Abstract
Background
Lifestyle habits have a key role in cardiometabolic health. The effects of combined aerobic training (AT) and high protein intake (HPI) on cardiometabolic parameters in older adults are not well established.
Aims
To investigate the association of AT and HPI with blood pressure (BP), blood glucose, and total blood cholesterol levels in a sample of Italian older adults enrolled in the Longevity Check-up 7 + (Lookup 7 +) study.
Methods
Lookup 7 + is an ongoing project started in June 2015 and conducted in unconventional settings (e.g., exhibitions, malls, health promotion campaigns) across Italy with the aim of fostering adoption of healthy lifestyles in the general population. For the present investigation, analyses were conducted in participants 65 + years and with body mass index values ≥ 18.5 kg/m2 (n = 3219). Systolic (SBP) and diastolic BP (DBP), blood glucose, and total blood cholesterol were measured. Protein intake was estimated using a 12-item food frequency questionnaire. HPI was operationalized as a daily protein intake ≥ 0.8 g/kg of body weight. AT was operationalized as the practice of running and/or swimming for 60 + minutes at least twice weekly during the previous year.
Results
The mean age of the 3219 participants was 72.7 ± 5.7 years, and 55.2% were women. Adherence to AT combined with a HPI was negatively and independently associated with SPB (β: − 4.976; 95% confidence interval: − 9.8 to − 0.08). No other significant associations were observed.
Discussion and conclusions
Our results indicate that AT combined with HPI was negatively associated with SBP in a large and relatively unselected sample of Italian older adults living in the community. These findings need confirmation by ad hoc designed studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Cardiovascular disease (CVD) is the leading cause of death around the world, killing almost 20 million people every year [1]. Older adults are highly affected by CVD, with ~ 70% of those 65 years and older showing some cardiometabolic risk factors [2, 3]. CVD burden in older adults is concerning due to its association with hospitalizations, healthcare costs, and mortality [2, 3]. The development of CVD in advanced age occurs as a consequence of age-related dysregulations of cardiometabolic homeostasis [4], encompassing high blood pressure (BP), abnormal glucose metabolism, and hyperlipidemia.
Physical activity and nutrition are two major modifiable lifestyle factors that may be harnessed for the prevention and treatment of cardiometabolic diseases [5,6,7,8,9]. Numerous studies have reported that aerobic training (AT), land- or water-based exercises in which muscle contractions are sustained for long periods [10], ameliorates cardiometabolic risk factors [11,12,13,14]. This evidence led experts in the field to recommend exercise training as part of non-pharmacological treatment for cardiometabolic diseases [5, 6, 9]. However, the effectiveness of AT is not supported by all studies [11,12,13,14], and more investigations are required.
The adherence to some dietary patterns also brings health benefits to the cardiovascular system. Besides carbohydrates and fats, dietary protein affects cardiometabolic parameters. However, the effects of protein intake vary according to their amino acid composition [15,16,17] and the simultaneous consumption of other macronutrients [18]. Indeed, conflicting results have been reported, with studies showing positive, negative, and null relationships between protein consumption and cardiometabolic parameters [19,20,21,22,23,24,25,26,27,28,29,30].
A few investigations tested the effects of exercise training combined with a high protein intake (HPI) on cardiometabolic risk factors. Amamou et al. [31] reported that a 16-week dynamic resistance training program combined with HPI did not produce additional reductions in cardiometabolic risk factors than HPI alone. However, the study involved a small sample size and did not include an exercise control group.
Randomized clinical trials are expensive and time-consuming, and new interventions are tested when evidence supports the investment and allows the design of the best approach. Currently, there is not sufficient information to support the conduction of a randomized clinical trial to test the effects of exercise combined with HPI on cardiometabolic health. On the other hand, observational studies allow examination of numerous covariates and mixed samples of people with different conditions. Such an approach is an excellent method to collect information for designing clinical trials.
Aims
The present study was conducted to investigate the association between AT and HPI with BP, blood glucose levels, and total blood cholesterol concentrations in a large sample of Italian older adults living in the community.
Methods
Data of the present investigation were gathered from the Longevity Check-up 7 + (Lookup 7 +) project database. Sampling characteristics, procedures, and other results have been published elsewhere [32,33,34,35,36,37,38,39]. Recruitment was conducted among people visiting public spaces (e.g., exhibitions, shopping centers) and those adhering to prevention campaigns promoted by our institution [34]. The Lookup 7 + protocol was approved by the Ethics Committee of the Università Cattolica del Sacro Cuore (protocol #: A.1220/CE/2011) and each participant provided written informed consent prior to enrolment. The manuscript was prepared in compliance with the STrengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines for observational studies [40].
Participants
From 1 June 2015 to 31 October 2021, 13,515 community-dwelling adults aged 18 + years were enrolled. Exclusion criteria were inability or unwillingness to provide written informed consent, self-reported pregnancy, and inability to perform the physical function tests. For the present investigation, analyses were conducted in participants aged 65 + years, with body mass index (BMI) values ≥ 18.5 kg/m2, and no missing data for the variables of interest, totaling 3219 participants (10,296 excluded).
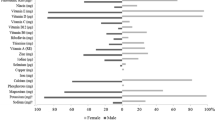
All participants received a structured questionnaire to collect information on personal characteristics and lifestyle habits, followed by measurement of anthropometric parameters. The BMI was calculated as the ratio between body weight (BW, kg) and the square of height (m2). An oscillometric monitor was used to measure BP (Omron M6 electronic sphygmomanometer, Omron, Kyoto, Japan) [41]. Blood concentrations of glucose and total cholesterol were measured from capillary blood samples using disposable electrode strips based on a reflectometric system with a portable device (MultiCare-In, Biomedical Systems International Srl, Florence, Italy) [42]. Participants were asked if they were fasting for at least 8 h. A food frequency questionnaire (FFQ) was used to collect information on how often participants consumed a standardized portion size of a list of 12 foods (meat, meat derivatives, fish, eggs, milk, cheese, yogurt, pasta, bread, rice, vegetables, cereals) on a weekly basis [42]. The mean daily intake of protein was calculated by multiplying the consumption frequency of a food item by the protein content of its standard portion [42], then dividing by seven (the days of a week), followed by the sum of all applicable items. HPI was defined as a protein consumption equal or greater than the recommended dietary allowance (RDA) (≥ 0.8 g/kg of BW per day). Physically inactive (PI) and exercise groups were operationalized based on self-report. Those who did not practice ≥ 60 min at least twice weekly during the previous year were defined PI. Engagement in AT was operationalized as involvement in running and/or swimming activities [38]. The interview did not include questions about exercise frequency, volume, intensity, or length of engagement in AT. Smoking status was categorized as active smoker (has smoked 100 + cigarettes in lifetime and currently smokes cigarettes) and no current smoker.
Statistical analysis
The normal distribution of variables was ascertained via the Shapiro–Wilk test. Continuous variables are expressed as the mean ± standard deviation (SD) or absolute numbers, percentages. Regression analysis was conducted to test the association between AT with or without HPI and cardiometabolic risk factors. PI was used as the reference group. The final model was adjusted for age, sex, BMI, energy intake, sodium, potassium, calcium, magnesium, active smoking, fasting state (blood glucose), and antihypertensive (for systolic and diastolic BP), cholesterol-lowering (for total blood cholesterol), and antidiabetic (for blood glucose) drugs. Significance was set at 5% (p < 0.05) for all tests. Significant results on regression analyses were expected to include a 95% confidence interval (CI 95%) that did not encompass the value of 1. All analyses were performed using the SPSS software (version 23.0, SPSS Inc., Chicago, IL, USA).
Results
Three-thousand two-hundred nineteen older adults of the Lookup 7 + database met the eligibility criteria to be included in the present study. The main characteristics of study participants according to adherence to AT and protein intake groups are shown in Table 1. Personal and anthropometric characteristics of participants 65 + years who were not included in the analyses (n = 206) did not significantly differ from those analyzed. Participants engaged in AT were younger than those in the PI group, whereas individuals with HPI were older than those who only practiced AT. HPI groups had greater absolute and adjusted protein consumption than PI and AT. Similar results were observed across groups for energy, calcium, magnesium, sodium, and potassium intake. Lower diastolic PB values and higher cholesterol levels were found in participants in the HPI group relative to PI. Significant differences in the frequency of women and participants on antihypertensive and cholesterol-lowering treatments were observed across groups.
Results of the linear regression for the association of adherence to AT with or without HPI and BP, blood glucose, and total blood cholesterol are shown in Tables 2, 3, 4, respectively. In the fully adjusted model, the practice of AT combined with a HPI was negatively associated with systolic BP values. No other significant associations were found.
Discussion
Findings of the present study indicate that adherence to AT alone was not associated with cardiometabolic benefits in a sample of community-dwelling Italian older adults enrolled in Lookup 7 + . However, lower systolic BP values were observed in those who engaged AT and had a HPI relative to the PI group. No significant associations were observed between AT + HPI and blood glucose or total blood cholesterol levels.
To the best of our knowledge, this is the first study that investigated the association between AT combined with HPI and cardiometabolic risk factors. A few investigations have been conducted on the subject. Amamou et al. [31] did not observe additional benefits on glucose, triglycerides, total cholesterol, or BP levels in older adults when a high-protein diet was combined with a 16 week dynamic resistance training program. In a study testing the acute effects of protein intake on cardiovascular responses to exercise tests, Bergia et al. [43] found that oral supplementation with 30 g of protein did not affect BP or nitric oxide bioavailability. Similar results were found in a randomized crossover trial testing the acute effects of a protein-rich drink in exercising young men [44].
The chronic effects of walking/running [45] and aquatic activities [46] on cardiometabolic parameters have been widely tested and meta-analyzed [11,12,13,14, 47]. Overall, results from a rigorous pooled analysis indicate that land- and water-based aerobic exercises may significantly reduce BP in older adults with different characteristics [48, 49]. Beneficial effects have also been observed on blood glucose and cholesterol levels [11,12,13,14, 50].
Differences between the results of the present study and the literature might be explained by exercise modalities and participant characteristics. Amelioration of cardiometabolic risk factors in response to exercise training is not easily achieved, and specific combinations of exercise variables might be required [51, 52]. Indeed, the effects of AT are more likely to be observed in women who perform moderate-to-high intensity programs at least 3 days a week for no less than 12 weeks [11]. The exercise volume (i.e., amount of activity) may also impact cardiometabolic parameters, with high-volume AT yielding larger reductions in blood cholesterol and glucose levels than moderate-volume AT [53] or high-intensity interval training [54]. Significant reductions in BP after swimming activities are observed when this exercise encompasses an endurance component, intermittent exercises, or specific swimming techniques (e.g., breaststroke kick) and is performed by hypertensive people [46]. Benefits of swimming on blood cholesterol are only observed for endurance-based activities and in young and middle-aged adults or those with dyslipidemia [50].
Based on these observations, it may be hypothesized that AT sessions of Lookup 7 + participants were not performed with sufficient frequency, intensity, or volume to elicit substantial cardiometabolic benefits. This view is supported by the lack of significant associations in the unadjusted analysis. However, exercise variables were not controlled for in the present study, which impedes from drawing solid conclusions. An increased access to adapted public spaces, the availability of inexpensive and easy-to-use equipment [55, 56], and the popularity of social media influencers [51] induce a growing number of individuals to exercise autonomously. However, supervised exercise may be necessary to elicit favorable adaptations [52].
The association between protein intake and cardiometabolic parameters has been widely explored. In relation to BP, investigations have produced conflicting results, with studies reporting positive, negative, or null relationships [19,20,21,22,23,24,25,26,27,28,29,30]. Protein intake was not associated with changes in BP or the incidence of hypertension in Dutch adults [25, 26]. Liu et al. [28] expanded these findings by reporting no associations of protein intake with the prevalence of hypertension in poorly nourished rural Chinese adults. Increased protein intake also failed to ameliorate hemodynamic parameters in randomized clinical trials [57]. In contrast, large cohort studies reported a negative association between protein intake and BP levels [19, 20, 23, 29, 57, 58].
Studies have also reported no associations between protein intake and blood lipid profile [22, 57]. Kohansal et al. [22] found no associations between protein intake and blood cholesterol levels in Iranian adults. Similar findings were reported by Tayyem et al. [57] in Jordanian adults. These divergent results might be due to protein consumption levels and amino acid composition.
Observation-based models have been proposed to explain the possible effects of protein intake on BP levels [59, 60]. Accordingly, individuals with HPI are expected to display low BP values, whereas those with moderate-to-low consumption might present normal or even high BP levels [59, 60]. Noticeably, participants of the present study reported a limited protein ingestion as shown by the small number of individuals in the HPI group and the low median protein intake in the whole sample (0.55 g/kg of BW). These observations suggest that, in our study population, the intake of protein might not be sufficient to lower BP.
The amino acid composition of dietary protein may have an influence on blood glucose and cholesterol levels. Hepatocytes incubated with homocysteine show increased cholesterol synthesis [61], and homocysteine intake is associated with greater cholesterol concentrations in the blood and liver [61, 62]. Rats fed a cystine-enriched diet displayed elevated liver, plasma, and serum cholesterol levels than controls [63, 64]. In contrast, taurine supplementation reduces total and low-density lipoprotein-cholesterol in rats on a high-cholesterol diet [65] and humans [66]. Similarly, leucine reduces systemic and liver cholesterol concentration in rats [67]. Hence, it is possible that participants of the present study might consume dietary protein whose amino acid composition was inadequate to ameliorate cardiometabolic parameters.
A question that remains is why the combination of AT and HPI was associated with low systolic BP, while either of them alone was not. A possible explanation is that the association of AT and HPI might improve endothelium-dependent vasodilation and increase blood flow and nutrient supply to muscle [68]. The greater metabolic demands of exercising muscles stimulate vasodilation and increase blood flow through a cascade of events that aims at matching the nutrient and oxygen supply to maintain physical work [69]. A repeated exposure to such an adaptation may eventually induce structural and functional alterations of the vascular bed that attenuate or even prevent the age-related decline in endothelium-dependent vasodilation [69, 70]. These changes in vascular structure and function might favor the vasodilating effects of dietary amino acids, such as l-arginine [71, 72]. Exercise may also reduce the concentration of l-arginine competitors for nitric oxide production [73, 74]. An alternative explanation is that amino acid ingestion, mainly branched-chain amino acids [75] combined with adequate AT designs [76, 77], may promote muscle hypertrophy. Indeed, community-dwelling older adults with low muscle mass frequently show elevated BP values and a high prevalence of hypertension [78, 79].
Although reporting novel findings, the present study is not free of limitations. First, our sample was exclusively composed of relatively young community-dwelling Caucasian older adults with well-controlled BP levels, and extrapolations to individuals of other ethnicities or with different age or BP values should be made with caution. In particular, ethnicity and hypertensive status may influence BP responses to exercise [49]. Second, studies have indicated that combined exercise protocols (aerobic plus resistance) produce greater improvements in cardiometabolic profile than individual exercise modalities [12, 13, 80]. Third, the FFQ used in the present study only included the weekly consumption of 12 food groups. This might explain the low protein intake observed in our study participants. Fourth, a meta-analysis found that high-protein diets did not affect fasting blood glucose but decreased HbA1C levels [81]. Hence, future studies should include more complex metabolic analyses to provide a deeper understanding of adaptations induced by dietary and exercise regimens. Fifth, participants were evaluated while they were attending events, in which they had to stand up and walk. Thus, we cannot exclude that the evaluation setting could have influenced BP values. Sixth, the presence of orthostatic hypotension [82] was not explored, and its impact on study results might not be ruled out. Seventh, participants with cardiovascular or cerebrovascular disease were not excluded. Eighth, data on physical activity habits were collected through self-report using simple questions embedded in the Lookup 7 + lifestyle questionnaire [38], instead of established tools for evaluation of physical activity in older adults (e.g., PASE, CHAMP, PAQE). Exercise variables (e.g., frequency, volume, intensity, length of engagement) were not collected or controlled for. Furthermore, subjective measures of physical activity may lead to over- or underestimation owing to inaccurate recall, misinterpretation, and response bias [83]. Limitations concerning physical activity and exercise assessment, albeit not negligible, are intrinsic to the design of Lookup 7 + and the unconventional settings where the survey is conducted. The collection of detailed information on lifestyle habits would substantially increase the duration of the assessments, which may deter many candidates from participating. Finally, the cross-sectional design of the study does not allow inference to be drawn on the time course of changes in the variables considered and on cause-effect relationships.
Conclusion
Findings of the present study indicate that AT combined with HPI is negatively associated with systolic BP in a large and relatively unselected sample of community-dwelling Italian older adults. These results, albeit promising, need to be confirmed by ad hoc designed studies using more established tools for assessing physical activity habits in older adults. Further research is also warranted to explore the effects of interventions involving AT with HPI in older adults with a high cardiometabolic risk profile.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
World Health Organization. (2021) World Health Organization — Cardiovascular diseases (CVDs)
Yazdanyar A, Newman AB (2009) The burden of cardiovascular disease in the elderly: morbidity, mortality, and costs. Clin Geriatr Med. https://doi.org/10.1016/j.cger.2009.07.007
Rodgers JL, Jones J, Bolleddu SI et al (2019) Cardiovascular risks associated with gender and aging. J Cardiovasc Dev Dis. https://doi.org/10.3390/jcdd6020019
Sinclair AJ, Abdelhafiz AH (2020) Cardiometabolic disease in the older person: prediction and prevention for the generalist physician. Cardiovasc Endocrinol Metab 9:90–95. https://doi.org/10.1097/XCE.0000000000000193
Garber CE, Blissmer B, Deschenes MR et al (2011) American college of sports medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc 43:1334–1359. https://doi.org/10.1249/MSS.0b013e318213fefb
Pescatello LS, MacDonald HV, Lamberti L et al (2015) Exercise for hypertension: a prescription update integrating existing recommendations with emerging research. Curr Hypertens Rep. https://doi.org/10.1007/s11906-015-0600-y
Martín-Peláez S, Fito M, Castaner O (2020) Mediterranean diet effects on type 2 diabetes prevention, disease progression, and related mechanisms. Rev Nutr 12:1–15
Sofi F, Macchi C, Abbate R et al (2013) Mediterranean diet and health status: an updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr 17:2769–2782
Colberg SR, Albright AL, Blissmer BJ et al (2010) Exercise and type 2 diabetes: American college of sports medicine and the American diabetes association: joint position statement. Exercise and type 2 diabetes. Med Sci Sports Exerc 42:2282–2303. https://doi.org/10.1249/MSS.0b013e3181eeb61c
Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA et al (2009) Exercise and physical activity for older adults. Med Sci Sports Exerc 41:1510–1530
Wewege MA, Thom JM, Rye K-A et al (2018) Aerobic, resistance or combined training: a systematic review and meta-analysis of exercise to reduce cardiovascular risk in adults with metabolic syndrome. Atherosclerosis 274:162–171. https://doi.org/10.1016/j.atherosclerosis.2018.05.002
Jamka M, Makarewicz-Bukowska A, Bokayeva K et al (2022) Comparison of the effect of endurance, strength and endurance-strength training on glucose and insulin homeostasis and the lipid profile of overweight and obese subjects: a systematic review and meta-analysis. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph192214928
Batrakoulis A, Jamurtas AZ, Metsios GS et al (2022) Comparative efficacy of 5 exercise types on cardiometabolic health in overweight and obese adults: a systematic review and network meta-analysis of 81 randomized controlled trials. Circ Cardiovasc Qual Outcomes 15:e008243. https://doi.org/10.1161/CIRCOUTCOMES.121.008243
Kelley GA, Kelley KS, Tran ZV (2005) Exercise, lipids, and lipoproteins in older adults: a meta-analysis. Prev Cardiol 8:206–214. https://doi.org/10.1111/j.0197-3118.2005.03769.x
Abukhodair AW, Abukhudair W, Alqarni MS (2021) The effects of L-arginine in hypertensive patients: a literature review. Cureus 13:e20485. https://doi.org/10.7759/cureus.20485
Blachier F, Lancha AH, Boutry C et al (2010) Alimentary proteins, amino acids and cholesterolemia. Amino Acids 38:15–22. https://doi.org/10.1007/s00726-009-0239-6
Altorf-van der Kuil W, Engberink MF, De Neve M et al (2013) Dietary amino acids and the risk of hypertension in a Dutch older population: the rotterdam study. Am J Clin Nutr 97:403–410. https://doi.org/10.3945/ajcn.112.038737
Lagiou P, Sandin S, Lof M et al (2012) Low carbohydrate-high protein diet and incidence of cardiovascular diseases in Swedish women: prospective cohort study. BMJ 344:e4026. https://doi.org/10.1136/bmj.e4026
Stamler J, Elliott P, Kesteloot H et al (1996) Inverse relation of dietary protein markers with blood pressure. Findings for 10,020 men and women in the INTERSALT Study. INTERSALT Cooperative Research Group. INTERnational study of SALT and blood pressure. Circulation 94:1629–1634. https://doi.org/10.1161/01.cir.94.7.1629
Hruby A, Jacques PF (2018) Dietary protein and changes in markers of cardiometabolic health across 20 years of follow-up in middle-aged Americans. Public Health Nutr 21:2998–3010. https://doi.org/10.1017/S1368980018001854
Buendia JR, Bradlee ML, Singer MR et al (2015) Diets higher in protein predict lower high blood pressure risk in framingham offspring study adults. Am J Hypertens 28:372–379. https://doi.org/10.1093/ajh/hpu157
Liu L, Ikeda K, Sullivan DH et al (2002) Epidemiological evidence of the association between dietary protein intake and blood pressure: a meta-analysis of published data. Hypertens Res 25:689–695. https://doi.org/10.1291/hypres.25.689
der Altorf-van Kuil W, Engberink MF, Vedder MM et al (2012) Sources of dietary protein in relation to blood pressure in a general Dutch population. PLoS ONE 7:e30582. https://doi.org/10.1371/journal.pone.0030582
Altorf-van der Kuil W, Engberink MF, Geleijnse JM et al (2012) Sources of dietary protein and risk of hypertension in a general Dutch population. Br J Nutr 108:1897–1903. https://doi.org/10.1017/S0007114512000049
Tielemans SMAJ, Kromhout D, Altorf-van der Kuil W et al (2014) Associations of plant and animal protein intake with 5-year changes in blood pressure: the Zutphen elderly study. Nutr Metab Cardiovasc Dis 24:1228–1233. https://doi.org/10.1016/j.numecd.2014.05.013
Liu R, Dang S, Yan H et al (2013) Association between dietary protein intake and the risk of hypertension: a cross-sectional study from rural western China. Hypertens Res 36:972–979. https://doi.org/10.1038/hr.2013.71
Mente A, Dehghan M, Rangarajan S et al (2017) Association of dietary nutrients with blood lipids and blood pressure in 18 countries: a cross-sectional analysis from the PURE study. Lancet Diabetes Endocrinol 5:774–787. https://doi.org/10.1016/S2213-8587(17)30283-8
Pasiakos SM, Lieberman HR, Fulgoni VL (2015) Higher-protein diets are associated with higher HDL cholesterol and lower BMI and waist circumference in US adults. J Nutr 145:605–614. https://doi.org/10.3945/jn.114.205203
Sekgala MD, Opperman M, Mpahleni B et al (2022) Association between macronutrient and fatty acid consumption and metabolic syndrome: a South African taxi driver survey. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph192315452
Kohansal A, Zangene A, Turki Jalil A et al (2022) Association between plant and animal proteins intake with lipid profile and anthropometric indices: a cross-sectional study. Nutr Health. https://doi.org/10.1177/02601060221104311
Amamou T, Normandin E, Pouliot J et al (2017) Effect of a high-protein energy-restricted diet combined with resistance training on metabolic profile in older individuals with metabolic impairments. J Nutr Health Aging 21:67–74. https://doi.org/10.1007/s12603-016-0760-8
Landi F, Calvani R, Picca A et al (2018) Body mass index is strongly associated with hypertension: results from the longevity check-up 7+ study. Nutrients. https://doi.org/10.3390/nu10121976
Landi F, Calvani R, Picca A et al (2018) Relationship between cardiovascular health metrics and physical performance in community-living people: results from the longevity check-up (Lookup) 7+ project. Sci Rep 8:16353. https://doi.org/10.1038/s41598-018-34746-4
Landi F, Calvani R, Martone AM et al (2020) Normative values of muscle strength across ages in a “real world” population: results from the longevity check-up 7+ project. J Cachexia Sarcopenia Muscle 11:1562–1569. https://doi.org/10.1002/jcsm.12610
Marzetti E, Calvani R, Picca A et al (2018) Prevalence of dyslipidaemia and awareness of blood cholesterol levels among community-living people: results from the longevity check-up 7+ (lookup 7+) cross-sectional survey. BMJ Open. https://doi.org/10.1136/bmjopen-2018-021627
Coelho-Junior HJ, Calvani R, Picca A et al (2020) Association between dietary habits and physical function in Brazilian and Italian older women. Nutrients. https://doi.org/10.3390/nu12061635
Landi F, Calvani R, Picca A et al (2018) Cardiovascular health metrics, muscle mass and function among Italian community-dwellers: the lookup 7+ project. Eur J Public Health 28:766–772. https://doi.org/10.1093/eurpub/cky034
Landi F, Calvani R, Picca A et al (2018) Impact of habitual physical activity and type of exercise on physical performance across ages in community-living people. PLoS ONE 13:e0191820. https://doi.org/10.1371/journal.pone.0191820
Cacciatore S, Calvani R, Marzetti E et al (2023) Low adherence to mediterranean diet is associated with probable sarcopenia in community-dwelling older adults: results from the longevity check-up (lookup) 7+ project. Nutrients 15:1026. https://doi.org/10.3390/nu15041026
von Elm E, Altman DG, Egger M et al (2007) The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 370:1453–1457. https://doi.org/10.1016/S0140-6736(07)61602-X
Chobanian AV, Bakris GL, Black HR et al (2003) Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 42:1206–1252. https://doi.org/10.1161/01.HYP.0000107251.49515.c2
Landi F, Calvani R, Tosato M et al (2017) Animal-derived protein consumption is associated with muscle mass and strength in community-dwellers: results from the milan expo survey. J Nutr Health Aging 21:1050–1056. https://doi.org/10.1007/s12603-017-0974-4
Bergia RE, Campbell WW, Roseguini BT et al (2020) A high-protein meal does not improve blood pressure or vasoactive biomarker responses to acute exercise in humans. Nutr Res 81:97–107. https://doi.org/10.1016/j.nutres.2020.08.001
Rontoyanni VG, Werner K, Sanders TAB et al (2015) Differential acute effects of carbohydrate- and protein-rich drinks compared with water on cardiac output during rest and exercise in healthy young men. Appl Physiol Nutr Metab 40:803–810. https://doi.org/10.1139/apnm-2014-0358
Ramos RM, Coelho-Júnior HJ, do Prado RCR et al (2018) Moderate aerobic training decreases blood pressure but no other cardiovascular risk factors in hypertensive overweight/obese elderly patients. Gerontol Geriatr Med 4:233372141880864. https://doi.org/10.1177/2333721418808645
Igarashi Y, Nogami Y (2018) The effect of regular aquatic exercise on blood pressure: a meta-analysis of randomized controlled trials. Eur J Prev Cardiol 25:190–199. https://doi.org/10.1177/2047487317731164
Herrod PJJ, Doleman B, Blackwell JEM et al (2018) Exercise and other nonpharmacological strategies to reduce blood pressure in older adults: a systematic review and meta-analysis. J Am Soc Hypertens 12:248–267. https://doi.org/10.1016/J.JASH.2018.01.008
Mann S, Beedie C, Jimenez A (2014) Differential effects of aerobic exercise, resistance training and combined exercise modalities on cholesterol and the lipid profile: review, synthesis and recommendations. Sports Med 44:211–221. https://doi.org/10.1007/s40279-013-0110-5
MacDonald HV, Johnson BT, Huedo-Medina TB et al (2016) Dynamic resistance training as stand-alone antihypertensive lifestyle therapy: a meta-analysis. J Am Heart Assoc. https://doi.org/10.1161/JAHA.116.003231
Igarashi Y, Nogami Y (2019) Response of lipids and lipoproteins to regular aquatic endurance exercise: a meta-analysis of randomized controlled trials. J Atheroscler Thromb 26:14–30. https://doi.org/10.5551/jat.42937
Vemulapalli S, Dolor RJ, Hasselblad V et al (2015) Supervised vs unsupervised exercise for intermittent claudication: a systematic review and meta-analysis. Am Heart J 169:924-937.e3. https://doi.org/10.1016/j.ahj.2015.03.009
Hodgson JM, Zhu K, Lewis JR et al (2012) Long-term effects of a protein-enriched diet on blood pressure in older women. Br J Nutr 107:1664–1672. https://doi.org/10.1017/S0007114511004740
Kraus WE, Houmard JA, Duscha BD et al (2002) Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med 347:1483–1492. https://doi.org/10.1056/NEJMoa020194
Nybo L, Sundstrup E, Jakobsen MD et al (2010) High-intensity training versus traditional exercise interventions for promoting health. Med Sci Sports Exerc 42:1951–1958. https://doi.org/10.1249/MSS.0b013e3181d99203
Uchida MC, Nishida MM, Sampaio RAC et al (2016) Thera-band(®) elastic band tension: reference values for physical activity. J Phys Ther Sci 28:1266–1271. https://doi.org/10.1589/jpts.28.1266
Durau J, Diehl S, Terlutter R (2022) Motivate me to exercise with you: the effects of social media fitness influencers on users’ intentions to engage in physical activity and the role of user gender. Digit Heal 8:20552076221102770. https://doi.org/10.1177/20552076221102769
Tayyem R, Hijjawi NS, Al-Awwad N et al (2018) Association between intakes of macro- and micro- nutrients and serum lipid profiles among Jordanian adults: a preliminary study. Prog Nutr 20:361–371. https://doi.org/10.2375/pn.v20i3.7259
Umesawa M, Sato S, Imano H et al (2009) Relations between protein intake and blood pressure in Japanese men and women: the circulatory risk in communities study (CIRCS). Am J Clin Nutr 90:377–384. https://doi.org/10.3945/ajcn.2008.27109
He J, Yu S, Fang A et al (2022) Association between protein intake and the risk of hypertension among Chinese men and women: a longitudinal study. Nutrients. https://doi.org/10.3390/nu14061276
Mehrabani S, Asemi M, Najafian J et al (2017) Association of animal and plant proteins intake with hypertension in Iranian adult population: isfahan healthy heart program. Adv Biomed Res 6:112. https://doi.org/10.4103/2277-9175.213877
Hirche F, Schröder A, Knoth B et al (2006) Effect of dietary methionine on plasma and liver cholesterol concentrations in rats and expression of hepatic genes involved in cholesterol metabolism. Br J Nutr 95:879–888. https://doi.org/10.1079/bjn20061729
Kern M, Ellison D, Marroquin Y et al (2002) Effects of soy protein supplemented with methionine on blood lipids and adiposity of rats. Nutrition 18:654–656. https://doi.org/10.1016/s0899-9007(02)00783-9
Aoyama Y, Amano N, Yoshida A (1999) Cholesterol synthesis and degradation in normal rats fed a cholesterol-free diet with excess cystine. Lipids 34:583–589. https://doi.org/10.1007/s11745-999-0402-8
Sérougne C, Férézou J, Rukaj A (1987) A new relationship between cholesterolemia and cholesterol synthesis determined in rats fed an excess of cystine. Biochim Biophys Acta 921:522–530. https://doi.org/10.1016/0005-2760(87)90080-4
Choi M-J, Kim J-H, Chang KJ (2006) The effect of dietary taurine supplementation on plasma and liver lipid concentrations and free amino acid concentrations in rats fed a high-cholesterol diet. Adv Exp Med Biol 583:235–242. https://doi.org/10.1007/978-0-387-33504-9_25
Mizushima S, Nara Y, Sawamura M et al (1996) Effects of oral taurine supplementation on lipids and sympathetic nerve tone. Adv Exp Med Biol 403:615–622. https://doi.org/10.1007/978-1-4899-0182-8_68
Zhao Y, Dai X, Zhou Z et al (2016) Leucine supplementation via drinking water reduces atherosclerotic lesions in apoE null mice. Acta Pharmacol Sin 37:196–203. https://doi.org/10.1038/aps.2015.88
DeSouza CA, Shapiro LF, Clevenger CM et al (2000) Regular aerobic exercise prevents and restores age-related declines in endothelium-dependent vasodilation in healthy men. Circulation 102:1351–1357. https://doi.org/10.1161/01.cir.102.12.1351
Padilla J, Simmons GH, Bender SB et al (2011) Vascular effects of exercise: endothelial adaptations beyond active muscle beds. Physiology 26:132–145. https://doi.org/10.1152/physiol.00052.2010
Green DJ, Smith KJ (2018) Effects of exercise on vascular function, structure, and health in humans. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a029819
Palloshi A, Fragasso G, Piatti P et al (2004) Effect of oral L-arginine on blood pressure and symptoms and endothelial function in patients with systemic hypertension, positive exercise tests, and normal coronary arteries. Am J Cardiol 93:933–935. https://doi.org/10.1016/j.amjcard.2003.12.040
Shiraseb F, Asbaghi O, Bagheri R et al (2022) Effect of l-arginine supplementation on blood pressure in adults: a systematic review and dose-response meta-analysis of randomized clinical trials. Adv Nutr 13:1226–1242. https://doi.org/10.1093/advances/nmab155
Tanahashi K, Akazawa N, Miyaki A et al (2014) Aerobic exercise training decreases plasma asymmetric dimethylarginine concentrations with increase in arterial compliance in postmenopausal women. Am J Hypertens 27:415–421. https://doi.org/10.1093/ajh/hpt217
Stuehr DJ (2004) Enzymes of the L-arginine to nitric oxide pathway. J Nutr 134:2748S-2751S. https://doi.org/10.1093/jn/134.10.2748S
Wilkinson DJ, Hossain T, Hill DS et al (2013) Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism. J Physiol 591:2911–2923. https://doi.org/10.1113/jphysiol.2013.253203
Aspenes ST, Karlsen T (2012) Exercise-training intervention studies in competitive swimming. Sport Med 42:527–543. https://doi.org/10.2165/11630760-000000000-00000
Nikolaidis PT, Knechtle B (2020) Force–velocity characteristics and maximal anaerobic power in male recreational marathon runners. Res Sport Med 28:99–110. https://doi.org/10.1080/15438627.2019.1608993
Han K, Park Y-M, Kwon H-S et al (2014) Sarcopenia as a determinant of blood pressure in older Koreans: findings from the Korea national health and nutrition examination surveys (KNHANES) 2008–2010. PLoS ONE 9:e86902. https://doi.org/10.1371/journal.pone.0086902
Bai T, Fang F, Li F et al (2020) Sarcopenia is associated with hypertension in older adults: a systematic review and meta-analysis. BMC Geriatr 20:279
Liang M, Pan Y, Zhong T et al (2021) Effects of aerobic, resistance, and combined exercise on metabolic syndrome parameters and cardiovascular risk factors: a systematic review and network meta-analysis. Rev Cardiovasc Med 22:1523–1533. https://doi.org/10.3108/j.rcm2204156
Dong J-Y, Zhang Z-L, Wang P-Y et al (2013) Effects of high-protein diets on body weight, glycaemic control, blood lipids and blood pressure in type 2 diabetes: meta-analysis of randomised controlled trials. Br J Nutr 110:781–789. https://doi.org/10.1017/S0007114513002055
Liguori I, Russo G, Coscia V et al (2018) Orthostatic hypotension in the elderly: a marker of clinical frailty? JAMDA 19:779–785. https://doi.org/10.1016/j.jamda.2018.04.018
Ogonowska-Slodownik A, Morgulec-Adamowicz N, Geigle PR et al (2022) Objective and self-reported assessment of physical activity of women over 60 years old. Ageing Int 47:307–320. https://doi.org/10.1007/s12126-021-09423-z
Funding
Open access funding provided by Università Cattolica del Sacro Cuore within the CRUI-CARE Agreement. This work was supported by the Università Cattolica del Sacro Cuore [D1.2020, D1.2022, and D1.2023], the Italian Ministry of Health [Ricerca Corrente 2023] and the nonprofit research foundation “Centro Studi Achille e Linda Lorenzon” [N/A]. The authors also acknowledge co-funding from Next Generation EU, in the context of the National Recovery and Resilience Plan, Investment PE8 – Project Age-It: “Ageing Well in an Ageing Society”. This resource was co-financed by the Next Generation EU [DM 1557 11.10.2022]. The views and opinions expressed are only those of the authors and do not necessarily reflect those of the European Union or the European Commission. Neither the European Union nor the European Commission can be held responsible for them.
Author information
Authors and Affiliations
Contributions
Conceptualization, FL, HJC-J, EM, RC, AP, and MT; Data curation, HJC-J, EM, RC, and MT; Formal analysis, HJC-J; Funding acquisition, FL and EM; Investigation, FL, GS, EM, RC, AP, and MT; Methodology, FL, HJC-J, GS, EM, and AP; Resources, FL; Supervision, FL and EM; Writing—original draft, HJC-J; Writing—review & editing, FL, EM, HJC-J, and AP.
Corresponding authors
Ethics declarations
Conflict of interests
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Lookup 7 + protocol was approved by the Ethics Committee of the Università Cattolica del Sacro Cuore (protocol #: A.1220/CE/2011) and each participant provided written informed consent prior to enrolment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Coelho-Júnior, H.J., Calvani, R., Picca, A. et al. Adherence to aerobic training combined with high protein intake is associated with low blood pressure in Italian older adults: a cross-sectional study. Aging Clin Exp Res 35, 2613–2621 (2023). https://doi.org/10.1007/s40520-023-02549-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-023-02549-x