Abstract
Purpose of Review
Malaria transmission depends on the development and survival in humans of mature sexual stage parasites, known as gametocytes which further develop within the mosquito midgut to eventually form sporozoites that are reintroduced to the human host to complete the transmission cycle. This review discusses some recent findings on phagocytic clearance of the gametocytes for novel transmission-blocking strategies towards malaria elimination.
Recent Findings
Antibodies to gametocytes are present after natural infection, and phagocytosis of immature gametocytes has been shown. There is evidence that the mature live gametocytes evade the phagocytic clearance pathway possibly providing transmission advantage. Dead gametocytes are effectively phagocytosed.
Summary
Phagocytosis of gametocytes by macrophages can result in decreased gametocyte load in humans while concurrently channel antigens into the antigen presentation pathways and induction of natural transmission-blocking antibodies. Thus, clearing gametocytes as they develop in the human host to block transmission or preventing subsequent development of the parasite in the mosquito should be an important component of malaria elimination strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Major malaria-causing parasites in humans, predominantly Plasmodium falciparum and Plasmodium vivax, are responsible for 300,000–500,000 deaths annually worldwide. The World Malaria Report 2016 indicates that there were 212 million new cases and 429,000 malaria deaths reported worldwide in 2015. Children under the age 5 years and pregnant women bear the greatest risk of malaria mortality. Significant reductions in morbidity and mortality globally due to malaria over the last decade have been attributed to the widespread use of insecticide-treated nets (ITNs), indoor residual spraying (IRS), and artemisinin combination therapies (ACT). In sub-Saharan Africa alone, 43% of the population were not covered by ITNs or IRS, 69% of pregnant women did not receive required three doses of intermittent preventive treatment in pregnancy (IPTp), and 36% of children with fever were not provided care (http://apps.who.int/iris/bitstream/10665/254912/1/WHO-HTM-GMP-2017.4-eng.pdf?ua=1). In many countries with a high malaria burden, health systems remain under-resourced and poorly accessible to those most at risk of malaria. Similarly, substantial gaps remain in malaria intervention programs in other parts of the world. In order to effectively control and reach the goal of malaria elimination, innovative approaches are still needed to understand the patterns of malaria infection and transmission because despite improvements in access to core malaria control tools, substantial gaps remain (http://www.who.int/malaria/publications/world-malaria-report-2016/en/).
The emergence of artemisinin-resistant parasites [1, 2] has threatened some of the reported positive trends. There are no effective vaccines currently available, drugs are continuously losing their therapeutic efficacy due to development of resistance to multiple antimalarial drugs, and mosquito vectors are becoming resistant to insecticides [3]. These factors underscore and stress the need for developing effective malaria vaccine(s) to help with the long-term goal of gradual malaria elimination and eradication [4, 5•]. In particular, vaccines targeting transmission of malaria are viewed as an important tool for malaria elimination [6]. There are currently no licensed vaccines against malaria or any other human parasite. The only malaria vaccine, Mosquirix™ (RTS,S/ASO1) based on P. falciparum circumsporozoite protein (PfCSP), and advanced through phase III trials, has shown < 40% protective efficacy [7]. Such a bleak outcome underscores the intricacy of malaria vaccine development and need for renewed vaccine discovery efforts towards better efficacious products to realize the goal of malaria elimination via transmission reduction [8]. In this focused report, we will review the current knowledge relating to malaria transmission reduction efforts targeting the parasite and speculate on future research directions that may help in achieving this goal.
Malaria Transmission and Plasmodium Life Cycle
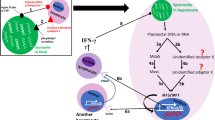
In general, asexually reproducing forms of the Plasmodium parasites within their human hosts are responsible for pathology and symptoms of malaria. But malaria transmission depends on the development and survival in humans of mature sexual stage parasites, known as gametocytes. Once ingested by a mosquito taking a blood meal, gametocytes must complete sexual reproduction within the mosquito midgut and eventually form sporozoites which are reintroduced to the human host to complete the transmission cycle. Transmission of the malaria parasite between humans and mosquitoes through several distinct developmental stages is required for malaria infection and disease symptoms to be manifest. The life cycle of the Plasmodium is a complex process involving the mosquito and the human. All four Plasmodium species responsible for the transmission from mosquitoes to humans, P. falciparum, P. vivax, P. ovale, and P. malariae, exhibit similar life cycle with minor variations. Malaria transmission depends upon the development of intraerythrocytic sexual stages (male and female gametocytes), ingestion by female anopheline mosquitoes, and subsequent sexual development leading to formation of sporozoites [9, 10]. Sporozoites injected by infected Anopheles mosquitoes invade hepatocytes, thus initiating malaria infection cycle. Each life cycle stage of the malaria parasite presents specific antigenic targets for vaccine development. Immune interventions aimed at blocking the liver stage development (pre-erythrocytic phase—PE) are expected to provide an effective way to prevent infection. Immune response to various erythrocytic life cycle forms (asexual and sexual) of Plasmodium plays an important role in resistance to malaria and in reducing malaria transmission through Anopheles mosquitoes.
To be an effective tool for malaria elimination and eradication, a vaccine must have a direct impact on malaria transmission. A vaccine based on the antigens expressed in the erythrocytic sexual and mosquito stages (gametocytes, gametes, zygotes, and ookinetes, Fig. 1) will have a direct impact on the transmission process. Previous studies have identified key target antigens of transmission-blocking vaccines (TBVs), which in P. falciparum include Pfs230 and Pfs48/45 (expressed in gametocytes and gametes) and Pfs25 and Pfs28 (zygotes and ookinetes), with known orthologs in P. vivax. Pre-clinical studies in mice and non-human primates with adjuvant-formulated recombinant Pfs25, Pvs28, and Pfs48/45 and clinical trials with Pvs28 and Pfs25 [11,12,13,14,15,16,17] have firmly established them as target antigens for the development of effective TBVs (Fig. 1).
Natural Immunity Against Malaria
Natural immunity against asexual stage parasites develops with repetitive exposure to malaria parasites and thus forms the basis of clinical immunity against malaria [18,19,20,21,22,23]. Though clinical immunity to malaria has been shown to develop in individuals, it does not prevent reinfection due to several factors including the complex life cycle of the parasite and insufficiency of the magnitude of effective antibodies. Susceptibility to P. falciparum was shown to be independent of the number of years of exposure to the parasite, and age-dependent protective immunity developed over a 2-year period of repeated exposure to infection pressure [19]. Studies have shown the presence of antibodies recognizing stage-specific parasite antigens in the sera of malaria-exposed individuals [24]. Some of these proteins are important for parasite invasion while others are responsible for sequestration of parasites in the blood vessels and in bone marrow niches among others. In addition to asexual stages, antigens expressed on erythrocytic sexual (male and female gametocytes) stages have also been shown to elicit antibody responses during natural infection and the antibody levels increase with repeat transmission cycles. Studies have established that some of these antigens are targets of antibody responses during natural infection [9] and are being pursued as candidate antigens for transmission-blocking vaccines.
Immune Responses to Gametocytes During Natural Infection
The development of gametocytes begins when some of the replicating asexual erythrocytic stages become either a male or a female gametocyte, which are non-dividing sexual stages [25]. The gametocytes undergo maturation through distinct antigenically and morphologically defined stages I through V [26]. Stages II–IV are sequestered in the bone marrow [27,28,29] while the mature stage V gametocytes are observed in the blood. These newly formed gametocytes developing as intraerythrocytic stages present several new antigens to the human immune system during each infection cycle. These immunologically identifiable forms are clear targets for the host immune system and both innate and adaptive immune responses are expected to become active against them. Naturally acquired antibodies against gametocytes exist [30,31,32,33] and may play a role in controlling transmission by limiting the gametocyte development in the circulation or by interrupting gamete development and fertilization in the mosquito following ingestion. In particular, antibodies against Pfs48/45 and Pfs230 gametocyte proteins effectively target male and female gamete surface and block mosquito stage development of the malaria parasites.
Analyses of investigations carried out in areas of different malaria endemicity have shown wide variation in the frequency and prevalence of these antibodies [9]. Studies have demonstrated that while natural immunity does develop against sexual stages, little is known about age-dependent pattern of the antibodies. Age dependency of the humoral response can be associated with repetitive exposure to the parasite [34,35,36,37], and as such, the development of the naturally occurring transmission-blocking antibodies can be assumed to be due to priming and boosting during repeated bouts of natural infection. Using cross-sectional design with only one sampling point, it has been reported that sera from 30 to 65% of exposed adults contain partially effective (> 65% transmission-blocking effect) antibodies. A much larger proportion (> 70%, approaching 100%) of sera from endemic populations have antibodies against an internal gametocyte protein (Pfg27) and it can be used as a proxy to gametocyte exposure [38]. Finally, an immune response may also target antigens present on the surface of gametocytes [39, 40]. Thus, age, frequency of infection, exposure to gametocytes, and multiplicity of infection are all likely to have an impact on natural transmission-reducing activity.
Analysis of antibody targets of naturally acquired immunity against gametocytes can be instrumental in discovery of novel molecules leading to the development of effective TBV approaches. Several reports provide in-depth analysis of the immunity against the sexual stages of Plasmodium which are critical for malaria transmission. Paul et al. provide an in-depth analysis of the immunity against the sexual stages of Plasmodium which are critical for malaria transmission [37], both during gametocyte development in humans, as well as after gametocyte activation in the mosquito midgut. Epitopes on the gametocyte-infected erythrocyte surface and on the gametes have been studied for potential transmission-blocking activity and future vaccine targets [41].
Role for Macrophages in Malaria Infection
While much is understood about the adaptive immune responses (both cellular and humoral), little is understood about the role of innate immune responses in malaria. Innate immune responses are the inherent defense mechanisms that are triggered by the host in response to a new or foreign antigen. Innate defense mechanisms are preexisting quiescent responses that are activated by the recognition of pathogen-associated molecular patterns (PAMP) or newly formed non-self antigens by immune cells of the host through its pattern recognition receptors (PRR). Resident phagocytic cells, predominantly macrophages, also known as scavenger cells, essentially remove biological debris and unwanted harmful pathogens from tissue sites by recognizing PAMP or non-self antigens on infected cells. These early immune responses are non-specific generated by PAMP-PRR interactions, and complement activation and opsonization of parasites by complement factors. Pre-formed antibodies specifically recognizing antigens on the surface of infected erythrocytes can also opsonize erythrocytes infected with asexual and sexual stages resulting in immune clearance via antibody-dependent cellular inhibition or cytotoxicity [42] or immunophagocytosis [43]. Opsonization triggers phagocytosis either via binding to complement receptors that bind complement factor-coated parasites or via Fc receptors that will bind antibody-coated parasites.
Previous studies have reported on phagocytosis of various opsonized stages of erythrocytic asexual stages of the parasite and these studies revealed evidence for phagocytosis of free merozoite [44] and erythrocytes infected with parasites at various degrees of maturity (ring, trophozoite, and schizont) [45, 46].
Phagocytosis of Gametocytes
Several published studies have suggested that leukocytes or white blood cells mediate reduction in transmission of P. falciparum primarily via their phagocytic activity [47, 48]. Smith et al. reported that immature stages I and II of gametocytes can be phagocytosed by macrophages [49]. However, there is scant information on phagocytic clearance of mature stage V gametocytes that are in circulation. It is only the mature gametocyte stages that circulate and mediate transmission to blood feeding mosquitoes. Recently, Bansal et al. for the first time demonstrated that live mature stages of P. falciparum gametocytes are not phagocytosed by activated monocytes in vitro. In contrast, when gametocytes were released as free forms by saponin treatment or heat killed (65 °C, 5–10 min), activated monocytes were able to phagocytose the gametocytes [50••]. Their studies evaluated phagocytic uptake of mature gametocytes by a human monocyte cell line in the absence of immune sera. Most previous published studies were not clear about phagocytic uptake of live vs killed gametocytes. Phagocytosis of the extracellular gametocytes of P. falciparum has been demonstrated in the blood meal in vitro (prior to ingestion) and in vivo within the mosquito midgut [47]. Infected blood fed to mosquitoes was examined over time for evidence of extracellular parasites by microscopy of Giemsa-stained slides. Up to 84% of the gametocytes were phagocytosed within two hours in vitro, compared to 7% in the same period in the mosquito gut.
Opsonization by circulating antibodies may further enhance phagocytosis of gametocytes, and we expect that opsonizing activity in the immune sera will demonstrate age-related patterns due to repeat exposures. Additionally, identification of parasite surface molecules recognized by opsonizing antibodies will identify novel targets of innate immune clearance of gametocytes.
Antibodies recognizing antigens on the surface of gametocytes and immune clearance by macrophages of such opsonized gametocytes may override the mechanism by which direct phagocytosis of live mature gametocytes is thwarted. It is reasonable to hypothesize that mature gametocytes not transmitted to mosquitoes in a blood meal are likely internalized by the phagocytic cells, either as dead or opsonized live parasites, resulting in reduced availability for transmission. Simultaneously, phagocytosed parasites undergo presentation of gametocyte antigens to the adaptive immune system resulting in elicitation of specific antibodies. Understanding these mechanisms will inform ways to effectively exploit innate immune parameters for inducing immune responses for transmission reduction. It is interesting to speculate on the mechanism by which live mature gametocyte evades being phagocytosed. Several investigators have published on the role of CD47, “a marker of self” molecule in the inhibition of phagocytosis by monocytes and macrophages. CD47 is a ubiquitously expressed cell surface glycoprotein which regulates the integrin responsiveness to RGD containing extracellular matrix proteins and was subsequently reported to be present on the surface of erythrocytes which do not express integrins [51]. Four different isoforms of CD47 attributed to tissue-specific expression are currently known. Immune cells are responsible for recognizing healthy host cells as self and not mount an immune response against them. This was recognized to be due to the recognition of the CD47 molecule on self cells by the signal regulatory protein-α (SIRP-α) which functions to inhibit the phagocytic property of the macrophage. Ayi et al. and Banerjee et al. demonstrated that expression of CD47 on the surface of erythrocytes prevents phagocytosis of the infected erythrocytes and also that infection leads to a decrease in CD47 expression. These studies were done with asexual stages of the parasite which are very different from the sexual stages and in particular the mature stage V gametocyte [51,52,53,54,55].
Conclusion
The consequences of immune responses to sexual stages of Plasmodium can have an important bearing on developing strategies in a malaria elimination plan. Since the sexual stages, most critically the mature gametocyte stage, are essential for the continued transmission cycle, any interruption to the further development of the malaria parasite will result in no transmission of viable parasites to initiate infection in humans. This will also result in decreased disease manifestation. Thus, understanding immune responses against the developing gametocytes in the human host can inform the design of more effective approaches to prevent transmission.
It is unclear whether gametocytes of varying degree of developmental maturity differ in their ability to be phagocytosed. Likewise, it is also not firmly established how natural antibody responses to the gametocytes came to be. For immune responses to be manifest, these circulating gametocytes clearly have had to be recognized by the antigen-presenting cells of the immune system, such as macrophages, and triggered a B cell response. If live gametocytes resist being phagocytosed [50••], clearly there are other mechanisms at play that led to the development of an antibody response. Opsonization by antibodies or complement factors provides a highly likely route for ingestion and antigen processing by macrophages. Dead or senesced circulating mature gametocytes likely are readily phagocytosed directly or after opsonization with specific antibodies present in immune sera from people exposed to malaria. Depending upon exposure frequency and intensity of infection, this may amount to efficient clearance of gametocytes and maintenance of natural transmission-reducing immune status. In general, malaria immunity during repeat infection is slow to develop, and it is expected that opsonizing activity by gametocyte-specific antibodies in the immune sera will demonstrate age-related patterns.
The unexpected findings of Bansal et al. that intact mature gametocyte stages are not phagocytosed suggest that the inability of macrophages to phagocytose mature gametocytes may actually provide transmission advantage to the malaria parasite. In contrast, mature gametocytes that are not picked up in a blood meal by mosquitoes and remain in the human host will eventually die and be phagocytosed. They will subsequently invoke immune responses that may possess transmission-blocking potential. Therefore, improved understanding of early immune responses to gametocytes initiated by macrophages may elucidate novel molecules that the parasite uses to avoid being phagocytosed, or identify additional mature gametocyte antigens that can be exploited for transmission-blocking strategies. Opsonic and non-opsonic phagocytosis-mediated immune activation and presentation of gametocyte-specific antigens in the form of dying parasites will together contribute to overall natural transmission-blocking immunity. It is logical to propose that, on the one hand, such clearance of parasites will decrease the parasite load and, on the other hand, it will also channel specific antigens for immune activations and induction of immunity targeting sexual stages. Both processes will account for reduced malaria transmission potential. Thus, vaccines aimed at generating an immune response against gametocyte antigens would be expected to influence survival of gametocytes that are crucial to the transmission process and prevent further sexual development in the mosquito midgut.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ariey F, Witkowski B, Amaratunga C, Beghain J, Langlois AC, Khim N, et al. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature. 2014;505(7481):50–5. https://doi.org/10.1038/nature12876.
Takala-Harrison S, Jacob CG, Arze C, Cummings MP, et al. Independent emergence of artemisinin resistance mutations among Plasmodium falciparum in Southeast Asia. J Infec Dis. 2015;211(5):670–79. https://doi.org/10.1093/infdis/jiu491.
Alout H, Roche B, Dabiré RK, Cohuet A. Consequences of insecticide resistance on malaria transmission. PLoS Pathog. 2017;13(9):e1006499. https://doi.org/10.1371/journal.ppat.1006499.
Alonso PL, Brown G, Arevalo-Herrera M, Binka F, Chitnis C, Collins F, et al. A research agenda to underpin malaria eradication. PLoS Med. 2011;8(1):e1000406. https://doi.org/10.1371/journal.pmed.1000406.
• Wu Y, Sinden RE, Churcher TS, Tsuboi T, Yusibov V. Chapter three-development of malaria transmission-blocking vaccines: from concept to product. Adv Parasitol. 2015;89:109–152. This paper reviews concept of transmission-blocking immunity against sexual and mosquito midgut stages. https://doi.org/10.1016/bs.apar.2015.04.001.
Lyke KE. Steady progress toward a malaria vaccine. Curr Opin Infect Dis. 2017;30(5):463–70. https://doi.org/10.1097/QCO.0000000000000393.
Wilby KJ, Lau TT, Gilchrist SE, Ensom MH. Mosquirix (RTS,S): a novel vaccine for the prevention of Plasmodium falciparum malaria. Ann Pharmacother. 2012;46(3):384–93. https://doi.org/10.1345/aph.1Q634.
The mal, E.R.A.C.G.o.V. A research agenda for malaria eradication: vaccines. PLoS Med. 2011;8(1):e1000398.
Bousema T, Drakeley C. Epidemiology and infectivity of Plasmodium falciparum and Plasmodium vivax gametocytes in relation to malaria control and elimination. Clin Microbiol Rev. 2011;24(2):377–410. https://doi.org/10.1128/CMR.00051-10.
Kumar N. A vaccine to prevent transmission of human malaria: a long way to travel on a dusty and often bumy road. Curr Sci. 2007;92(11):1535–44.
Malkin E, et al. Phase 1 vaccine trial of Pvs25H: a transmission blocking vaccine for Plasmodium vivax malaria. Vaccine. 2005;23(24):3131–8. https://doi.org/10.1016/j.vaccine.2004.12.019.
Wu Y, Ellis RD, Shaffer D, Fontes E, Malkin EM, Mahanty S, et al. Phase 1 trial of malaria transmission blocking vaccine candidates Pfs25 and Pvs25 formulated with montanide ISA 51. PLoS One. 2008;3(7):e2636. https://doi.org/10.1371/journal.pone.0002636.
Datta D, Bansal GP, Gerloff DL, Ellefsen B, Hannaman D, Kumar N. Immunogenicity and malaria transmission reducing potency of Pfs48/45 and Pfs25 encoded by DNA vaccines administered by intramuscular electroporation. Vaccine. 2017;35(2):264–72. https://doi.org/10.1016/j.vaccine.2016.11.072.
Cao Y, Bansal GP, Merino K, Kumar N. Immunological cross-reactivity between malaria vaccine target antigen P48/45 in Plasmodium vivax and P. falciparum and cross-boosting of immune responses. PLoS One. 2016;11(7):e0158212. https://doi.org/10.1371/journal.pone.0158212.
Kumar R, Nyakundi R, Kariuki T, Ozwara H, Nyamongo O, Mlambo G, et al. Functional evaluation of malaria Pfs25 DNA vaccine by in vivo electroporation in Olive baboons. Vaccine. 2013;31(31):3140–7. https://doi.org/10.1016/j.vaccine.2013.05.006.
Kumar R, Angov E, Kumar N. Potent malaria transmission-blocking antibody responses elicited by Plasmodium falciparum Pfs25 expressed in Escherichia coli after successful protein refolding. Infect Immun. 2014;82(4):1453–9. https://doi.org/10.1128/IAI.01438-13.
Kumar R, Ledet G, Graves R, Datta D, Robinson S, Bansal GP, et al. Potent functional immunogenicity of Plasmodium falciparum transmission-blocking antigen (Pfs25) delivered with nanoemulsion and porous polymeric nanoparticles. Pharm Res. 2015;32(12):3827–36. https://doi.org/10.1007/s11095-015-1743-x.
Baird JK. Host age as a determinant of naturally acquired immunity to Plasmodium falciparum. Parasitol Today. 1995;11(3):105–11. https://doi.org/10.1016/0169-4758(95)80167-7.
Baird JK, et al. Age-dependent acquired protection against Plasmodium falciparum in people having two years exposure to hyperendemic malaria. The American Journal of Tropical Medicine and Hygiene. 1991;45(1):65–76. https://doi.org/10.4269/ajtmh.1991.45.65.
Baird JK. Age dependent characteristics of protection v. susceptibility to Plasmodium falciparum. Ann Trop Med Parasitol. 1998;92(4):367–90. https://doi.org/10.1080/00034989859366.
Day KP, Marsh K. Naturally acquired immunity to Plasmodium faldparum. Immunol Today. 1991;12(3):A68–71. https://doi.org/10.1016/S0167-5699(05)80020-9.
Hviid L. Naturally acquired immunity to Plasmodium falciparum malaria in Africa. Acta Trop. 2005;95(3):270–5. https://doi.org/10.1016/j.actatropica.2005.06.012.
Trape J-F, et al. The Dielmo project: a longitudinal study of natural malaria infection and the mechanisms of protective immunity in a community living in a holoendemic area of Senegal. The American Journal of Tropical Medicine and Hygiene. 1994;51(2):123–37. https://doi.org/10.4269/ajtmh.1994.51.123.
Vermeulen A, et al. Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med. 1985;162(5):1460–76. https://doi.org/10.1084/jem.162.5.1460.
Guttery DS, Roques M, Holder AA, Tewari R. Commit and transmit: molecular players in plasmodium sexual development and zygote differentiation. Trends Parasitol. 2015;31(12):676–85. https://doi.org/10.1016/j.pt.2015.08.002.
Carter R, Miller LH. Recent developments in production and purification of malaria antigens: evidence for environmental modulation of gametocytogenesis in Plasmodium falciparum in continuous culture. Bull World Health Organ. 1979;57(Suppl):37–52.
Farfour E, Charlotte F, Settegrana C, Miyara M, Buffet P. The extravascular compartment of the bone marrow: a niche for Plasmodium falciparum gametocyte maturation. Malar J. 2012;11(1):285. https://doi.org/10.1186/1475-2875-11-285.
Aguilar R, Magallon-Tejada A, Achtman AH, Moraleda C, Joice R, Cistero P, et al. Molecular evidence for the localization of Plasmodium falciparum immature gametocytes in bone marrow. Blood. 2014;123(7):959–66. https://doi.org/10.1182/blood-2013-08-520767.
Joice R, Nilsson SK, Montgomery J, Dankwa S, Egan E, Morahan B, et al. Plasmodium falciparum transmission stages accumulate in the human bone marrow. Sci Transl Med. 2014;6(244):244re5. https://doi.org/10.1126/scitranslmed.3008882.
Gebru T, Ajua A, Theisen M, Esen M, Ngoa UA, Issifou S, et al. Recognition of Plasmodium falciparum mature gametocyte-infected erythrocytes by antibodies of semi-immune adults and malaria-exposed children from Gabon. Malar J. 2017;16(1):176. https://doi.org/10.1186/s12936-017-1827-7.
Paul NH, Vengesai A, Mduluza T, Chipeta J, Midzi N, Bansal GP, et al. Prevalence of Plasmodium falciparum transmission reducing immunity among primary school children in a malaria moderate transmission region in Zimbabwe. Acta Trop. 2016;163:103–8. https://doi.org/10.1016/j.actatropica.2016.07.023.
Bansal GP, Vengesai A, Cao Y, Mduluza T, Kumar N. Antibodies elicited during natural infection in a predominantly Plasmodium falciparum transmission area cross-react with sexual stage-specific antigen in P. vivax. Acta Trop. 2017;170:105–11. https://doi.org/10.1016/j.actatropica.2017.02.032.
Graves P, et al. Antibodies to Plasmodium faciparum gamete surface antigens in Papua New Guinea sera. Parasite Immunol. 1988;10(2):209–18. https://doi.org/10.1111/j.1365-3024.1988.tb00215.x.
Drakeley C, Sutherland C, Bousema JT, Sauerwein RW, Targett GAT. The epidemiology of Plasmodium falciparum gametocytes: weapons of mass dispersion. Trends Parasitol. 2006;22(9):424–30. https://doi.org/10.1016/j.pt.2006.07.001.
Ouedraogo AL, et al. Naturally acquired immune responses to Plasmodium falciparum sexual stage antigens Pfs48/45 and Pfs230 in an area of seasonal transmission. Infect Immun. 2011;79(12):4957–64. https://doi.org/10.1128/IAI.05288-11.
Graves PM, Wirtz RA, Carter R, Burkot TR, Looker M, Targett GA. Naturally occurring antibodies to an epitope on Plasmodium falciparum gametes detected by monoclonal antibody-based competitive enzyme-linked immunosorbent assay. Infect Immun. 1988;56(11):2818–21.
Paul RE, Bonnet S, Boudin C, Tchuinkam T, Robert V. Age-structured gametocyte allocation links immunity to epidemiology in malaria parasites. Malar J. 2007;6(1):123. https://doi.org/10.1186/1475-2875-6-123.
Riley EM, Bennett S, Jepson A, Hassan-King M, Whittle H, Olerup O, et al. Human antibody responses to Pfs 230, a sexual stage-specific surface antigen of Plasmodium falciparum: non-responsiveness is a stable phenotype but does not appear to be genetically regulated. Parasite Immunol. 1994;16(2):55–62.
Saeed M, Roeffen W, Alexander N, Drakeley CJ, Targett GAT, Sutherland CJ. Plasmodium falciparum antigens on the surface of the gametocyte-infected erythrocyte. PLoS One. 2008;3(5):e2280. https://doi.org/10.1371/journal.pone.0002280.
Sutherland C. Surface antigens of Plasmodium falciparum gametocytes-a new class of transmission-blocking vaccine targets? Mol Biochem Parasitol. 2009;166(2):93–8. https://doi.org/10.1016/j.molbiopara.2009.03.007.
Stone WJ, et al. Naturally acquired immunity to sexual stage P. falciparum parasites. Parasitology. 2016;143(2):187–98. https://doi.org/10.1017/S0031182015001341.
Bouharoun-Tayoun H, Attanath P, Sabchareon A, Chongsuphajaisiddhi T, Druilhe P. Antibodies that protect humans against Plasmodium falciparum blood stages do not on their own inhibit parasite growth and invasion in vitro, but act in cooperation with monocytes. J Exp Med. 1990;172(6):1633–41. https://doi.org/10.1084/jem.172.6.1633.
Ferrante A, Kumaratilake L, Rzepczyk CM, Dayer JM. Killing of Plasmodium falciparum by cytokine activated effector cells (neutrophils and macrophages). Immunol Lett. 1990;25(1–3):179–87. https://doi.org/10.1016/0165-2478(90)90112-4.
Kumaratilake LM, Ferrante A. Opsonization and phagocytosis of Plasmodium falciparum merozoites measured by flow cytometry. Clin Diagn Lab Immunol. 2000;7(1):9–13.
Ayi K, Patel SN, Serghides L, Smith TG, Kain KC. Nonopsonic phagocytosis of erythrocytes infected with ring-stage Plasmodium falciparum. Infect Immun. 2005;73(4):2559–63. https://doi.org/10.1128/IAI.73.4.2559-2563.2005.
McGilvray ID, Serghides L, Kapus A, Rotstein OD, Kain KC. Nonopsonic monocyte/macrophage phagocytosis of Plasmodium falciparum-parasitized erythrocytes: a role for CD36 in malarial clearance. Blood. 2000;96(9):3231–40.
Sinden R, Smalley M. Gametocytes of Plasmodium falciparum: phagocytosis by leucocytes in vivo and in vitro. Trans R Soc Trop Med Hyg. 1976;70(4):344–5. https://doi.org/10.1016/0035-9203(76)90096-1.
Lensen A, et al. Leukocytes in a Plasmodium falciparum-infected blood meal reduce transmission of malaria to Anopheles mosquitoes. Infect Immun. 1997;65(9):3834–7.
Smith TG, Serghides L, Patel SN, Febbraio M, Silverstein RL, Kain KC. CD36-mediated nonopsonic phagocytosis of erythrocytes infected with stage I and IIA gametocytes of Plasmodium falciparum. Infect Immun. 2003;71(1):393–400. https://doi.org/10.1128/IAI.71.1.393-400.2003.
•• Bansal GP, Weinstein CS, Kumar N. Insight into phagocytosis of mature sexual (gametocyte) stages of Plasmodium falciparum using a human monocyte cell line. Acta Trop. 2016;157:96–101. These studies provide evidence that live mature intraerythrocytic gametocytes are not phagocytosed whereas free or killed gametocytes are. https://doi.org/10.1016/j.actatropica.2016.01.033.
Oldenborg P-A. CD47: a cell surface glycoprotein which regulates multiple functions of hematopoietic cells in health and disease. ISRN Hematology. 2013;2013:614619.
Ayi K, Lu Z, Serghides L, Ho JM, Finney C, Wang JCY, et al. CD47-SIRPα interactions regulate macrophage uptake of Plasmodium falciparum-infected erythrocytes and clearance of malaria in vivo. Infect Immun. 2016;84(7):2002–11. https://doi.org/10.1128/IAI.01426-15.
Banerjee R, Khandelwal S, Kozakai Y, Sahu B, Kumar S. CD47 regulates the phagocytic clearance and replication of the Plasmodium yoelii malaria parasite. Proc Natl Acad Sci U S A. 2015;112(10):3062–7. https://doi.org/10.1073/pnas.1418144112.
Tsai RK, Rodriguez PL, Discher DE. Self inhibition of phagocytosis: the affinity of ‘Marker of Self’ CD47 for SIRPα dictates potency of inhibition but only at low expression levels. Blood Cells Mol Dis. 2010;45(1):67–74. https://doi.org/10.1016/j.bcmd.2010.02.016.
Sosale NG, Ivanovska II, Tsai RK, Swift J, Hsu JW, Alvey CM, et al. “Marker of Self” CD47 on lentiviral vectors decreases macrophage-mediated clearance and increases delivery to SIRPA-expressing lung carcinoma tumors. Molecular Therapy Methods & Clinical Development. 2016;3:16080. https://doi.org/10.1038/mtm.2016.80.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This review article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Parasitology
Rights and permissions
About this article
Cite this article
Bansal, G.P., Kumar, N. Immune Responses in Malaria Transmission. Curr Clin Micro Rpt 5, 38–44 (2018). https://doi.org/10.1007/s40588-018-0078-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40588-018-0078-x