Abstract
Background
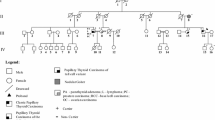
The familial nonmedullary thyroid cancer (FNMTC) is suspected to be a Mendelian condition in up to 3–8% of thyroid cancers. The susceptibility chromosomal loci and genes of 95% of FNMTC cases remain to be characterized. The inheritance of FNMTC appears to be autosomal dominant with incomplete penetrance and variable expressivity. The finding of the causative gene of FNMTC and the identification of patients at risk that need genetic testing were our aim.
Methods
We analyzed by whole-exome sequencing patients and non-affected relatives of five families with at least two family members affected by papillary thyroid cancer, selecting for new or extremely rare variants with predicted pathogenic value.
Results
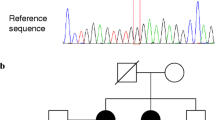
A family showed, in all three affected members, a new loss-of-function variant (frameshift deletion) in BROX gene at 1q41 that was absent from all internal and external databases. In a second family with three affected relatives, we found an additional new BROX variant. The smaller families presented no variants in BROX or in the other causative genes studied.
Conclusions
BROX could be a new causative gene for FNMTC. Variants in BROX may result in the haploinsufficiency of a key gene involved in the morphogenesis of MVBs, in the endosomal sorting of cargo proteins, and in EGFR. Functional studies are needed to support this result. The thorough genomic analysis by NGS in all families with three or more affected members should become a routine approach to obtain a comprehensive genetic view and find confirmative second cases.



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Durante C, Grani G, Lamartina L, Filetti S, Mandel SJ, Cooper DS (2018) The diagnosis and management of thyroid nodules: a review. JAMA 319:914–924
Howlader N, Noone AM, Krapcho M, Miller D, Bishop K, Kosary CL, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA (eds) (2017) SEER cancer statistics review, 1975–2014, National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/csr/1975_2014/, based on November 2016 SEER data submission, posted to the SEER web site.
Grossman RF, Tu SH, Duh QY, Siperstein AE, Novosolov F, Clark OH (1995) Familial nonmedullary thyroid cancer. An emerging entity that warrants aggressive treatment. Arch Surg 130:892–897
Loh KC, Lo JC, Greenspan FS, Miller TR, Yeo PP (1997) Familial papillary thyroid cancer: a case report. Ann Acad Med Singapore 26:503–506
Alsanea O, Wada N, Ain K, Wong M, Taylor K, Ituarte PH, Treseler PA, Weier HU, Freimer N, Siperstein AE, Duh QY, Takami H, Clark OH (2000) Is familial non-medullary thyroid carcinoma more aggressive than sporadic thyroid cancer? A multicenter series. Surgery 128:1043–1050
Uchino S, Noguchi S, Kawamoto H, Yamashita H, Watanabe S, Yamashita H, Shuto S (2002) Familial nonmedullary thyroid carcinoma characterized by multifocality and a high recurrence rate in a large study population. World J Surg 26:897–902
Lee YM, Yoon JH, Yi O, Tae-Yon Sung TY, Chung KW, Kim WB, Hong SJ (2014) Familial history of non-medullary thyroid cancer is an independent prognostic factor for tumor recurrence in younger patients with conventional papillary thyroid carcinoma. J Surg Oncol 109:168–173
Yu Y, Dong L, Li D, Chuai S, Wu Z, Zheng X, Cheng Y, Han L, Yu J, Gao M (2015) Targeted DNA sequencing detects mutations related to susceptibility among familial non-medullary thyroid cancer. Sci Rep 5:16129
Gara SK, Jia L, Merino MJ, Agarwal SK, Zhang L, Cam M, Patel D, Kebebew E (2013) Germline HABP2 mutation causing familial nonmedullary thyroid cancer. N Engl J Med 373:448–455
Sahasrabudhe R, Stultz J, Williamson J, TCUKIN et al (2016) The HABP2 G534E variant is an unlikely cause of familial non-medullary thyroid cancer. J Clin Endocrinol Metab 10:1098–1103
Alzahrani AS, Murugan AK, Qasem E, Al-Hindi H (2016) HABP2 Gene mutations do not cause familial or sporadic non-medullary thyroid cancer in a highly inbred middle eastern population. Thyroid 26:667–671
Weeks AL, Wilson SG, Ward L, Goldblatt J, Hui J, Walsh JP (2016) HABP2 germline variants are uncommon in familial nonmedullary thyroid cancer. BMC Med Genet 17:60
Peiling Yang S, Ngeow J (2016) Familial non-medullary thyroid cancer: unraveling the genetic maze. Endocr Relat Cancer 23:577–595
Haugen BR, Alexander EK, Bible KC et al (2015) (2016) American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer. Thyroid 26:1–133
Nikiforov YE, Carty SE, Chiosea SI, Coyne C, Duvvuri U, Ferris RL, Gooding WE, Hodak SP, LeBeau SO, Ohori NP, Seethala RR, Tublin ME, Yip L, Nikiforova MN (2014) Highly accurate diagnosis of cancer in thyroid nodules with follicular neoplasm/suspicious for a follicular neoplasm cytology by ThyroSeq v2 next-generation sequencing assay. Cancer 120:3627–3634
Nikiforova MN, Mercurio S, Wald AI, Barbi de Moura M, Callenberg K, Santana-Santos L, Gooding WE, Yip L, Ferris RL, Nikiforov YE (2018) Analytical performance of the ThyroSeq v3 genomic classifier for cancer diagnosis in thyroid nodules. Cancer 124:1682–1690
Agrawal N, Akbani R, Aksoy BA, on behalf of Cancer Genome Atlas Research Network (2014) Integrated genomic characterization of papillary thyroid carcinoma. Cell 159:676–690
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132
Morris LG, Sikora AG, Tosteson TD, Davies L (2013) The increasing incidence of thyroid cancer: the influence of access to care. Thyroid 23:885–891
Vriens MR, Suh I, Moses W, Kebebew E (2009) Clinical features and genetic predisposition to hereditary nonmedullary thyroid cancer. Thyroid 19:1343–1349
Khan A, Smellie J, Nutting C, Harrington K, Newbold K (2010) Familial nonmedullary thyroid cancer: a review of the genetics. Thyroid 20:795–801
Fagerberg L, Hallström BM, Oksvold P et al (2014) Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol Cell Proteomics 13:397–406
Pradhan-Sundd T, Verheyen EM (2014) The role of Bro1- domain-containing protein Myopic in endosomal trafficking of Wnt/Wingless. Dev Biol 392:93–107
McCullough J, Fisher RD, Whitby FG, Sundquist WI, Hill CP (2008) ALIX-CHMP4 interactions in the human ESCRT pathway. Proc Natl Acad SciUSA 105:7687–7691
Mu R, Dussupt V, Jiang J, Sette P, Rudd V, Chuenchor W, Bello NF, Bouamr F, Xiao TS (2012) Two distinct binding modes define the interaction of Brox with the C-terminal tails of CHMP5 and CHMP4B. Structure 20:887–898
Matoulkova E, Michalova E, Vojtesek B, Hrstka R (2012) The role of the 3' untranslated region in post-transcriptional regulation of protein expression in mammalian cells. RNA Biol 95:563–576
Klubo-Gwiezdzinska J, Yang L, Merkel R, Patel D, Nilubol N, Merino MJ, Skarulis M, Sadowski SM, Kebebew E (2017) Results of screening in familial non-medullary thyroid cancer. Thyroid 27:1017–1024
Capezzone M, Marchisotta S, Cantara S, Busonero G, Brilli L, Pazaitou-Panayiotou K, Carli AF, Caruso G, Toti P, Capitani S, Pammolli A, Pacini F (2008) Familial non-medullary thyroid carcinoma displays the features of clinical anticipation suggestive of a distinct biological entity. Endocr Relat Cancer 15:1075–1081
Capezzone M, Secchi C, Fralassi N, Cantara S, Brilli L, Ciuoli C, Pilli T, Maino F, Forleo R, Pacini F, Castagna MG (2019) Should familial disease be considered as a negative prognostic factor in micropapillary thyroid carcinoma? J Endocrinol Invest 42:1205–1213
Wang X, Cheng W, Li J, Su A, Wei T, Liu F, Zhu J (2015) Endocrine tumours: familial nonmedullary thyroid carcinoma is a more aggressive disease: a systematic review and meta-analysis. Eur J Endocrinol 172:253–262
Hińcza K, Kowalik A, Kowalska A (2019) Current knowledge of germline genetic risk factors for the development of non-medullary thyroid cancer. Genes (basel) 10:482
Ye F, Gao H, Xiao L et al (2019) Whole exome and target sequencing identifies MAP2K5 as novel susceptibility gene for familial non-medullary thyroid carcinoma. Int J Cancer 144:1321–1330
Cirello V, Colombo C, Persani L, Fugazzola L (2019) Absence of the MAP2K5 germline variants c.G961A and c.T1100C in a wide series of familial nonmedullary thyroid carcinoma Italian families. Int J Cancer. 145:600
Wang X, Cheng W, Li J, Su A, Wei T, Liu F, Zhu J (2019) Identification of rare variants predisposing to thyroid cancer. Thyroid 29:946–955
Acknowledgements
We thank the study participants, their families, and all the investigators and study staff members. VN Supported by grants from Telethon 2015 “Medicina Traslazionale in Oncologia: Dalla Ricerca alla Terapia PON01_02418”, Telethon 2016–2018 “Telethon Undiagnosed Disease Program”, 2018-22 EU Research Funding H2020-HEALTH-SC1-2017-RTD: “SOLVE RD”.
Funding
VN Supported by grants from Telethon 2015 “Medicina Traslazionale in Oncologia: Dalla Ricerca alla Terapia PON01_02418”, Telethon 2016–2018 “Telethon Undiagnosed Disease Program”, 2018-22 EU Research Funding H2020-HEALTH-SC1-2017-RTD: “SOLVE RD”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have nothing to disclose.
Ethics approval
The study was approved by the local ethical committee of the Università della Campania “Luigi Vanvitelli” protocol n°54 del 2/5/2019 on Horizon 2020 Solve the Unsolved project.
Informed consent
The informed consent was obtained from the patients included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pasquali, D., Torella, A., Accardo, G. et al. BROX haploinsufficiency in familial nonmedullary thyroid cancer. J Endocrinol Invest 44, 165–171 (2021). https://doi.org/10.1007/s40618-020-01286-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-020-01286-6