Abstract
Purpose of Review
Environmental exposures during early stages of life may be particularly relevant for cancer etiology because of the rapid hormonal and tissue changes that occur during puberty and, in women, through first birth. We review evidence from the past 5 years on environmental exposures during childhood/adolescence through first birth and the risk of breast and other cancers during adulthood.
Recent Findings
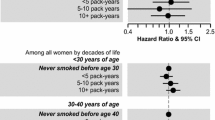
The studies of breast cancer (n = 14) reported associations for childhood/adolescent environmental tobacco smoke (ETS), smoking initiation, pesticides, hair dye use, and living on a road with high traffic. Smoking before first childbirth was also associated with increased breast cancer risk. We identified 12 studies on other cancers, with only 1–2 studies per cancer type, with most focused on ETS or active smoking.
Summary
Despite studies suggesting an important role of exposure to environmental factors during early life and cancer risk in adulthood, few studies have been conducted. Future studies could utilize stored biologic samples from relevant periods or complete residential histories for geographically-based exposures.
Similar content being viewed by others
Data Availability
This article did not involve the use of raw data.
References
President’s Cancer Panel. Reducing environmental cancer risk: what can we do now. Bethesda: National Cancer Institute; 2010.
Russo J, Russo IH. Development of the human breast. Maturitas. 2004;49(1):2–15.
Macias H, Hinck L. Mammary gland development. Wiley Interdiscip Rev Dev Biol. 2012;1(4):533–57.
Terry MB, Michels KB, Brody JG, Byrne C, Chen S, Jerry DJ, Malecki KMC, Martin MB, Miller RL, Neuhausen SL, et al. Environmental exposures during windows of susceptibility for breast cancer: a framework for prevention research. Breast Cancer Res. 2019;21(1):96.
Fenton SE. Endocrine-disrupting compounds and mammary gland development: early exposure and later life consequences. Endocrinology. 2006;147(6 Suppl):S18–24.
Martinson HA, Lyons TR, Giles ED, Borges VF, Schedin P. Developmental windows of breast cancer risk provide opportunities for targeted chemoprevention. Exp Cell Res. 2013;319(11):1671–8.
Russo J, Russo IH. Experimentally induced mammary tumors in rats. Breast Cancer Res Treat. 1996;39(1):7–20.
Russo J, Hu YF, Yang X, Russo IH. Developmental, cellular, and molecular basis of human breast cancer. J Natl Cancer Inst Monogr. 2000;27:17–37.
Bodicoat DH, Schoemaker MJ, Jones ME, McFadden E, Griffin J, Ashworth A, Swerdlow AJ. Timing of pubertal stages and breast cancer risk: the Breakthrough Generations Study. Breast Cancer Res. 2014;16(1):R18.
Goldberg M, D’Aloisio AA, O'Brien KM, Zhao S, Sandler DP. Pubertal timing and breast cancer risk in the Sister Study cohort. Breast Cancer Res. 2020;22(1):112.
Biro FM, Huang B, Wasserman H, Gordon CM, Pinney SM. Pubertal growth, IGF-1, and windows of susceptibility: puberty and future breast cancer risk. J Adolesc Health. 2021;68(3):517–22.
Henderson BE, Feigelson HS. Hormonal carcinogenesis. Carcinogenesis. 2000;21(3):427–33.
Henderson BE, Ross RK, Pike MC, Casagrande JT. Endogenous hormones as a major factor in human cancer. Cancer Res. 1982;42(8):3232–9.
Yu H, Rohan T. Role of the insulin-like growth factor family in cancer development and progression. J Natl Cancer Inst. 2000;92(18):1472–89.
Mantovani A, Fucic A. Puberty dysregulation and increased risk of disease in adult life: possible modes of action. Reprod Toxicol (Elmsford, NY). 2014;44:15–22.
Kripke M, Brody JG, Hawk E, Hernandez AB, Hoppin PJ, Jacobs MM, Rudel RA, Rebbeck TR. Rethinking environmental carcinogenesis. Cancer Epidemiol Biomark Prevent. 2020;29(10):1870–5.
Ben-Shlomo Y, Kuh D. A life course approach to chronic disease epidemiology: conceptual models, empirical challenges and interdisciplinary perspectives. Int J Epidemiol. 2002;31(2):285–93.
Williams PG, Holmbeck GN, Greenley RN. J Consult Clin Psychol. 2002;70(3):828.
Dorn LD, Hostinar CE, Susman EJ, Pervanidou P. Conceptualizing puberty as a window of opportunity for impacting health and well-being across the life span. J Res Adolesc. 2019;29(1):155–76.
Gaston SA, James-Todd T, Harmon Q, Taylor KW, Baird D, Jackson CL. Chemical/straightening and other hair product usage during childhood, adolescence, and adulthood among African-American women: potential implications for health. J Expos Sci Environ Epidemiol. 2020;30(1):86–96.
Hart LB, Walker J, Beckingham B, Shelley A, Alten Flagg M, Wischusen K, Sundstrom B. A characterization of personal care product use among undergraduate female college students in South Carolina, USA. J Expos Sci Environ Epidemiol. 2020;30(1):97–106.
Harley KG, Kogut K, Madrigal DS, Cardenas M, Vera IA, Meza-Alfaro G, She J, Gavin Q, Zahedi R, Bradman A, et al. Reducing phthalate, paraben, and phenol exposure from personal care products in adolescent girls: findings from the HERMOSA Intervention Study. Environ Health Perspect. 2016;124(10):1600–7.
Zota AR, Singla V, Adamkiewicz G, Mitro SD, Dodson RE. Reducing chemical exposures at home: opportunities for action. J Epidemiol Community Health. 2017;71(9):937–40.
Colditz GA, Bohlke K. Priorities for the primary prevention of breast cancer. CA Cancer J Clin. 2014;64(3):186–94.
Hiatt RA, Haslam SZ, Osuch J. The breast cancer and the environment research centers: transdisciplinary research on the role of the environment in breast cancer etiology. Environ Health Perspect. 2009;117(12):1814–22.
Holman DM, Rodriguez JL, Peipins L, Watson M, White MC. Highlights from a workshop on opportunities for cancer prevention during preadolescence and adolescence. J Adolesc Health. 2013;52(5 Supplement):S8–S14.
Mahabir S, Aagaard K, Anderson LM, Herceg Z, Hiatt RA, Hoover RN, Linet MS, Medina D, Potischman N, Tretli S, et al. Challenges and opportunities in research on early-life events/exposures and cancer development later in life. Cancer Causes Control. 2012;23(6):983–90.
Rodgers KM, Udesky JO, Rudel RA, Brody JG. Environmental chemicals and breast cancer: an updated review of epidemiological literature informed by biological mechanisms. Environ Res. 2018;160:152–82.
White AJ, D'Aloisio AA, Nichols HB, DeRoo LA, Sandler DP. Breast cancer and exposure to tobacco smoke during potential windows of susceptibility. Cancer Causes Control. 2017;28(7):667–75.
Lee PN, Hamling JS. Environmental tobacco smoke exposure and risk of breast cancer in nonsmoking women. An updated review and meta-analysis. Inhal Toxicol. 2016;28(10):431–54.
Gram IT, Park SY, Maskarinec G, Wilkens LR, Haiman CA, Le Marchand L. Smoking and breast cancer risk by race/ethnicity and oestrogen and progesterone receptor status: the Multiethnic Cohort (MEC) study. Int J Epidemiol. 2019;48(2):501–11.
Jones ME, Schoemaker MJ, Wright LB, Ashworth A, Swerdlow AJ. Smoking and risk of breast cancer in the Generations Study cohort. Breast Cancer Res. 2017;19(1):118.
Gaudet MM, Carter BD, Brinton LA, Falk RT, Gram IT, Luo J, Milne RL, Nyante SJ, Weiderpass E, Beane Freeman LE, et al. Pooled analysis of active cigarette smoking and invasive breast cancer risk in 14 cohort studies. Int J Epidemiol. 2017;46(3):881–93.
Li H, Terry MB, Antoniou AC, Phillips KA, Kast K, Mooij TM, Engel C, Noguès C, Stoppa-Lyonnet D, Lasset C, et al. Alcohol consumption, cigarette smoking, and risk of breast cancer for BRCA1 and BRCA2 mutation carriers: results from the BRCA1 and BRCA2 cohort consortium. Cancer Epidemiol Biomark Prevent. 2020;29(2):368–78.
Andersen ZJ, Jørgensen JT, Grøn R, Brauner EV, Lynge E. Active smoking and risk of breast cancer in a Danish nurse cohort study. BMC Cancer. 2017;17(1):556.
Bachelet D, Verner M-A, Neri M, Cordina Duverger É, Charlier C, Arveux P, Haddad S, Guénel P. Breast Cancer and exposure to organochlorines in the CECILE study: associations with plasma levels measured at the time of diagnosis and estimated during adolescence. Int J Environ Res Public Health. 2019;16(2):271.
Cohn BA, Cirillo PM, Terry MB. DDT and breast cancer: prospective study of induction time and susceptibility windows. J Natl Cancer Inst. 2019;111(8):803–10.
Niehoff NM, Nichols HB, White AJ, Parks CG, D'Aloisio AA, Sandler DP. Childhood and adolescent pesticide exposure and breast cancer risk. Epidemiology (Cambridge, Mass). 2016;27(3):326–33.
Shmuel S, White AJ, Sandler DP. Residential exposure to vehicular traffic-related air pollution during childhood and breast cancer risk. Environ Res. 2017;159:257–63.
Rai R, Glass DC, Heyworth JS, Saunders C, Fritschi L. Occupational exposures to engine exhausts and other PAHs and breast cancer risk: a population-based case-control study. Am J Ind Med. 2016;59(6):437–44.
White AJ, Gregoire AM, Taylor KW, Eberle C, Gaston S, O'Brien KM, Jackson CL, Sandler DP. Adolescent use of hair dyes, straighteners and perms in relation to breast cancer risk. Int J Cancer. 2021;148(9):2255–63.
Vieira VM, VoPham T, Bertrand KA, James P, DuPré N, Tamimi RM, Laden F, Hart JE. Contribution of socioeconomic and environmental factors to geographic disparities in breast cancer risk in the Nurses’ Health Study II. Environ Epidemiol (Philadelphia, Pa). 2020;4(1):e080.
Wang T, Townsend MK, Vinci C, Jake-Schoffman DE, Tworoger SS. Early life exposure to tobacco smoke and ovarian cancer risk in adulthood. Int J Epidemiol. 2021.
Moirano G, Zugna D, Grasso C, Mirabelli D, Lista P, Ciuffreda L, Segnan N, Merletti F, Richiardi L. Postnatal risk factors for testicular cancer: the EPSAM case-control study. Int J Cancer. 2017;141(9):1803–10.
Abdel-Rahman O. Incidence and mortality of lung cancer among never smokers in relationship to secondhand smoking: findings from the PLCO trial. Clin Lung Cancer. 2020;21(5):415–420.e412.
Molina-Montes E, Van Hoogstraten L, Gomez-Rubio P, Löhr M, Sharp L, Molero X, Márquez M, Michalski CW, Farré A, Perea J, et al. Pancreatic cancer risk in relation to lifetime smoking patterns, tobacco type, and dose-response relationships. Cancer Epidemiol Biomark Prevent. 2020;29(5):1009–18.
Wong JYY, Downward GS, Hu W, Portengen L, Seow WJ, Silverman DT, Bassig BA, Zhang J, Xu J, Ji BT, et al. Lung cancer risk by geologic coal deposits: a case-control study of female never-smokers from Xuanwei and Fuyuan, China. Int J Cancer. 2019;144(12):2918–27.
Würtz ET, Hansen J, Røe OD, Omland Ø. Asbestos exposure and haematological malignancies: a Danish cohort study. Eur J Epidemiol. 2020;35(10):949–60.
Deziel NC, Warren JL, Huang H, Zhou H, Sjodin A, Zhang Y. Exposure to polychlorinated biphenyls and organochlorine pesticides and thyroid cancer in Connecticut women. Environ Res. 2021;192:110333.
Utada M, Brenner AV, Preston DL, Cologne JB, Sakata R, Sugiyama H, Sadakane A, Grant EJ, Cahoon EK, Ozasa K, et al. Radiation risks of uterine cancer in atomic bomb survivors: 1958–2009. JNCI Cancer Spectrum. 2019;2(4).
Weinberg CR, Shore DL, Umbach DM, Sandler DP. Using risk-based sampling to enrich cohorts for endpoints, genes, and exposures. Am J Epidemiol. 2007;166(4):447–55.
Oh SM, Ryu BT, Lee SK, Chung KH. Antiestrogenic potentials of ortho-PCB congeners by single or complex exposure. Arch Pharm Res. 2007;30(2):199–209.
Cohn BA, Wolff MS, Cirillo PM, Sholtz RI. DDT and breast cancer in young women: new data on the significance of age at exposure. Environ Health Perspect. 2007;115(10):1406–14.
Li M, Liu X, Zhang L. The relationship of indoor coal use and environmental tobacco smoke exposure with lung cancer in China: a meta-analysis. J Cancer Res Ther. 2018;14(8):7–13.
Yan H, Ying Y, Xie H, Li J, Wang X, He L, Jin K, Tang J, Xu X, Zheng X. Secondhand smoking increases bladder cancer risk in nonsmoking population: a meta-analysis. Cancer Manag Res. 2018;10:3781–91.
van Osch FH, Jochems SH, van Schooten F-J, Bryan RT, Zeegers MP. Quantified relations between exposure to tobacco smoking and bladder cancer risk: a meta-analysis of 89 observational studies. Int J Epidemiol. 2016;45(3):857–70.
Lee PN, Thornton AJ, Hamling JS. Epidemiological evidence on environmental tobacco smoke and cancers other than lung or breast. Regul Toxicol Pharmacol. 2016;80:134–63.
International Agency for Research on Cancer. Personal habits and indoor combustions, vol. 100E. Lyon: World Health Organization; 2012.
Moon MK. Concern about the safety of bisphenol A substitutes. Diabetes Metab J. 2019;43(1):46–8.
Birnbaum LS, Grandjean P. Alternatives to PFASs: perspectives on the science. Environ Health Perspect. 2015;123(5):A104–5.
Hamra GB, Buckley JP. Environmental exposure mixtures: questions and methods to address them. Curr Epidemiol Rep. 2018;5(2):160–5.
Breast Cancer and the Environment Research Program [https://epi.grants.cancer.gov/bcerp/]
Code Availability
This article did not involve the use of code
Funding
This research was supported by the Intramural Research Program of the National Institute of Health, National Institute of Environmental Health Sciences, project number ZIAES103332-01
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Not applicable
Consent to Participate
Not applicable
Consent for Publication
Not applicable
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cancer Epidemiology
Rights and permissions
About this article
Cite this article
Niehoff, N.M., Goldberg, M. & White, A.J. The Importance of Addressing Early-Life Environmental Exposures in Cancer Epidemiology. Curr Epidemiol Rep 9, 49–65 (2022). https://doi.org/10.1007/s40471-022-00289-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40471-022-00289-6