on markers of bone metabolism during endurance cycling exercise in male athletes. Calcif Tissue Int 74:407–414. https://doi.org/10.1007/s00223-003-0070-0
Qvist P, Christgau S, Pedersen BJ et al (2002) Circadian variation in the serum concentration of C-terminal telopeptide of type I collagen (serum CTx): Effects of gender, age, menopausal status, posture, daylight, serum cortisol, and fasting. Bone 31:57–61. https://doi.org/10.1016/S8756-3282(02)00791-3
Acknowledgements
The authors thank all the participants for participating in our study, and the study coordinator (Pearl Lynne Law), the Human Performance Laboratory technician (Hoang Pham), UCI ICTS nurses and the Pediatric Exercise and Genomics Research Center Senior Biostatistician (Annamarie Stehli) for their assistance with different parts of the study.
Funding
This study was funded by NIH U01-TR002004 and PERC Systems Biology Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by Institutional Review Board at the University of California Irvine and carried out in accordance with the principles set out in the Declaration of Helsinki.
Informed consent
Written informed consent was obtained from all participants upon enrollment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dror, N., Carbone, J., Haddad, F. et al. Sclerostin and bone turnover markers response to cycling and running at the same moderate-to-vigorous exercise intensity in healthy men. J Endocrinol Invest 45, 391–397 (2022). https://doi.org/10.1007/s40618-021-01659-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-021-01659-5