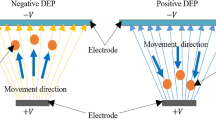
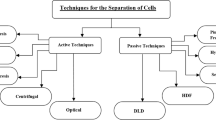
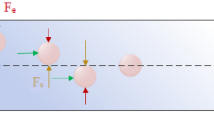
Abstract
Investigating and analyzing circulating tumor cells (CTCs) have shown to be an invaluable tool for early cancer detection and diagnosis. Microfluidic devices, which are inexpensive and simple to use, have recently gained a lot of attention for the enumeration and separation of CTCs. In this research, a novel sheathless double-loop spiral-based lab-on-a-chip is proposed dependent upon the functionality of inertial focusing for separating multiple CTCs such as MCF-7 (breast cancer CTCs) and A549 (lung cancer CTCs) distinctly from the normal cells like WBCs (white blood cells) and RBCs (red blood cells). The chip is designed and examined in numerical simulation using COMSOL Multiphysics 5.4 tool at various average flow velocities and Reynolds numbers (Re). In this study, the separation purities and recoveries of \(\sim \) 100% is gained by the chip at the Re values ranges from 71.75 \({\text{to}}\) 76.87 (flowrate of 87.8\(-\)94.1 ml/h), which indicates the high capability of separating multiple CTCs distinctly with high throughput.










Similar content being viewed by others
Data Availability
All data are included in the manuscript.
References
Lin D, Shen L, Luo M, Zhang K, Li J, Yang Q, Zhu F, Zhou D, Zheng S, Chen Y, Zhou J (2021) Circulating tumor cells: biology and clinical significance. Signal Transduct Target Ther 6(1):1–24
Ozbey A, Karimzadehkhouei M, Kocaturk NM, Bilir SE, Kutlu O, Gozuacik D, Kosar A (2019) Inertial focusing of cancer cell lines in curvilinear microchannels. Micro Nano Eng 2:53–63
He S, Yu S, Wei J, Ding L, Yang X, Wu Y (2022) New horizons in the identification of circulating tumor cells (CTCs): an emerging paradigm shift in cytosensors. Biosens Bioelectron 203:114043
Esteva FJ, Hubbard-Lucey VM, Tang J, Pusztai L (2019) Immunotherapy and targeted therapy combinations in metastatic breast cancer. Lancet Oncol 20(3):e175–e186
Pantel K, Alix-Panabières C (2010) Circulating tumour cells in cancer patients: challenges and perspectives. Trends Mol Med 16(9):398–406
Vignot S, Besse B, André F, Spano JP, Soria JC (2012) Discrepancies between primary tumor and metastasis: a literature review on clinically established biomarkers. Crit Rev Oncol Hematol 84(3):301–313
Sun Y, Haglund TA, Rogers AJ, Ghanim AF, Sethu P (2018) Microfluidics technologies for blood-based cancer liquid biopsies. Anal Chim Acta 1012:10–29
Warkiani ME, Khoo BL, Wu L, Tay AKP, Bhagat AAS, Han J, Lim CT (2016) Ultra-fast, label-free isolation of circulating tumor cells from blood using spiral microfluidics. Nat Protoc 11(1):134–148
Zhou Y, Ma Z, Ai Y (2018) Sheathless inertial cell focusing and sorting with serial reverse wavy channel structures. Microsyst Nanoeng 4(1):1–14
Belotti Y, Lim CT (2021) Microfluidics for liquid biopsies: Recent advances, current challenges, and future directions. Anal Chem 93(11):4727–4738
Sivaramakrishnan M, Kothandan R, Govindarajan DK, Meganathan Y, Kandaswamy K (2020) Active microfluidic systems for cell sorting and separation. Curr Opin Biomed Eng 13:60–68
Bayareh M (2020) An updated review on particle separation in passive microfluidic devices. Chem Eng Process-Process Intensif 153:107984
Xu X, Huang X, Sun J, Wang R, Yao J, Han W, Wei M, Chen J, Guo J, Sun L, Yin M (2021) Recent progress of inertial microfluidic-based cell separation. Analyst 146(23):7070–7086
Zhao Q, Yuan D, Zhang J, Li W (2020) A review of secondary flow in inertial microfluidics. Micromachines 11(5):461
Tang W, Zhu S, Jiang D, Zhu L, Yang J, Xiang N (2020) Channel innovations for inertial microfluidics. Lab Chip 20(19):3485–3502
Kim GY, Han JI, Park JK (2018) Inertial microfluidics-based cell sorting. BioChip J 12(4):257–267
Chung AJ (2019) A minireview on inertial microfluidics fundamentals: Inertial particle focusing and secondary flow. BioChip J 13(1):53–63
Liu N, Petchakup C, Tay HM, Li KHH, Hou HW (2019) Spiral inertial microfluidics for cell separation and biomedical applications. Appl Microfluid Syst Biol Med 7:99–150
Sun J, Li M, Liu C, Zhang Y, Liu D, Liu W, Hu G, Jiang X (2012) Double spiral microchannel for label-free tumor cell separation and enrichment. Lab Chip 12(20):3952–3960
Rzhevskiy AS, Razavi Bazaz S, Ding L, Kapitannikova A, Sayyadi N, Campbell D, Walsh B, Gillatt D, Ebrahimi Warkiani M, Zvyagin AV (2019) Rapid and label-free isolation of tumour cells from the urine of patients with localised prostate cancer using inertial microfluidics. Cancers 12(1):81
Akbarnataj K, Maleki S, Rezaeian M, Haki M, Shamloo A (2023) Novel size-based design of spiral microfluidic devices with elliptic configurations and trapezoidal cross-section for ultra-fast isolation of circulating tumor cells. Talanta 254:124125
Zhu Z, Wu D, Li S, Han Y, Xiang N, Wang C, Ni Z (2021) A polymer-film inertial microfluidic sorter fabricated by jigsaw puzzle method for precise size-based cell separation. Anal Chim Acta 1143:306–314
Omrani V, Targhi MZ, Rahbarizadeh F, Nosrati R (2023) High-throughput isolation of cancer cells in spiral microchannel by changing the direction, magnitude and location of the maximum velocity. Sci Rep 13(1):3213
Ookawara S, Street D, Ogawa K (2006) Numerical study on development of particle concentration profiles in a curved microchannel. Chem Eng Sci 61(11):3714–3724
Bhagat AAS, Kuntaegowdanahalli SS, Papautsky I (2008) Continuous particle separation in spiral microchannels using dean flows and differential migration. Lab Chip 8(11):1906–1914
Kye HG, Park BS, Lee JM, Song MG, Song HG, Ahrberg CD, Chung BG (2019) Dual-neodymium magnet-based microfluidic separation device. Sci Rep 9(1):1–10
Di Carlo D, Irimia D, Tompkins RG, Toner M (2007) Continuous inertial focusing, ordering, and separation of particles in microchannels. Proc Natl Acad Sci 104(48):18892–18897
Amini H, Lee W, Di Carlo D (2014) Inertial microfluidic physics. Lab Chip 14(15):2739–2761
Chen H (2018) A triplet parallelizing spiral microfluidic chip for continuous separation of tumor cells. Sci Rep 8(1):1–8
Abdulla A, Liu W, Gholamipour-Shirazi A, Sun J, Ding X (2018) High-throughput isolation of circulating tumor cells using cascaded inertial focusing microfluidic channel. Anal Chem 90(7):4397–4405
Hou HW, Warkiani ME, Khoo BL, Li ZR, Soo RA, Tan DSW, Lim WT, Han J, Bhagat AAS, Lim CT (2013) Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci Rep 3(1):1–8
Shiriny A, Bayareh M (2021) Inertial focusing of CTCs in a novel spiral microchannel. Chem Eng Sci 229:116102
Shamloo A, Boodaghi M (2018) Design and simulation of a microfluidic device for acoustic cell separation. Ultrasonics 84:234–243
Altay R, Yapici MK, Koşar A (2022) A hybrid spiral microfluidic platform coupled with surface acoustic waves for circulating tumor cell sorting and separation: a numerical study. Biosensors 12(3):171
Acknowledgements
It is appropriate to express gratitude to the Bio-MEMS Laboratory at the NIT Nagaland, Dimapur-797103, for providing the software and their continued supports for the research project.
Funding
The study’s execution received no financing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no declared conflicts of interest between the authors and the subject matter of this paper.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pakhira, W., Kumar, R. & Ibrahimi, K.M. Numerical Simulation of a Sheathless Multi-CTC Separator Lab-on-a-Chip Using Inertial Focusing Method. Comp. Part. Mech. (2024). https://doi.org/10.1007/s40571-024-00770-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40571-024-00770-7