Abstract
Introduction
Patients with chronic kidney disease (CKD) are vulnerable to adverse-drug events from cardiovascular drugs.
Aim
To evaluate awareness and knowledge for appropriate dose adjustment of cardiovascular drugs in CKD patients among Internal Medicine house-staff (IMHS).
Methods
Cross-sectional convenience sample survey in Fall 2015 among 341 IMHS from multiple academic institutions in the suburban New York City metropolitan area. Awareness was whether drug dose adjustment was needed. Knowledge was correct GFR level for drug dose adjustment. Multivariate logistic regression was conducted.
Results
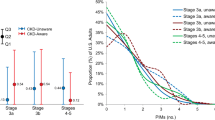
We found overall high percentages and high odds for all cardiovascular drugs for incorrect awareness and knowledge. Postgraduate year (PGY)-1 had greater odds than PGY-3 for Carvedilol (OR: 5.56, 95% CI: 2.19–14.12, p < 0.001) and Digoxin (OR: 3.87, 95% CI: 1.37–10.95, p < 0.05), and lesser odds than PGY3 for Atenolol (OR: 0.31, 95% CI: 0.10–0.91, p < 0.05). Nephrology exposure during medical school rotation, renal clinic, or family history had lesser odds for Carvedilol (OR: 0.45, 95% CI: 0.21–0.97, p < 0.05), Simvastatin (OR: 0.40, 95% CI: 0.16–0.97, p < 0.05), and Hydralazine (OR: 0.31, 95% CI: 0.12–0.81, p < 0.05). Nephrology exposure during residency (OR: 1.96, 95% CI: 1.10–3.50, p < 0.05) and US osteopathic graduates (OR: 2.40, 95% CI: 1.04–5.50, p < 0.05) each had greater odds for Enalapril (OR: 2.40, 95% CI: 1.04–5.50, p < 0.05). International medical graduates had lesser odds than US graduates for Amlodipine (OR: 0.30, 95% CI: 0.11–0.82, p < 0.05).
Conclusions
IMHS had overall poor awareness and knowledge for dose adjustment for common cardiovascular drugs in patients with CKD. As the majority of CKD patients are managed by their primary care providers, training programs should ensure that IMHS have adequate education in Nephrology during their residency training.
Similar content being viewed by others
References
Vrettos I, Voukelatou P, Katsoras A, Theotoka D, Kalliakmanis A. Diseases linked to polypharmacy in elderly patients. Curr Gerontol Geriatr Res. 2017;2017:4276047. https://doi.org/10.1155/2017/4276047.
Centers for Disease Control and Prevention. National Chronic Kidney Disease Fact Sheet, 2017. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention; 2017.
United States Renal Data System. 2018 USRDS annual data report: epidemiology of kidney disease in the United States. Bethesda: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2018.
Sutaria A, Liu L, Ahmed Z. Multiple medication (polypharmacy) and chronic kidney disease in patients aged 60 and older: a pharmacoepidemiologic perspective. Ther Adv Cardiovasc Dis. 2016;10(4):242–50.
Ardhanari S, Alpert MA, Aggarwal K. Cardiovascular disease in chronic kidney disease: risk factors, pathogenesis, and prevention. Adv Perit Dial. 2014;30:40–53.
Subbiah AK, Chhabra YK, Mahajan S. Cardiovascular disease in patients with chronic kidney disease: a neglected subgroup. Heart Asia. 2016;8(2):56–61.
Bajait C, Pimpalkhute S, Sontakke S, Jaiswal K, Dawri A. Prescribing pattern of medicines in chronic kidney disease with emphasis on phosphate binders. Indian J Pharmacol. 2014;46(1):35.
Centers for Disease Control and Prevention. Chronic kidney disease surveillance system—United States. http://www.cdc.gov/ckd.
Gholami K, Ziaie S, Shalviri G. Adverse drug reactions induced by cardiovascular drugs in outpatients. Pharm Pract (Granada). 2008;6(1):51–5.
Milton JC, Jackson SH. Inappropriate polypharmacy: reducing the burden of multiple medication. Clin Med. 2007;7(5):514–7.
Giardina C, Cutroneo PM, Mocciaro E, et al. Adverse drug reactions in hospitalized patients: results of the FORWARD (Facilitation of Reporting in Hospital Ward) Study. Front Pharmacol. 2018;9. https://doi.org/10.3389/fphar.2018.00350.
Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients a meta-analysis of prospective studies. JAMA. 1998;279(15):1200–5.
Classen DC, Pestotnik SL, Evans RS, Lloyd JF, Burke JP. Adverse drug events in hospitalized patients. Excess length of stay, extra costs, and attributable mortality. JAMA. 1997;277(4):301–6.
Johnson JA, Bootman JL. Drug-related morbidity and mortality. A cost-of-illness model. Arch Intern Med. 1995;155(18):1949–56.
Sakhuja A, Hyland J, Simon JF. Managing advanced chronic kidney disease: a primary care guide. CCJM. 2014;81(5):289–99.
Allen AS, Forman JP, Orav EJ, et al. Primary care management of chronic kidney disease. J Gen Intern Med. 2011;26:386. https://doi.org/10.1007/s11606-010-1523-6.
Desmedt S, Spinewine A, Jadoul M, et al. Impact of a clinical decision support system for drug dosage in patients with renal failure. Int J Clin Pharamacol. 2018;40:1225–33.
Dreischulte T, Guthrie B. High-risk prescribing and monitoring in primary care: how common is it, and how can it be improved? Ther Adv Drug Saf. 2012;3(4):175–84.
Surana S, Kumar N, Vasudeva A, et al. Awareness and knowledge among internal medicine house-staff for dose adjustment of commonly used medications in patients with CKD. BMC Nephrol. 2017;18(1):26.
Consumer Reports Health. Evaluating statin drugs to treat: high cholesterol and heart disease.https://article.images.consumerreports.org/prod/content/dam/cro/news_articles/health/PDFs/StatinsUpdate-FINAL.pdf. Accessed 12 June 2020.
Cannon CP, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. NEJM. 2004;350(15):1495–504.
Yancy C, Jessup M, Bozkurt B, et al. 2017 ACC/AFA/AFSA focused updated of the ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2017;136(6):e137-161.
Holmboe ES, Edgar L, Hamstra S. The milestones guidebook. Chicago: ACGME; 2016. http://www.acgme.org/Portals/0/MilestonesGuidebook.pdf.
DiNicolantonio JJ, Fares H, Niazi AK, et al. β-Blockers in hypertension, diabetes, heart failure and acute myocardial infarction: a review of the literature. Open Heart. 2015;2(1):e000230. https://doi.org/10.1136/openhrt-2014-000230.
Sawhney S, Fraser SD. Epidemiology of AKI: utilizing large databases to determine the burden of AKI. Adv Chronic Kidney Dis. 2017;24(4):194–204.
Townsend R., et al. Major side effects of angiotension-converting enzyme inhibitors and angiotensin II receptor blockers. UpToDate. 2018 Jun 27.
Wouters O, Kanavos P, McKee M, et al. Comparing generic drug markets in Europe and the United States: prices, volumes, and spending. Milbank Q. 2017;95(3):554–601. https://doi.org/10.1111/1468-0009.12279.
World Health Organization. Model list of essential medicines, 21st list, 2019. Geneva: World Health Organization; 2019. Licence: CC BY-NC-SA 3.0 IGO.
Fundamental Osteopathic Competencies. Guidelines for osteopathic medical licensure and the practice of osteopathic medicine. Chicago: National Board of Osteopathic Medical Examiners; 2009.
Accreditation Council for Graduate Medical Education. ACGME data resource book academic year 2017–2018. Chicago: Accreditation Council for Graduate Medical Education; 2018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None for all authors.
Conflicts of interest/Competing interests
None declared for all authors.
Ethics approval
Study approved by local IRB.
Consent to participate
All subjects in the study gave informed consent.
Consent for publication
All authors approve the manuscript for publication.
Availability of data and material
Not applicable. Not able to share the data at present as we still in the process of doing sub-group analyses.
Code availability
Not applicable.
Author’s contributions
KD interpreted the data and drafted the manuscript for important intellectual content. SP gathered the data and drafted the manuscript for important intellectual content. FR interpreted the data and drafted the manuscript for important intellectual content. JF designed the study, analyzed the data, interpreted the data, and critically reviewed and revised the manuscript for important intellectual content. DC designed the study, interpreted the data, and critically reviewed and revised the manuscript for important intellectual content. SR conceptualized and designed the study, gathered the data, interpreted the data, and critically reviewed and revised the manuscript for important intellectual content.
Rights and permissions
About this article
Cite this article
D’Angelo, K., Paul, S., Ranjeeta, F. et al. Awareness and Knowledge Among Internal Medicine House-Staff for Dose Adjustment of Cardiovascular Drugs in Chronic Kidney Disease. High Blood Press Cardiovasc Prev 28, 177–184 (2021). https://doi.org/10.1007/s40292-021-00438-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-021-00438-w