Abstract
Background
Some studies have employed machine learning (ML) methods for mobility prediction modeling in older adults. ML methods could be a helpful tool for life-space mobility (LSM) data analysis.
Aim
This study aimed to evaluate the predictive value of ML algorithms for the restriction of life-space mobility (LSM) among elderly people and to identify the most important risk factors for that prediction model.
Methods
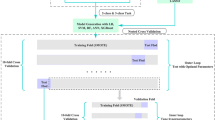
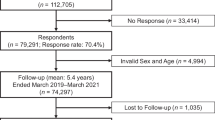
A 2-year LSM reduction prediction model was developed using the ML-based algorithms decision tree, random forest, and eXtreme gradient boosting (XGBoost), and tested on an independent validation cohort. The data were collected from the International Mobility in Aging Study (IMIAS) from 2012 to 2014, comprising 372 older patients (≥ 65 years of age). LSM was measured by the Life-Space Assessment questionnaire (LSA) with five levels of living space during the month before assessment.
Results
According to the XGBoost algorithm, the best model reached a mean absolute error (MAE) of 10.28 and root-mean-square error (RMSE) of 12.91 in the testing portion. The variables frailty (39.4%), mobility disability (25.4%), depression (21.9%), and female sex (13.3%) had the highest importance.
Conclusion
The model identified risk factors through ML algorithms that could be used to predict LSM restriction; these risk factors could be used by practitioners to identify older adults with an increased risk of LSM reduction in the future. The XGBoost model offers benefits as a complementary method of traditional statistical approaches to understand the complexity of mobility.

Similar content being viewed by others
References
Baker PS, Bodner EV, Allman RM (2003) Measuring life-space mobility in community-dwelling older adults. J Am Geriatr Soc 51:1610–1614
Webber SC, Porter MM, Menec VH (2010) Mobility in older adults: a comprehensive framework. Gerontologist 50:443–450
Lo AX, Rundle AG, Buys D et al (2016) Neighborhood disadvantage and life-space mobility are associated with incident falls in community-dwelling older adults. J Am Geriatr Soc 64:2218–2225
James BD, Boyle PA, Buchman AS et al (2011) Life space and risk of Alzheimer disease, mild cognitive impairment, and cognitive decline in old age. Am J Geriatr Psychiatry 19:961–969
Portegijs E, Rantakokko M, Viljanen A et al (2016) Is frailty associated with life-space mobility and perceived autonomy in participation outdoors? A longitudinal study. Age Ageing 45:550–553
Fathi R, Bacchetti P, Haan MN et al (2017) Life-space assessment predicts hospital readmission in home-limited adults. J Am Geriatr Soc 65:1004–1011
Kennedy RE, Williams CP, Sawyer P et al (2019) Life-space predicts health care utilization in community-dwelling older adults. J Aging Health 31:280–292
Sheppard KD, Sawyer P, Ritchie CS et al (2013) Life-space mobility predicts nursing home admission over 6 years. J Aging Health 25:907–920
Rantakokko M, Portegijs E, Viljanen A et al (2015) Changes in life-space mobility and quality of life among community-dwelling older people: a 2-year follow-up study. Qual Life Res 25:1189–1197
Kennedy RE, Sawyer P, Williams CP et al (2017) Life-space mobility change predicts 6-month mortality. J Am Geriatr Soc 65:833–838
Taylor JK, Buchan IE, van der Veer SN (2019) Assessing life-space mobility for a more holistic view on wellbeing in geriatric research and clinical practice. Aging Clin Exp Res 31:439–445
Rajkomar A, Dean J, Kohane I (2019) machine learning in medicine. N Engl J Med 380:1347–1358
Beam AL, Kohane IS (2018) Big data and machine learning in health care. JAMA J Am Med Assoc 319:1317–1318
Odden MC, Melzer D (2019) Machine learning in aging research. J Gerontol A Biol Sci Med Sci 74:1901–1902
Chang W, Liu Y, Xiao Y et al (2019) A machine-learning-based prediction method for hypertension outcomes based on medical data. Diagnostics 9:178
Budholiya K, Shrivastava SK, Sharma V (2020) An optimized XGBoost based diagnostic system for effective prediction of heart disease. J King Saud Univ Comput Inf Sci. https://doi.org/10.1016/j.jksuci.2020.10.013
Wang L, Wang X, Chen A et al (2020) Prediction of type 2 diabetes risk and its effect evaluation based on the xgboost model. Healthc 8:1–11
Zhang L, Wang Y, Niu M et al (2020) Machine learning for characterizing risk of type 2 diabetes mellitus in a rural Chinese population: the Henan Rural Cohort Study. Sci Rep 10:1–10
Byeon H (2021) Development of a physical impairment prediction model for korean elderly people using synthetic minority over-sampling technique and XGBoost. Int J Adv Comput Sci Appl 12:34–41
Ye C, Li J, Hao S et al (2020) Identification of elders at higher risk for fall with statewide electronic health records and a machine learning algorithm. Int J Med Inform 137:104105
Noh B, Youm C, Goh E et al (2021) XGBoost based machine learning approach to predict the risk of fall in older adults using gait outcomes. Sci Rep 11:1–9. https://doi.org/10.1038/s41598-021-91797-w
Kheirkhahan M, Tudor-Locke C, Axtell R et al (2016) Actigraphy features for predicting mobility disability in older adults. Physiol Meas 37:1813–1833. https://doi.org/10.1088/0967-3334/37/10/1813
Hirata K, Suzuki M, Iso N et al (2021) Using machine learning to investigate the relationship between domains of functioning and functional mobility in older adults. PLoS ONE 16:e0246397. https://doi.org/10.1371/journal.pone.0246397
do Nascimento CF, Batista AFM, Duarte YAO et al (2022) Early identification of older individuals at risk of mobility decline with machine learning. Arch Gerontol Geriatr 100:104625. https://doi.org/10.1016/j.archger.2022.104625
Speiser JL, Callahan KE, Ip EH et al (2022) Predicting future mobility limitation in older adults: a machine learning analysis of health ABC study data. J Gerontol A Biol Sci Med Sci 77:1072–1078. https://doi.org/10.1093/gerona/glab269
Zunzunegui MV, Alvarado BE, Guerra R et al (2015) The mobility gap between older men and women: the embodiment of gender. Arch Gerontol Geriatr 61:140–148
Gomez F, Zunzunegui MV, Alvarado B et al (2018) Cohort profile: the international mobility in aging study (IMIAS). Int J Epidemiol 47:1393
Curcio C-L, Alvarado E, Gomez F et al (2013) Life-space assessment scale to assess mobility: validation in Latin American older women and men. Aging Clin Exp Res 25:553–560
Sawyer P, Allman RM (2010) Resilience in mobility in the context of chronic disease and aging: cross-sectional and prospective findings from the University of Alabama at Birmingham (UAB) study of aging. In: New frontiers in resilient aging: life-strengths and well-being in late life, pp 310–39
Johnson J, Rodriguez M, Al Snih S (2020) Life-space mobility in the elderly: current perspectives. Clin Interv Aging 15:1665–1674
De Yébenes MJ, Otero A, Zunzunegui MV et al (2003) Validation of a short cognitive tool for the screening of dementia in elderly people with low educational level. Int J Geriatr Psychiatry 18:925–936
Roadolff LS (1977) The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas 1:385–401
Ylli A, Miszkurka M, Phillips SP et al (2016) Clinically relevant depression in old age: An international study with populations from Canada, Latin America and Eastern Europe. Psychiatry Res 241:236–241
de Alencar Caldas VV, Zunzunegui MV, FalcãoFreire ADN et al (2012) Translation, cultural adaptation and psychometric evaluation of the leganés cognitive test in a low educated elderly Brazilian population. Arq Neuropsiquiatr 70:22–27
Fried LP, Tangen CM, Walston J et al (2001) Frailty in older adults: evidence for a phenotype. J Gerontol Ser A Biol Sci Med Sci 56:146–157
Fernandes J, dos Santos Gomes C, Guerra RO et al (2021) Frailty syndrome and risk of cardiovascular disease: analysis from the International Mobility in Aging Study. Arch Gerontol Geriatr 92:104279
Nagi SZ (1976) An epidemiology of disability among adults in the United States. Milbank Mem Fund Q Health Soc 54:439
Chen T, Guestrin C (2016) XGBoost: A scalable tree boosting system. In Proc ACM SIGKDD int conf knowl discov data min. 13–17-Augu, pp 785–94
Pedregosa F, Varoquaux G, Gramfort A et al (2011) Scikit-learn: machine learning in python. J Mach Learn Res 12:2825–2830
Peel C, Baker PS, Roth DL et al (2005) Assessing mobility in older adults: the UAB Study of Aging Life-Space Assessment. Phys Ther 85:1008–1119
Choi M, O’Connor ML, Mingo CA et al (2016) Gender and racial disparities in life-space constriction among older adults. Gerontologist 56:1153–1160
Xue QL, Fried LP, Glass TA et al (2008) Life-space constriction, development of frailty, and the competing risk of mortality: The women’s health and aging study I. Am J Epidemiol 167:240–248
Tsai LT, Portegijs E, Rantakokko M et al (2015) The association between objectively measured physical activity and life-space mobility among older people. Scand J Med Sci Sport 25:e368–e373
Polku H, Mikkola TM, Portegijs E et al (2015) Life-space mobility and dimensions of depressive symptoms among community-dwelling older adults. Aging Ment Health 19:781–789
Kuspinar A, Verschoor CP, Beauchamp MK et al (2020) Modifiable factors related to life-space mobility in community-dwelling older adults: results from the Canadian Longitudinal Study on Aging. BMC Geriatr 20:35
Miyashita T, Tadaka E, Arimoto A (2021) Cross-sectional study of individual and environmental factors associated with life-space mobility among community-dwelling independent older people. Environ Health Prev Med 26:1–8
Ansai JH et al (2019) Performance of different timed up and go subtasks in frailty syndrome. J Geriatr Phys Ther 42:287–293
Al Snih S, Peek KM, Sawyer P et al (2012) Life-space mobility in Mexican Americans aged 75 and older. J Am Geriatr Soc 60:532–537
Gonzalez BCS, Delgado LH, Quevedo JEC et al (2013) Life- space mobility, perceived health, and depression symptoms in a sample of Mexican older adults. Hisp Health Care Int 11:14–20. https://doi.org/10.1891/1540-4153.11.1.14
Acknowledgements
We are grateful to all the older adults who have given their time and confidence to this research. We would also like to thank our universities and research groups for providing continuous support.
Funding
This work was supported by The Canadian Institutes of Health Research (Grant No. AAM 108751).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was approved by the ethics committees of each site.
Human and animal rights statement
The study including human participants has been performed in accordance with the ethical standards of the declaration of Helsinki and its later ammendments. Institutional review board statement this study was approved by the ethical committee of Universidad de Caldas.
Informed consent
Written informed consent was obtained from all subjects before their participation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pérez-Trujillo, M., Curcio, CL., Duque-Méndez, N. et al. Predicting restriction of life-space mobility: a machine learning analysis of the IMIAS study. Aging Clin Exp Res 34, 2761–2768 (2022). https://doi.org/10.1007/s40520-022-02227-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-022-02227-4