Abstract
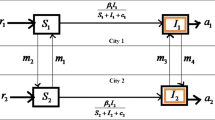
To describe the impact of population mobility between different cities on the spread of infectious disease, a new infectious disease complex dynamical model is proposed. Moreover, we obtain the basic regeneration number of the model based on applied spectral analysis. And the disease-free equilibrium points and local equilibrium points of the model are discussed, and it is found that two kind equilibrium points are globally asymptotically stable. In addition, the final scale of the presented model is analyzed and an expression for the final scale is obtained. Furthermore, we analyze the impact of population mobility on the spread of infectious diseases via numerical simulations. Our results reveal that the increase of population mobility between two cities leads to more intense disease transmission. Finally, the influence of media effects on the spread of infectious diseases is investigated. It is shown that the spread of diseases is suppressed because of the increase of individual's self-isolation rate. Therefore, controlling the population mobility is an effective initiative to curb outbreaks of infectious diseases throughout the network. These results can provide a theoretical basis for preventing and controlling the spreading of infectious diseases.










Similar content being viewed by others
Availability of data and materials
We do not analyse or generate any datasets, because our work proceeds within a theoretical and mathematical approach.
Code availability
Code will be made available on request.
References
Koziol JA, Schnitzer JE (2022) Déjà vu all over again: racial, ethnic and age disparities in mortality from influenza 1918–19 and COVID-19 in the United States. Heliyon 8(4):e09299
Chowell G, Viboud C, Simonsen L et al (2011) The 1918–1920 influenza pandemic in Peru. Vaccine 29:B21–B26
Ghosh JK, Ghosh U (2023) Three dimensional epidemic model with non-monotonic incidence and saturated treatment: a case study of SARS infection of Hong Kong 2003 Scenario. Results Control Optim 11:100239
Rodriguez T, Dobrovolny HM (2021) Quantifying the effect of trypsin and elastase on in vitro SARS-COV infections. Virus Res 299:198423
Basyouni KS, Khan AQ (2022) Discrete-time COVID-19 epidemic model with chaos, stability and bifurcation. Results Phys 43:106038
Song H, Wang R, Liu S et al (2022) Global stability and optimal control for a COVID-19 model with vaccination and isolation delays. Results Phys 42:106011
Akhtar MU, Liu J, Liu X et al (2023) NRAND: An efficient and robust dismantling approach for infectious disease network. Inf Process Manag 60(2):103221
Nunner H, Buskens V, Teslya A et al (2022) Health behavior homophily can mitigate the spread of infectious diseases in small-world networks. Soc Sci Med 312:115350
Voinson M, Smadi C, Billiard S (2022) How does the host community structure affect the epidemiological dynamics of emerging infectious diseases? Ecol Model 472:110092
Schimit PHT, Monteiro LHA (2010) Who should wear mask against airborne infections? Altering the contact network for controlling the spread of contagious diseases. Ecol Model 221(9):1329–1332
Stensen DB, Cañadas RA, Småbrekke L et al (2022) Social network analysis of Staphylococcus aureus carriage in a general youth population. Int J Infect Dis 123:200–209
Lymperopoulos IN (2021) Stay home to contain Covid-19: neuro-SIR-neuro dynamical epidemic modeling of infection patterns in social networks. Expert Syst Appl 165:113970
Bouveret G, Mandel A (2021) Social interactions and the prophylaxis of SI epidemics on networks. J Math Econ 93:102486
Yao Y, Guo Z, Huang X et al (2023) Gauging urban resilience in the United States during the COVID-19 pandemic via social network analysis. Cities 138:104361
Marqués SP, Martínez FMC, Leirós R et al (2023) Leadership and contagion by COVID-19 among residence hall students: a social network analysis approach. Soc Netw 73:80–88
Zhao D, Gao B, Wang Y et al (2018) Optimal dismantling of interdependent networks based on inverse explosive percolation. IEEE Trans Circuits Syst II Express Briefs 65(7):953–957
Zhao D, Wang L, Wang Z et al (2018) Virus propagation and patch distribution in multiplex networks: modeling, analysis, and optimal allocation. IEEE Trans Inf Forensics Secur 14(7):1755–1767
Wang X, Wang Z (2022) Bifurcation and propagation dynamics of a discrete pair SIS epidemic model on networks with correlation coefficient. Appl Math Comput 435:127477
Zhu L, Liu W, Zhang Z (2020) Delay differential equations modeling of rumor propagation in both homogeneous and heterogeneous networks with a forced silence function. Appl Math Comput 370:124925
Li M, Ling Y, Ma J (2023) The dynamics of the risk perception on a social network and its effect on disease dynamics. Infect Dis Model 8(3):632–344
Ghosh S, Kundu P et al (2023) Dimension reduction in higher-order contagious phenomena. Chaos 33:053117
Das P, Upadhyay RK et al (2021) Mathematical model of COVID-19 with comorbidity and controlling using non-pharmaceutical interventions and vaccination. Nonlinear Dyn 106(2):1213–1227
Ghosh S, Senapati A et al (2021) Reservoir computing on epidemic spreading: a case study on COVID-19 cases. Phys Rev E 104:014308
Wu Q, Hadzibeganovic T (2018) Pair quenched mean-field approach to epidemic spreading in multiplex networks. Appl Math Model 60:244–254
Wang X, Wang Z, Shen H (2019) Dynamical analysis of a discrete-time SIS epidemic model on complex networks. Appl Math Lett 94:292–299
Xia C, Wang Z, Zheng C et al (2019) A new coupled disease-awareness spreading model with mass media on multiplex networks. Inf Sci 471:185–200
Zhang H, Shu P, Wang Z et al (2017) Preferential imitation can invalidate targeted subsidy policies on seasonal-influenza diseases. Appl Math Comput 294:332–342
Huang S, Chen F, Chen L (2017) Global dynamics of a network-based SIQRS epidemic model with demographics and vaccination. Commun Nonlinear Sci Numer Simul 43:296–310
Gong Y, Small M (2018) Epidemic spreading on meta population networks including migration and demographics. Chaos Interdiscip J Nonlinear Sci 28(8):083102
Jing W, Jin Z, Peng XL (2018) Adaptive SIS epidemic models on heterogeneous networks with demographics and risk perception. J Biol Syst 26(02):247–273
Jin Z, Sun G, Zhu H (2014) Epidemic models for complex networks with demographics. Math Biosci Eng 11(6):1295–1317
Pan W, Sun GQ, Jin Z (2015) How demography-driven evolving networks impact epidemic transmission between communities. J Theor Biol 382:309–319
Yao Y, Zhang J (2016) A two-strain epidemic model on complex networks with demographics. J Biol Syst 24(04):577–609
Giffin A, Gong W, Majumder S et al (2022) Estimating intervention effects on infectious disease control: the effect of community mobility reduction on Coronavirus spread. Spatial Stat 52:100711
Ensoy MC, Nguyen MH, Hens N et al (2023) Spatio-temporal model to investigate COVID-19 spread accounting for the mobility among municipalities. Spatial Spatio Temporal Epidemiol 45:100568
Rollier M, Miranda GHB, Vergeynst J et al (2023) Mobility and the spatial spread of sars-cov-2 in Belgium. Math Biosci 360:108957
Rafiq R, Ahmed T, Uddin MYS (2022) Structural modeling of COVID-19 spread in relation to human mobility. Transp Res Interdiscip Perspect 13:100528
Xia C, Wang Z, Zheng C (2019) A new coupled disease-awareness spreading model with mass media on multiplex networks. Inf Sci Int J 471:185–200
Wang S (2019) The impact of awareness diffusion on SIR-like epidemics in multiplex networks. Appl Math Comput 349:134–147
Ghosh S, Senapati A et al (2021) Optimal test-kit-based intervention strategy of epidemic spreading in heterogeneous complex networks. Chaos 31:071101
Van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Acknowledgements
First of all, I would like to give my heartfelt thanks to all the people who have ever helped me in this paper. My sincere and hearty thanks and appreciations go firstly to my supervisor, Mrs Yang Lixin, whose suggestions and encouragement have given me much insight into these translation studies. It has been a great privilege and joy to study under his guidance and supervision. Furthermore, it is my honor to benefit from his personality and diligence, which I will treasure my whole life. My gratitude to him knows no bounds. I am also extremely grateful to all my friends and classmates who have kindly provided me assistance and companionship in the course of preparing this paper. In addition, I would like to thank the National Natural Science Foundation of China for its support. This research was supported by The National Natural Science Foundation of China under Grant No.11702195.
Funding
This research was supported by The National Natural Science Foundation of China (11702195).
Author information
Authors and Affiliations
Contributions
YQ: Writing-Original Draft, Validation, Formal analysis, Visualization, Methodology. LY: Methodology, Writing-Review&Editing, Funding acquisition, Supervision, Resources. ZG: Resources, Visualization.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose. The authors have no competing interests to declare that are relevant to the content of this article. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. The authors have no financialor proprietary interests in any material discussed in this article. No conflict of interest exits in the submission of this manuscript, and manuscript is approved by all authors for publication. I would like to declare on behalf of my co-authors that the work described was original research that has not been published previously, and not under consideration for publication elsewhere, in whole or in part.
Appendix A: Specific derivation of stability analysis
Appendix A: Specific derivation of stability analysis
Stability plays a crucial role in the dynamic analysis of infectious disease models. The following content is the theoretical derivation of stability analysis. Let's first derive the global stability of the disease-free equilibrium point at \(R_{0} < 1\).
Firstly, the disease-free equilibrium \(E_{0}\) of system (2) is \(\left( {S_{i,k}^{0} ,S_{{q_{i,k} }}^{0} ,0,0,0} \right)\).
Where
System (2) is rewritten in the form.
Where \(X = \left( {S_{i,k} \left( t \right),S_{{q_{i,k} }} \left( t \right)} \right)^{\prime }\)\(Z = \left( {E_{i,k} \left( t \right),I_{i,k} \left( t \right),Q_{i,k} \left( t \right)} \right)^{\prime }\).
\(U_{0} = \left( {X_{0} ,0} \right) = \left( {S_{i,k}^{0} ,S_{{q_{i,k} }}^{0} ,0,0,0} \right)\) denotes the disease-free equilibrium of system.
Then, we have the following two conditions:
\(\left( {H_{1} } \right)\) For \(\frac{dX}{{dt}} = F\left( {X,0} \right) = \left( \begin{gathered} \delta_{i,k} A_{i} + q_{1} S_{{q_{i,k} }} - \left( {q + d_{1} } \right)S_{i,k} \hfill \\ qS_{i,k} - d_{1} S_{{q_{i,k} }} - q_{1} S_{{q_{i,k} }} \hfill \\ \end{gathered} \right)\),\(x_{0} = \left( {S_{i,k}^{0} ,S_{{q_{i,k} }}^{0} ,0,0,0} \right)\) is a globally asymptomatically stable equilibrium of \(\frac{dx}{{dt}} = F\left( {x,0} \right)\). Hence,\(U_{0}\) is globally asymptomatically stable.
And \({\varvec{A}}_{21} \user2{ = - V}_{21}\), \({\mathbf{A}}_{22} {\mathbf{ = }} - {\mathbf{V}}_{22}\), \({\varvec{A}}_{31} \user2{ = } - {\varvec{V}}_{31}\), \({\varvec{A}}_{32} \user2{ = } - {\varvec{V}}_{32}\), \({\varvec{A}}_{33} \user2{ = } - {\varvec{V}}_{33}\).
Where \(\rho_{1l} = \left( {\beta_{2} + \lambda_{2} } \right)k\xi_{2} \omega_{2} \frac{{S_{1,l}^{0} }}{{N_{2} }},\rho_{2l} = \left( {\beta_{1} + \lambda_{1} } \right)k\xi_{1} \omega_{1} \frac{{S_{2,l}^{0} }}{{N_{1} }},l = 1 \cdots m.\)
Note that \({\varvec{A}}\) is an Metzler matrix,\(\user2{ - A}\) is an M-matrix, and all eigenvalues with respect to \({\varvec{A}}\) have negative real parts when \(R_{0} < 1\).We derive that \(Z\left( t \right)\) is globally asymptomatically stable, which means.
Then, the disease-free equilibrium \(E_{0}\) is globally asymptomatically stable when \(R_{0} < 1\).
Next, we discuss the global stability of the local equilibrium point at \(R_{0} { > }1\).
Proof
Consider the following Lyapunov function candidate
where \(h_{1} ,h_{2} ,h_{3} ,h_{4}\) and \(h_{5}\) are positive constants to be determined later. Differentiating the function with respect to time yields.
Substituting system (A5) into the above formula and sorted out:
where \(x_{1} = \frac{{S_{i,k} }}{{S_{i,k}^{*} }},x_{2} = \frac{{S_{{q_{i,k} }} }}{{S_{{q_{i,k} }}^{*} }},x_{3} = \frac{{E_{i,k} }}{{E_{i,k}^{*} }},x_{4} = \frac{{I_{i,k} }}{{I_{i,k}^{*} }},x_{5} = \frac{{Q_{i,k} }}{{Q_{i,k}^{*} }}\).
The above formula can be simplified
Considering \(h_{3} = 1\), by setting the coefficients of \(x_{2} ,x_{3} ,x_{4} ,x_{5}\) to 0 and solving for \(h_{1} ,h_{2} ,h_{4}\) yields.
\(h_{1} = {{\left[ {\left( {q + d_{1} } \right) + a} \right]} \mathord{\left/ {\vphantom {{\left[ {\left( {q + d_{1} } \right) + a} \right]} q}} \right. \kern-0pt} q}.\)\(h_{2} = {{\left[ {\alpha_{i}^{2} \varepsilon_{i} + \alpha_{i}^{1} \left( {d_{1} + d_{2} + \alpha_{i}^{2} + \gamma_{i}^{2} } \right)} \right]} \mathord{\left/ {\vphantom {{\left[ {\alpha_{i}^{2} \varepsilon_{i} + \alpha_{i}^{1} \left( {d_{1} + d_{2} + \alpha_{i}^{2} + \gamma_{i}^{2} } \right)} \right]} {\left[ {\alpha_{i}^{2} \left( {d_{1} + \varepsilon_{i} + \alpha_{i}^{1} } \right)} \right]}}} \right. \kern-0pt} {\left[ {\alpha_{i}^{2} \left( {d_{1} + \varepsilon_{i} + \alpha_{i}^{1} } \right)} \right]}}\).\(h_{4} = {{\left[ {\left( {d_{1} + d_{2} + \alpha_{i}^{2} + \gamma_{i}^{2} } \right)} \right]} \mathord{\left/ {\vphantom {{\left[ {\left( {d_{1} + d_{2} + \alpha_{i}^{2} + \gamma_{i}^{2} } \right)} \right]} {\alpha_{i}^{2} }}} \right. \kern-0pt} {\alpha_{i}^{2} }}.\)\(a = \left( {\beta_{i} + \lambda_{i} } \right)k\left( {1 - \xi_{i} } \right)\theta_{i} + \left( {\beta_{{i^{\prime}}} + \lambda_{{i^{\prime}}} } \right)\left( {\frac{{I_{{i^{\prime}}} + E_{{i^{\prime}}} }}{{N_{{i^{\prime}}} }}} \right)k\xi_{i} \omega_{i}\).
Using the arithmetic–geometric means inequality, it is found that \(L\) is less or equal to zero with equality only if \(x_{1} = 1,x_{2} = 1,x_{3} = 1,x_{4} = 1,x_{5} = 1\). By LaSalle’s invariance principle, we know the largest invariant set in \(\Omega\) is reduced to the endemic equilibrium \(E^{*}\), and the largest invariant set contained in
Then, we conclude that the endemic equilibrium is globally asymptotically stable in \(\Omega\).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qin, Y., Yang, L. & Gu, Z. Dynamics behavior of a novel infectious disease model considering population mobility on complex network. Int. J. Dynam. Control (2024). https://doi.org/10.1007/s40435-023-01371-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40435-023-01371-7