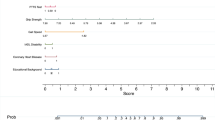
Abstract
Objective
This study sought to develop and validate a 6-year risk prediction model in older adults with cognitive frailty (CF).
Methods
In the secondary analysis of Chinese Longitudinal Healthy Longevity Survey (CLHLS), participants from the 2011–2018 cohort were included to develop the prediction model. The CF was assessed by the Chinese version of Mini-Mental State Exam (CMMSE) and the modified Fried criteria. The stepwise regression was used to select predictors, and the logistic regression analysis was conducted to construct the model. The model was externally validated using the temporal validation method via the 2005–2011 cohort. The discrimination was measured by the area under the curve (AUC), and the calibration was measured by the calibration plot. A nomogram was conducted to vividly present the prediction model.
Results
The development dataset included 2420 participants aged 60 years or above, and 243 participants suffered from CF during a median follow-up period of 6.91 years (interquartile range 5.47–7.10 years). Six predictors, namely, age, sex, residence, body mass index (BMI), exercise, and physical disability, were finally used to develop the model. The model performed well with the AUC of 0.830 and 0.840 in the development and external validation datasets, respectively.
Conclusion
The study could provide a practical tool to identify older adults with a high risk of CF early. Furthermore, targeting modifiable factors could prevent about half of the new-onset CF during a 6-year follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
The global population of older adults was approximately 1 billion in 2019 and will increase to 1.4 billion by 2030 [1]. Ageing contributes to many chronic conditions, such as cognitive impairment and frailty, which have become increasingly significant public health problems [2, 3]. Frailty is influenced by multidomain factors, including age, sex, risk of malnutrition, and chronic diseases, as well as disability [4, 5]. Frailty and cognitive impairment interact in the ageing process, increasing the risk of adverse outcomes [6] such as dementia, disability, and mortality, but they have historically been studied separately. Consequently, the International Consensus Group from the International Academy of Nutrition and Aging (IANA) and the International Association of Gerontology and Geriatrics (IAGG) have proposed that cognitive frailty (CF) is a clinical condition characterized by the occurrence of both physical frailty and cognitive impairment and in the absence of dementia diagnosis [7]. CF may contribute to a higher risk of adverse outcomes than healthy older adults or those with physical frailty or cognitive impairment alone [8,9,10]. A meta-analysis indicated that the pooled prevalence of CF among community-dwelling older adults was 9% [11].
In order to identify individuals at high risk of CF and to facilitate the implementation of appropriate preventive measures and interventions [12,13,14], some prediction models have been developed [15,16,17]. There were some limitations in existed CF prediction models, such as selecting predictors based on univariable analysis and lacking calibration and external validation [15]. For instance, Peng et al. [16] developed and internally validated a prediction model for diagnosing CF in elderly Chinese patients with multimorbidity, incorporating non-traditional factors. The final model included nine predictors, yielding an area under the curve (AUC) of 0.9908. However, the predictors of this model were chosen through univariable analysis, and no external validation was performed, potentially compromising the prediction performance of the final model. The accessibility of predictors was another question. Sargent et al. [17] constructed a prediction model for CF using 32 single nucleotide polymorphisms (SNPs) and 155 protein biomarkers, achieving a model AUC of 0.88. However, the high cost and limited accessibility of predictors restricted the generalizability and applicability of models.
Therefore, we aimed to develop and validate a CF prediction model among older adults using easily obtainable predictors and adhering strictly to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) statement [18]. The study’s findings might be more helpful for primary medical staffs in the early detection of CF.
Methods
This study was a secondary analysis of the Chinese Longitudinal Healthy Longevity Survey (CLHLS) and strictly followed the TRIPOD statement [18]. The detailed information is given in Supplementary Table 1.
Source of data and participants
Participants were chosen from CLHLS, a prospective multicenter cohort study covering about 113,000 older adults in China. Surveys of the CLHLS began in 1998 and were conducted every three years, with eight surveys conducted from 1998 to 2018. We selected the 2011–2018 cohort for model development in this study. The included criteria were the followings: (1) older adults aged 60 years and over; (2) absence of CF at baseline; and (3) at least one follow-up.
Exclusion criteria encompassed individuals with dementia and cancer at baseline. The study excluded individuals with dementia to emphasize the focus on CF, involving reversible cognitive impairment, particularly mild cognitive impairment, as opposed to irreversible dementia [19]. Additionally, the exclusion of individuals diagnosed with cancer at the baseline was aimed at minimizing potential attrition rates over the extended 6-year follow-up duration due to their shorter life expectancy.
Additionally, we used the 2005–2011 cohort of CLHLS for external validation, and the inclusion and exclusion criteria were consistent with the development cohort.
Assessment of cognitive frailty
In this study, CF was defined as the coexistence of cognitive impairment and physical frailty. Cognitive impairment was measured by the Chinese version of Mini-Mental State Exam (CMMSE) [20]. The CMMSE consists of 24 items, scored from 0 to 30, with higher scores indicating better cognitive function. The definition of cognitive impairment was corrected by the educational levels [21, 22]. Participants had been diagnosed cognitive impairment with CMMSE score between 16 to 19 for those illiterate people, 20 to 22 for those with elementary school education, and 23 to 26 for those with a middle school education or higher score.
Physical frailty was evaluated by the modified Fried criteria which consisted of five domains: exhaustion, shrink, weakness, low mobility, and inactivity [23]. Each domain was assessed with a binary response (yes or no). Individuals were categorized as frailty if they reported three or more domains as "yes". Exhaustion was defined if the participants answered “always”, “often” or “sometimes” to the question “I felt old and useless”. Shrink was defined as body mass index (BMI) < 18.5 kg/m2, calculated by dividing the weight by the square of the height. Weakness was defined as the participants’ self-reported inability to lift a bag weighing 5 kg. Low mobility was defined as the participants’ self-reported inability to walk one km. Inactivity was defined if participants reported that they engaged in activities once a week or less. Activities included housework, outside activity, gardening, keeping a pet, livestock breeding, playing cards or mah-jong, and social activity.
Candidate predictors
Candidate predictors were selected based on previous studies [24,25,26,27,28], medical knowledge and data available in the database. A total of 12 candidate predictors were chosen, and the detailed information is shown in Table 1. The physical disability was measured using the instrumental activity of daily living (IADL) and the basic activity of daily living (BADL), and it was defined if any item of IADL or BADL was judged as dependence [29]. Most variables were self-reported by the older adults or their families except BMI.
Sample size
According to the Tool to Assess Risk of Bias and Application of Prediction Model Studies (PROBAST) [30], a minimum of 20 events per variable (EPVs) was suggested for model development and at least 100 participants with the outcome for model validation to minimize overfitting. More than 240 older adults with CF must be in the development cohort, and there should be at least 100 participants with CF in the validation cohort.
Missing data
The missing data were dealt with multivariate imputation by chained equation (MICE) if the missing data in each variable were random and accounted for less than 50% [31]. Five imputations were generated using multiple chains. The dataset with the lowest Akaike Information Criterion (AIC) value was chosen.
Statistical analysis
The continuous data were presented as mean with standard deviation (SD) if the data were normally distributed, verified using histogram and Kolmogorov–Smirnov test. Non-normally distributed data were presented as median with interquartile range (IQR). The categorical data were presented as the number with proportions. BMI was classified into four categories [32]. More details can be found in Supplementary Table 2. The categarical data were analyzed using the chi-square test (for theoretical frequencies T ≥ 5) or continuity correction for theoretical frequencies (for theoretical frequencies T < 5). For continuous data, an independent samples t-test was utilized for normally distributed data; otherwise, the non-parametric Wilcoxon rank-sum test was applied.
For model development, we used stepwise binary logistic regression analysis to select predictors and develop a model with the lowest AIC value. For internal validation, we evaluated the model’s performances by discrimination and calibration. The discrimination, often measured by AUC, was the extent to distinguish those at higher or lower risk of having an event [33]. The discrimination was evaluated as acceptable with AUC of 0.7–0.8, excellent with AUC of 0.8–0.9, and outstanding with AUC of 0.9 [34]. The calibration evaluated the conformity of predicted and actual risks, measured by the calibration plot. The calibration plot represented the predictive probability on the x-axis and the observed probability on the y-axis. A 45-degree line in the calibration plot suggested perfect prediction [35].
To test the robustness of the model, we conducted several sensitivity analysis. First, we excluded the BMI from the model to investigate the effect of incorporation bias. Then, we excluded participants with missing data to explore selection or attrition bias.
Additionally, we calculated the population attributable fraction (PAF) for the proportion of people exposed to risk factors. The formula is as follows:
where the Pi is the population proportion at exposure level i; Pi’ is the counterfactual or ideal level of exposure; RRi is the risk ratio at exposure level i, and n is the number of exposure levels.
All statistical analyses were performed using the R version 4.1.3 software with major packages of mice, MASS, regplot, rms, and pROC.
Internal and external validation
The model was internally validated using the bootstrap resampling for 1000 times method. To check the external validation, we performed the temporal validation using data from the CLHLS 2005–2011 cohort. There was no difference between the development and validation datasets in setting, CF assessment, and predictors.
Results
Participants
The development dataset consisted of 2420 participants, among whom 243 older adults (10.04%) were observed to have CF during a median follow-up period of 6.91 years (IQR: 5.47–7.10 years). In the external validation dataset, a final analysis of 3512 participants was conducted, revealing 404 older adults with CF. The detailed flowchart of the study population is shown in Fig. 1. The characteristics of the participants are shown in Table 1. Compared with those having complete data, individuals with missing data showed no statistical difference in all variables except physical disability (p < 0.001). The characteristics of the participants between those with missing data and complete data are shown in Supplementary Table 3.
Predictors selection and model development
According to the lowest AIC value, the stepwise binary logistic regression analysis was used for predictors selection and model development. Finally, six predictors were included to develop the model (i.e., age, sex, residence, BMI, exercise, and physical disability). The importance of the six predictors was ranked as follows: age, physical disability, BMI, sex, residence, and exercise, according to difference value between full model pseudo-R2 and models with any excluded variable. And the details are shown in the Supplementary Table 4. Table 2 displays the model’s coefficient and odds ratio (OR).
Model performance
The discrimination of CF prediction model was excellent [34], indicated by an AUC of 0.830 (95% CI 0.802–0.858). The model's cut-off point was – 2.271, with a specificity of 0.844 and a sensitivity of 0.907. The calibration plot revealed a high level of consistency between the predicted risk of CF and the observed risk.
In the external validation, the model performed better, with a higher discrimination AUC of 0.840 (95% CI 0.820–0.860) and a well-fitted calibration curve. Figures 2 and 3 display the prediction performance.
Model presentation
The nomogram was employed to illustrate the model, allowing the calculation and summation of points based on participants' characteristics. The total score was calculated by summing the values of each predictor for an older adult. By drawing a line down corresponding to this total score, one could obtain the predicted probability of CF (Supplementary Fig. 1).
Sensitivity analysis
After excluding BMI from the model, the AUC slightly attenuated, with AUC of 0.824 (95% CI 0.795–0.852) (Supplementary Fig. 2). After excluding participants with missing data, the AUC of the model was 0.836 (95% CI 0.786–0.888) (Supplementary Fig. 3), which proved that the model was robust.
Population attributable fraction
Among the six predictors, three were modifiable: BMI, exercise, and physical disability. The results of PAF suggested that targeting the three modifiable predictors would theoretically decrease 53.79% of the 6-year risk of CF.
Discussion
We developed a CF prediction model using multivariable logistic regression with the CLHLS database. Predictors considered in this model encompassed age, sex, residence, exercise, BMI, and physical disability. The internal and external validation results exhibited that the model was with excellent discriminative ability and calibration.
Consistent with other studies [28, 36], aging and female were risk factors of CF, while living in rural areas posed a higher risk than living in the city settings. Aging has been identified as a catalyst for chronic inflammation, and was often correlated with compromised physical functionality and diminished muscle mass. This augmented inflammatory state could have significant ramifications on cognitive function, particularly memory and executive function, thereby elevating the risk of CF [37]. Regarding the gender, the increased risk of CF in women may be related to hormonal factors, compared with men. Studies suggested that the decrease in testosterone and other androgens might be linked to the progression of frailty and cognitive decline [38, 39]. Testosterone plays a protective role in cognition by promoting synaptic plasticity in the hippocampus and regulating the accumulation of amyloid-beta proteins [39]. Additionally, a reduction in testosterone levels can lead to the decline in muscle mass [38]. Rural older residents in mainland China had a higher risk of CF, possibly due to unequal urban–rural resources, lower income, low education levels among rural inhabitants, and limited access to healthcare services and insurance [40].
Our model showed that underweight, physical inactivity and physical disability increased the risk of CF. These findings were consistent with previous studies [41,42,43]. Low BMI and physical inactivity can increase the content of metabolites and inflammatory cells, affecting structure in the brain [44,45,46]. Additionally, previous studies showed that inflammation was significantly associated with poor physical function and muscle strength in older adults, thereby leading to an increased risk of CF [47, 48]. The nutritional and exercise intervention could effectively improve muscle strength and physical function among older adults, thereby reducing the risk of CF [49, 50]. One study showed that participants in the physical activity group had 21% lower odds of worsening CF over 24 months than those in the health education group [51]. Nevertheless, the best doses of type, intensity, and timing of exercise for preventing CF still need to be explored [52]. Despite the lack of significant impact on CF, exercise remained a constituent of the final model. The predictors were computed following the AIC methodology and were informed by scientific insights, underscoring the pivotal role of exercise in CF [12, 52].
Strengths
Our study had some strengths. Firstly, the participants were representative because they were from a large population-based cohort. Secondly, the predictors included were non-invasive, low-cost, and easy to obtain, so the prediction model could be used in the primary care settings [53]. Thirdly, we performed external validation for portability and generalization, and the model displayed excellent discrimination and calibration in external validation. Lastly, we calculated the PAF to explore how modifiable risk factors contribute to CF.
Limitations
Our study also had some limitations. Firstly, most predictors were self-reported by older adults, potentially introducing information bias. Nevertheless, self-reported predictors were more easy-to-obtain and practical [54]. Secondly, some critical predictors were not included due to data limitations, such as depression [28], likely impacting the prediction performance. Thirdly, BMI, a part of the CF assessment, was selected as a predictor, which might introduce incorporation bias and optimist estimates of model performance [55]. However, the sensitivity analysis indicated that after removing the BMI, the prediction model maintained excellent performance. Therefore, the incorporation bias may have a negligible effect on the model performance.
Implications and clinical practice
This study presents some insights for future research. Firstly, the selection of predictive factors, encompassing easily accessible, non-invasive, and cost-effective variables, plays a pivotal role in prediction models applicable to clinical practice, especially within community healthcare and diverse clinical settings. Subsequent research endeavors should consider integrating addition predictive factors that share the accessibility, non-invasiveness, and cost-effectiveness criteria. Secondly, our study is grounded in the application of the logistic regression method for model development. Future research could explore alternative methodologies, such as machine learning techniques, to foster the evolution of predictive models. Lastly, the model's foundation is rooted in the Chinese population, prompting the necessity to examine its transferability to other demographics. This calls for comprehensive validation in diverse populations to establish its broader applicability in the future.
This study developed a prediction model for CF based on the characteristics of the Chinese population, utilizing practical, non-invasive, cost-effective, and easily obtainable variables. It can be applied in secondary prevention, enabling early identification, diagnosis, and treatment of CF. In tertiary disease prevention, the predictive model can be used to forecast recurrence, reduce mortality and disability [56]. Furthermore, it can provide community staffs with insights into the progression of CF in the older adults, allowing the identification of potential contributing factors for tailored preventive interventions [54].
Conclusion
The CF prediction model, following the TRIPOD statement, has been established and validated for older adults. It integrates six easily obtainable predictors and demonstrates excellent prediction performance. This model helps healthcare practitioners and nurses to identify older adults at a heightened risk of CF development over a six-year period and intervene proactively.
Data availability
The CLHLS data can be obtained by applying on the website. https://opendata.pku.edu.cn/dataset.xhtml?persistentId=doi:10.18170/DVN/WBO7LK&version=2.0.
References
World Health Organization (2018) Ageing. https://www.who.int/news-room/facts-in-pictures/detail/ageing. Accessed 28 Feb 2018
Dent E, Lien C, Lim WS et al (2017) The Asia-Pacific clinical practice guidelines for the management of frailty. J Am Med Dir Assoc 18:564–575. https://doi.org/10.1016/j.jamda.2017.04.018
Yu R, Wong M, Chong KC et al (2018) Trajectories of frailty among Chinese older people in Hong Kong between 2001 and 2012: an age-period-cohort analysis. Age Ageing 47:254–261. https://doi.org/10.1093/ageing/afx170
Qin Y, Hao X, Lv M et al (2023) A global perspective on risk factors for frailty in community-dwelling older adults: a systematic review and meta-analysis. Arch Gerontol Geriatr 105:104844. https://doi.org/10.1016/j.archger.2022.104844
He B, Ma Y, Wang C et al (2019) Prevalence and risk factors for frailty among community-dwelling older people in china: a systematic review and meta-analysis. J Nutr Health Aging 23:442–450. https://doi.org/10.1007/s12603-019-1179-9
Lee Y, Kim J, Chon D et al (2018) The effects of frailty and cognitive impairment on 3-year mortality in older adults. Maturitas 107:50–55. https://doi.org/10.1016/j.maturitas.2017.10.006
Kelaiditi E, Cesari M, Canevelli M et al (2013) Cognitive frailty: rational and definition from an (i.A.N.A./i.A.G.G.) international consensus group. J Nutr Health Aging 17:726–734. https://doi.org/10.1007/s12603-013-0367-2
Chen C, Park J, Wu C et al (2020) Cognitive frailty in relation to adverse health outcomes independent of multimorbidity: results from the china health and retirement longitudinal study. Aging (Albany NY) 12:23129–23145. https://doi.org/10.18632/aging.104078
Shimada H, Doi T, Lee S et al (2018) Cognitive frailty predicts incident dementia among community-dwelling older people. J Clin Med. https://doi.org/10.3390/jcm7090250
Zhang XM, Jiao J, Zhu C et al (2021) Cognitive frailty and 30-day mortality in a national cohort of older Chinese inpatients. Clin Interv Aging 16:389–401. https://doi.org/10.2147/cia.S294106
Qiu Y, Li G, Wang X et al (2022) Prevalence of cognitive frailty among community-dwelling older adults: a systematic review and meta-analysis. Int J Nurs Stud 125:104112. https://doi.org/10.1016/j.ijnurstu.2021.104112
Merchant RA, Chan YH, Hui RJY et al (2021) Motoric cognitive risk syndrome, physio-cognitive decline syndrome, cognitive frailty and reversibility with dual-task exercise. Exp Gerontol 150:111362. https://doi.org/10.1016/j.exger.2021.111362
Ruan Q, Yu Z, Chen M et al (2015) Cognitive frailty, a novel target for the prevention of elderly dependency. Ageing Res Rev 20:1–10. https://doi.org/10.1016/j.arr.2014.12.004
Hoogendijk EO, Afilalo J, Ensrud KE et al (2019) Frailty: implications for clinical practice and public health. Lancet 394:1365–1375. https://doi.org/10.1016/s0140-6736(19)31786-6
Huang J, Zeng X, Hu M et al (2023) Prediction model for cognitive frailty in older adults: A systematic review and critical appraisal. Front Aging Neurosci 15:1119194. https://doi.org/10.3389/fnagi.2023.1119194
Peng S, Zhou J, Xiong S et al (2023) Construction and validation of cognitive frailty risk prediction model for elderly patients with multimorbidity in Chinese community based on non-traditional factors. BMC Psychiatry 23:266. https://doi.org/10.1186/s12888-023-04736-6
Sargent L, Nalls M, Amella EJ et al (2020) Shared mechanisms for cognitive impairment and physical frailty: a model for complex systems. Alzheimers Dement (N Y) 6:e12027. https://doi.org/10.1002/trc2.12027
Collins GS, Reitsma JB, Altman DG et al (2015) Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (tripod): the tripod statement. BMJ 350:g7594. https://doi.org/10.1136/bmj.g7594
Zhang XX, Tian Y, Wang ZT et al (2021) The epidemiology of Alzheimer’s disease modifiable risk factors and prevention. J Prev Alzheimers Dis 8:313–321. https://doi.org/10.14283/jpad.2021.15
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198. https://doi.org/10.1016/0022-3956(75)90026-6
Luo G, Han J, QQ et al (2002) The suitable application the mmse on old people in urban and rural areas [Chinese]. Chin J Clin Psychol 01:10–13
Yang Z, Holt HK, Fan J-H et al (2016) Optimal cutoff scores for alzheimer’s disease using the chinese version of mini-mental state examination among chinese population living in rural areas. Am J Alzheimers Dis Other Demen 31:650–657. https://doi.org/10.1177/1533317516662336
Wang HY, Lv X, Du J et al (2021) Age- and gender-specific prevalence of frailty and its outcomes in the longevous population: the Chinese longitudinal healthy longevity study. Front Med (Lausanne) 8:719806. https://doi.org/10.3389/fmed.2021.719806
Chen Y, Zhang Z, Zuo Q et al (2022) Construction and validation of a prediction model for the risk of cognitive frailty among the elderly in a community [Chinese]. Chin J Nurs 57:197–203. https://doi.org/10.3761/j.issn.0254-1769.2022.02.012
Tseng SH, Liu LK, Peng LN et al (2019) Development and validation of a tool to screen for cognitive frailty among community-dwelling elders. J Nutr Health Aging 23:904–909. https://doi.org/10.1007/s12603-019-1235-5
Wen F, Chen M, Zhao C et al (2021) Development of a cognitive frailty prediction model for elderly patients with stable coronary artery disease [Chinese]. J Nurs Sci 36:21–26. https://doi.org/10.3870/j.issn.1001-4152.2021.10.021
Yang Z, Zhang H (2021) A nomogram for predicting the risk of cognitive frailty in community-dwelling elderly people with chronic diseases [Chinese]. J Nurs Sci 36:86–89. https://doi.org/10.3870/j.issn.1001-4125.2021.12.086
Zhang T, Ren Y, Shen P et al (2021) Prevalence and associated risk factors of cognitive frailty: a systematic review and meta-analysis. Front Aging Neurosci 13:755926. https://doi.org/10.3389/fnagi.2021.755926
Zhang Y, Xiong Y, Yu Q et al (2021) The activity of daily living (adl) subgroups and health impairment among Chinese elderly: a latent profile analysis. BMC Geriatr 21:30. https://doi.org/10.1186/s12877-020-01986-x
Wolff RF, Moons KGM, Riley RD et al (2019) Probast: a tool to assess the risk of bias and applicability of prediction model studies. Ann Intern Med 170:51–58. https://doi.org/10.7326/m18-1376
Steyerberg EW (2009) Clinical prediction models: a practical approach to development, validation, and updating. Springer, Cham
Gao M, Wei YX, Lyu J et al (2019) the cut-off points of body mass index and waist circumference for predicting metabolic risk factors in Chinese adults. Zhonghua Liu Xing Bing Xue Za Zhi 40:1533–1540. https://doi.org/10.3760/cma.j.issn.0254-6450.2019.12.006
Alba AC, Agoritsas T, Walsh M et al (2017) Discrimination and calibration of clinical prediction models: Users’ guides to the medical literature. JAMA 318:1377–1384. https://doi.org/10.1001/jama.2017.12126
Mandrekar JN (2010) Receiver operating characteristic curve in diagnostic test assessment. J Thorac Oncol 5:1315–1316. https://doi.org/10.1097/JTO.0b013e3181ec173d
Steyerberg EW, Vickers AJ, Cook NR et al (2010) Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology 21:128–138. https://doi.org/10.1097/EDE.0b013e3181c30fb2
Vatanabe IP, Pedroso RV, Teles RHG et al (2022) A systematic review and meta-analysis on cognitive frailty in community-dwelling older adults: risk and associated factors. Aging Ment Health 26:464–476. https://doi.org/10.1080/13607863.2021.1884844
Robertson DA, Savva GM, Kenny RA (2013) Frailty and cognitive impairment—a review of the evidence and causal mechanisms. Ageing Res Rev 12:840–851. https://doi.org/10.1016/j.arr.2013.06.004
Muller M, Grobbee DE, Thijssen JH et al (2003) Sex hormones and male health: effects on components of the frailty syndrome. Trends Endocrinol Metab 14:289–296. https://doi.org/10.1016/s1043-2760(03)00083-3
Maggio M, Dall’Aglio E, Lauretani F et al (2012) The hormonal pathway to cognitive impairment in older men. J Nutr Health Aging 16:40–54. https://doi.org/10.1007/s12603-012-0002-7
Ma L, Tang Z, Zhang L et al (2018) Prevalence of frailty and associated factors in the community-dwelling population of china. J Am Geriatr Soc 66:559–564. https://doi.org/10.1111/jgs.15214
Shimada H, Makizako H, Lee S et al (2016) Impact of cognitive frailty on daily activities in older persons. J Nutr Health Aging 20:729–735. https://doi.org/10.1007/s12603-016-0685-2
Liu H, Jiao J, Zhu C et al (2020) Potential associated factors of functional disability in Chinese older inpatients: a multicenter cross-sectional study. BMC Geriatr 20:319. https://doi.org/10.1186/s12877-020-01738-x
Rietman ML, van der AD, van Oostrom SH et al (2018) The association between bmi and different frailty domains: a u-shaped curve? J Nutr Health Aging 22:8–15. https://doi.org/10.1007/s12603-016-0854-3
Deng YT, Li YZ, Huang SY et al (2022) Association of life course adiposity with risk of incident dementia: A prospective cohort study of 322,336 participants. Mol Psychiatry 27:3385–3395. https://doi.org/10.1038/s41380-022-01604-9
Frodermann V, Rohde D, Courties G et al (2019) Exercise reduces inflammatory cell production and cardiovascular inflammation via instruction of hematopoietic progenitor cells. Nat Med 25:1761–1771. https://doi.org/10.1038/s41591-019-0633-x
Alessa HB, Chomistek AK, Hankinson SE et al (2017) Objective measures of physical activity and cardiometabolic and endocrine biomarkers. Med Sci Sports Exerc 49:1817–1825. https://doi.org/10.1249/mss.0000000000001287
Miller SL, Wolfe RR (2008) The danger of weight loss in the elderly. J Nutr Health Aging 12:487–491. https://doi.org/10.1007/bf02982710
Cesari M, Penninx BW, Pahor M et al (2004) Inflammatory markers and physical performance in older persons: the inchianti study. J Gerontol A Biol Sci Med Sci 59:242–248. https://doi.org/10.1093/gerona/59.3.m242
Luger E, Dorner TE, Haider S et al (2016) Effects of a home-based and volunteer-administered physical training, nutritional, and social support program on malnutrition and frailty in older persons: a randomized controlled trial. J Am Med Dir Assoc 17:671.e679-671.e616. https://doi.org/10.1016/j.jamda.2016.04.018
Hsu KJ, Liao CD, Tsai MW et al (2019) Effects of exercise and nutritional intervention on body composition, metabolic health, and physical performance in adults with sarcopenic obesity: a meta-analysis. Nutrients. https://doi.org/10.3390/nu11092163
Liu Z, Hsu FC, Trombetti A et al (2018) Effect of 24-month physical activity on cognitive frailty and the role of inflammation: the life randomized clinical trial. BMC Med 16:185. https://doi.org/10.1186/s12916-018-1174-8
Li X, Zhang Y, Tian Y et al (2022) Exercise interventions for older people with cognitive frailty-a scoping review. BMC Geriatr 22:721. https://doi.org/10.1186/s12877-022-03370-3
Zhou ZR, Wang WW, Li Y et al (2019) In-depth mining of clinical data: the construction of clinical prediction model with r. Ann Transl Med 7:796. https://doi.org/10.21037/atm.2019.08.63
Hu M, Shu X, Yu G et al (2021) A risk prediction model based on machine learning for cognitive impairment among Chinese community-dwelling elderly people with normal cognition: development and validation study. J Med Internet Res 23:e20298. https://doi.org/10.2196/20298
Moons KGM, Wolff RF, Riley RD et al (2019) Probast: a tool to assess risk of bias and applicability of prediction model studies: explanation and elaboration. Ann Intern Med 170:W1-w33. https://doi.org/10.7326/m18-1377
Langa KM, Levine DA (2014) The diagnosis and management of mild cognitive impairment: A clinical review. JAMA 312:2551–2561. https://doi.org/10.1001/jama.2014.13806
Acknowledgements
The authors thank the work and contribution of all members of the CLHLS.
Funding
This study was supported by the National Natural Science Foundation of China (Grant Numbers: 72374224, 72174212), National Key R&D Program of China (Grant number 2020YFC2008602, 2020YFC2008603), Natural Science Foundation of Hunan province (2023JJ30704), the Special Funding for the Construction of Innovative Provinces in Hunan (2020SK2055), and the Changsha County Science and Technology Plan Project (2022037-2).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The CLHLS study was approved by the research ethics committees of Duke University and Peking University (IRB00001052-13074).
Informed consent
For this type of study, consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, J., Zeng, X., Ning, H. et al. Development and validation of prediction model for older adults with cognitive frailty. Aging Clin Exp Res 36, 8 (2024). https://doi.org/10.1007/s40520-023-02647-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40520-023-02647-w