Abstract
Background
Considerable evidence on the costs and cost-effectiveness of biomedical, non-surgical interventions to prevent human immunodeficiency virus (HIV) transmission has been generated over the last decade. This study aims to synthesize findings and identify remaining knowledge gaps to suggest future research priorities.
Methods
A systematic literature review was carried out in August 2020 using the MEDLINE, Embase, Global Health and EconLit databases to retrieve economic evaluations and costing studies of oral pre-exposure prophylaxis (PrEP), injectable long-acting PrEP, vaginal microbicide rings and gels, HIV vaccines and broadly neutralizing antibodies. Studies reporting costs from the provider or societal perspective were included in the analysis. Those reporting on behavioural methods of prevention, condoms and surgical approaches (voluntary medical male circumcision) were excluded. The quality of reporting of the included studies was assessed using published checklists.
Results
We identified 3007 citations, of which 87 studies were retained. Most were set in low- and middle-income countries (LMICs; n = 53) and focused on the costs and/or cost-effectiveness of oral PrEP regimens (n = 70). Model-based economic evaluations were the most frequent study design; only two trial-based cost-effectiveness analyses and nine costing studies were found. Less than half of the studies provided practical details on how the intervention would be delivered by the health system, and only three of these, all in LMICs, explicitly focused on service integration and its implication for delivery costs. ‘Real-world’ programme delivery mechanisms and costs of intervention delivery were rarely considered. PrEP technologies were generally found to be cost-effective only when targeting high-risk subpopulations. Single-dose HIV vaccines are expected to be cost-effective for all groups despite substantial uncertainty around pricing.
Conclusions
A lack of primary, detailed and updated cost data, including above-service level costs, from a variety of settings makes it difficult to evaluate the cost-effectiveness of specific delivery modes at scale, or to evaluate strategies for services integration. Closing this evidence gap around real-world implementation is vital, not least because the strategies targeting high-risk groups that are recommended by PrEP models may incur substantially higher costs and be of limited practical feasibility in some settings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Our literature review identified a large volume of literature on the costs and cost-effectiveness of biomedical, non-surgical interventions to prevent human immunodeficiency virus (HIV) transmission. Most literature focuses on oral PrEP regimens, is set in low- and middle-income countries and largely covers interventions targeting the general population. |
PrEP technologies were generally found to be cost-effective only when targeting high-risk subpopulations, although relatively little evidence is available on more recently developed technologies. Single-dose HIV vaccines are expected to be cost-effective for all groups despite substantial uncertainty around pricing. |
A lack of primary, detailed and updated cost data, including above service-level costs, from a variety of settings makes it difficult to evaluate the cost-effectiveness of specific delivery modes at scale, or to evaluate strategies for services integration. To effectively assist priority setting, more practical detail is needed on how modelled interventions would be implemented by the health system. |
1 Background
The human immunodeficiency virus (HIV) epidemic continues to cause extensive morbidity and mortality globally. Despite global progress towards reaching the 90–90–90 targets, only 87% of people living with HIV who knew their status accessed antiretroviral therapy (ART) in 2020 [1], and gaps in HIV prevention and treatment contributed to 1.5 million new infections and 690,000 HIV-related deaths [2].
With the exception of voluntary medical male circumcision, the existing arsenal of biomedical HIV prevention tools, which includes oral pre-exposure prophylaxis (PrEP), condoms and medication-assisted treatment for people who inject drugs (PWIDs), requires frequent usage, which contributes to implementation challenges and high recurrent costs. Forthcoming products that offer longer duration of protection are poised to have a significant impact on HIV prevention efforts. For example, the dapivirine vaginal ring for women received a positive opinion from the European Medicines Agency as a monthly prevention option [3] and has been recommended by the World Health Organization (WHO) as a new choice for HIV pevention for women at substantial risk of HIV infection [4]. Phase III clinical trials showed that the ring reduced HIV infection risk by approximately 30% overall [5]. Additionally, long-acting PrEP with cabotegravir administered as a bi-monthly injection was found to be 89% more effective than oral PrEP at preventing HIV acquisition [6], and has been approved by the US FDA [7]. Furthermore, several HIV prevention monoclonal antibody candidates are currently advancing through clinical development [8,9,10], and a number of putatively more potent combinations and engineered antibodies are also undergoing early clinical evaluation [11].
Information on the costs and cost-effectiveness of new HIV prevention interventions is required to inform priority setting, particularly drawing from and reflecting on ‘real world’ implementation. Funding for HIV programmes is not growing, which results in mounting pressure to improve the efficiency of HIV spending, for example by promoting the integration of HIV service delivery with other health services [12, 13]. Moreover, gaining insight into the drivers of cost and cost-effectiveness can help shape key choices in the early development of biomedical HIV prevention products that may have long-term implications for affordability and uptake, including pricing strategies, dosing regimens, coadministration strategies and delivery approaches.
Therefore, the aim of this literature review was to collate evidence on the costs and cost-effectiveness of biomedical, non-surgical HIV prevention interventions, to identify gaps and suggest future research priorities.
2 Methods
A systematic literature review was carried out using the Medline, Embase, Global Health, EconLit and Web of Science databases between 26 and 27 August 2020. The Cochrane Database of systematic reviews was also searched to retrieve other reviews on the same topic and hand search the reference lists of any relevant records. Searches were limited to full texts published after 2010, written in English and focusing on human subjects. The search strategy combined keywords on human immunodeficiency virus (HIV) and acquired immunodeficiency syndrome (AIDS), HIV prevention interventions and economic evaluation. Published cost and cost-effectiveness filters were used for MEDLINE and adapted for other databases [14]. The full search strategies and number of records retrieved for each database are reported in the electronic supplementary material (ESM). Reporting follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and checklist [15].
2.1 Screening, Data Extraction and Analysis
The abstract and titles of all unique records were screened and articles were further excluded based on the following criteria: (1) language other than English; (2) topic not related to human health; (3) no reference to HIV prevention interventions; (4) no reference to cost, cost-effectiveness, efficiency or priority setting models; and (5) ineligible article types (e.g. conference abstracts, comments, letters, review articles, methods papers). The full texts of the remaining articles were then reviewed and retained if they made reference to biomedical health products or technologies for preventing HIV infection in uninfected individuals (e.g. pre-exposure prophylaxis, or ‘PrEP’, or HIV vaccination). Behavioural methods of prevention, condoms and surgical approaches (voluntary medical male circumcision) were excluded. Articles reporting costs exclusively from the perspective of patients were also excluded.
Data were extracted from the retained records in the following categories: study setting; objective; population and demographics of interest; intervention and comparator; type of economic analysis, including perspective, cost analysis methods, data sources and types of costs; intervention delivery method (stand-alone or integrated) including any shared costs; type of model, if any, including disease transmission model characteristics where applicable; main results and sources of uncertainty. The data were summarized using descriptive statistics and narratively following thematic analysis of the contents of the articles.
The quality of included studies was assessed using the Global Health Costing Consortium (GHCC) reference case for studies that have a substantial primary cost data collection component [16], and the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist for economic evaluation studies [17]. While the GHCC reference case sets out methodological standards that costing studies should adhere to, the CHEERS checklist lays out reporting standards and is not intended to assess methodological quality.
3 Results
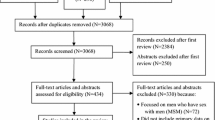
We identified 3007 citations, of which approximately 1 in 20 were eligible for full-text screening. The PRISMA flowchart with details of the study screening and selection process is shown in Fig. 1. After the selection process was completed, 87 studies were retained for analysis. All studies meeting the inclusion criteria were included in the review, regardless of quality assessment score. The main study characteristics are summarized in the ESM.
3.1 Study Characteristics
3.1.1 Setting
Approximately two-thirds of the studies (n = 53) focused on low- or middle-income countries (LMICs), with South Africa (n = 27) and Kenya (n = 9) being the countries most represented in the sample. Two of the largest PrEP demonstration projects were also based in these countries [18, 19]. Thirty-four studies analyzed data from high-income countries (HICs) and two studies were global. The majority of studies focusing on specific countries analyzed national-level data, while approximately one in four studies focused on individual regions, districts or urban areas. Countries in all UNAIDS regions were individually represented in the sample as follows: Eastern and Southern Africa (21%, n = 18), Western and Central Europe and North America (18%, n = 16), Asia and the Pacific (n = 6), Latin America and the Caribbean (2%, n = 2), Western and Central Africa (2%, n = 2), Eastern Europe and Central Asia (1%, n = 1), and Middle East and North Africa (1%, n = 1).
3.1.2 Study Populations
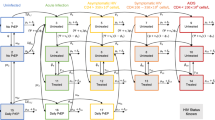
Most studies analyzed interventions for the general population (n = 49). Studies focusing on key populations overwhelmingly evaluated interventions targeting men who have sex with men (MSM; n = 38), nine studies focused on female sex workers (FSW), and eight studies focused on PWIDs. Some of the studies (n = 17) compared the costs or cost-effectiveness of interventions across different populations or subgroups, such as adults versus adolescents/young adults or high-risk groups within a subpopulation. A breakdown of study populations investigated in the most represented geographical regions is presented in Fig. 2.
3.1.3 Interventions and Comparators
The interventions analyzed in the retrieved records are also summarized in Fig. 2, stratified by study population. Eighty percent of retrieved studies (n = 70) focused on the costs and/or cost-effectiveness of oral PrEP regimens, mostly on their own [18, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61] rather than in combination with other prevention interventions such as condom use, treatment as prevention, and HIV counselling and testing [35, 62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86]. All oral PrEP studies examined emtricitabine/tenofovir disoproxil fumarate regimens, whether branded or generic, except for one paper comparing this regimen with the newer emtricitabine/tenofovir alafenamide [58]. The most common comparator for cost-effectiveness studies was the status quo or no intervention. A few studies compared the relative cost-effectiveness of PrEP between subpopulations (e.g. all adults) and high-risk groups (e.g. FSWs only). Another common comparison was the relative cost-effectiveness of daily PrEP and event-driven (‘on-demand’) PrEP, either limited to high-risk sexual encounters or around specific time periods, such as pregnancy and breastfeeding or as a bridge to ART for HIV-infected partners in serodiscordant couples.
Eleven studies investigated the cost-effectiveness of HIV vaccines compared with no intervention [37, 82, 87,88,89,90]; comparing different vaccination strategies (e.g. one-off versus revaccination strategies, different target populations) [91,92,93]; or comparing HIV vaccination with oral PrEP [62, 94].
Fewer records retrieved focused on non-oral forms of PrEP, including six studies on microbicide gel [76, 80, 95,96,97,98] and four studies each on long-acting injectable PrEP [80, 82, 99, 100] and vaginal rings [80,81,82, 101]. Only one study reported results on broadly neutralizing HIV-1 antibodies (bNABs), a promising novel approach to prevention that engages with the host immune response to neutralize free virus, clear infected cells and inhibit cell-to-cell transmission of HIV-1 [82]. All of these studies compared the new prevention technologies, alone or in combination with other interventions, to current standards of preventive care, likely reflecting the early stage of development of the technologies and thus the exploratory nature of the research questions.
3.1.4 Intervention Design
Less than half of the studies (43%) provided practical details on the platforms through which the intervention of interest would be delivered, either through routine public health services, pharmacies or other distribution systems, and only three of these explicitly focused on service integration and its implication for delivery costs [45, 52, 100].
Among the studies that explicitly mentioned delivery modes, 11 stated that the intervention was delivered through a stand-alone vertical programme, with dedicated resources (diagnostics, staff, program management) but likely assuming no shared costs; 10 studies described interventions delivered through routine HIV services; 7 studies reported on interventions integrated with an existing HIV service (e.g. a vaginal microbicide programme integrated with a condom distribution programme); and another 10 studies reported on interventions integrated with other non-HIV health services (e.g. HIV vaccine delivered through the expanded programme on immunization, or long-acting injectable PrEP delivered during family planning visits for injectable contraception).
3.2 Study Methods
3.2.1 Cost and Cost-Effectiveness Analyses
A small number of studies carried out cost analyses (10%) only, while the majority were economic evaluations. Of these, more than half were cost-utility analyses, using either cost per quality-adjusted life-year (QALY) gained or per disability-adjusted life-year (DALY) averted as outcome measures. Approximately 30% were cost-effectiveness analyses, most commonly reporting cost per infection averted or per life-year gained. The majority of economic evaluations that used cost-effectiveness thresholds to establish whether the interventions represented good value for money used gross domestic product-based thresholds proposed by the WHO.
Lastly, 10% of the studies sought to optimize resource allocation or intervention scale-up for a given budget. Common objectives of these studies included maximizing intervention impact and/or coverage under a set of budget constraints.
3.2.2 Model-Based and Trial-Based Analyses
Only two of the studies presented the results of trial-based cost-effectiveness analyses [26, 46]. The majority of studies (83%) were instead based on mathematical models of disease transmission. The three main transmission model types—deterministic compartmental models, stochastic models and agent-based simulations—were present. No pattern was observed in terms of the chosen mathematical model structure for specific interventions, with different structures being used interchangeably to accommodate different technologies and answer diverse study questions (Fig. 3).
Thirty-three (44%) of the transmission model-based analyses were pre-trial analyses that posed exploratory strategic questions (e.g., is oral PrEP cost-effective in a given population?), while the remainder addressed more tactical questions of relevance once the efficacy of the technology has been demonstrated (e.g. which population groups should be targeted during scale-up? What is the most cost-effective way of delivering HIV vaccines?). The most common time horizons for the models were in the 10- to 20-year range (62%), and 21% of the models projected costs and outcomes over a lifetime horizon.
3.2.3 Costing Methods
The vast majority of studies took a provider or payer perspective (86%) and calculated economic costs, taking opportunity costs into account (90%) rather than financial costs. Only approximately 30% of studies mentioned the inclusion of above service-level costs, defined as the costs of support services provided by central administrations such as training, laboratory services or sensitization campaigns.
Only nine studies (10%) in the sample collected primary data, seven from bottom-up microcosting exercises [18, 26, 31, 44, 46, 71, 86] and two allocating programme costs using top-down gross costing methods [27, 52]. Most studies (93%) used secondary cost data sources to parameterize economic models, including published and grey literature, public procurement price lists and other national-level routine sources such as insurance reimbursement prices (particularly in HICs), and ‘off-the shelf’ estimates such as the Lives Saved Tool [82]. The remaining studies either made assumptions about the prices of novel drugs or technologies, or validated assumptions or costs extrapolated from the literature through key informant/expert consultations.
Lastly, 10 studies modelled the optimal costs of the intervention or programme of interest for it to achieve a certain objective, such as remaining cost-effective or optimizing resource allocation under a budget constraint [20, 22, 61, 65, 66, 73, 87, 89, 90, 93].
3.2.4 Quality Assessment
The quality of studies collecting primary cost data was high overall, with an average score of 80% based on the criteria laid out in the GHCC reference case. Overall, the reference case principles with the lowest average scores were #14 (reporting on the use of shadow prices), #15 (reporting variation in costs by size and subpopulation) and #16 (uncertainty is appropriately characterized).
The quality of economic evaluations, assessed using the CHEERS checklist, was also good overall, with an average score of 70%. The criteria with the lowest average score were #13 and #14 (supplying details of resources and costs estimation and currency conversion), and #18 (reporting values, ranges, references and, if used, probability distributions of all study parameters).
3.3 Study Findings
3.3.1 Oral PrEP
Daily oral PrEP was generally found to be costly, with the potential to become cost-effective only at lower or generic prices, or when targeting high-risk groups, although findings were not conclusive across settings and subpopulations. In settings with generalized HIV epidemics, with sustained transmission in the general population, PrEP programmes targeting the general population would not be included in an optimal package of prevention interventions, which would rather favour scaling-up existing interventions such as universal or early ART and voluntary medical male circumcision [20, 21, 48, 64, 76, 77, 79, 82, 102].
Findings among subpopulations are less uniform. Several studies across both high- and low-income settings found that daily oral PrEP can be cost-effective when high-risk population groups are targeted, while uniform scale-up to the entire population (or subpopulation) is usually not cost-effective [34, 39, 42, 59, 65, 103]. For example, MacFadden et al. found an incremental cost-effectiveness ratio (ICER) of between US$640,000 and US$1,025,600 per QALY gained by introducing oral PrEP for all MSM in Toronto, Canada, while targeting the highest-risk decile yielded an ICER of US$45,000–90,000 per QALY [39]. Similar findings were reported by Nichols et al. in Macha, Zambia, where targeting the highest sexual activity groups in the population was found to be highly cost-effective compared with no targeting of oral PrEP [42]. Jamieson et al. found that in South Africa, self-selection of higher-risk individuals in subpopulations (adolescents, young adults) can be up to 10 times more cost-effective compared with targeting the entire sub population to receive daily oral PrEP [32].
Oral PrEP was also found to be cost-effective among young women and FSWs in LMICs [48, 80, 97]. However, there is evidence that it might not be cost-effective in all high-risk groups and across settings. Roberts et al. report that the costs of daily oral PrEP delivery targeting young women could be up to 40% lower when the intervention is integrated into the routine family planning services provided at Kenyan government clinics [52]. Among PWIDs, adding PrEP to existing interventions such as methadone maintenance therapy, needles and syringe exchange programmes, naloxone and addiction counselling was found to not be cost-effective at current PrEP drug prices in the USA and Ukraine [36, 63, 67, 68, 84], while one study found that daily oral PrEP coupled with increased frequency of HIV testing (every 6 months) among PWIDs in India was cost-effective at a reference price negotiated with manufacturers [35].
Among MSM, several studies in high- and upper middle-income settings found daily oral PrEP was not cost-effective at current prices [22, 24, 26, 27, 40, 46, 50, 55, 60, 73, 75]. Two stochastic modelling analyses, one with a global scope and one focusing on the USA, found that in addition to high prices, PrEP among MSM is not cost-effective at low levels of adherence (defined as efficacy <92%, as observed in clinical trials) and low background levels of HIV prevalence [24, 40]. Similarly, studies from Lima, Peru and Hong Kong found that PrEP for MSM is not cost-effective at coverage levels currently attainable within the budget envelope [28, 60]. Studies from The Netherlands and San Francisco, USA, respectively, found that PrEP for MSM is more likely to be cost-effective in the absence of risk compensation (the increase in risk-taking behaviour triggered by a decrease in perceived risk) [50], and when background ART coverage is low [54]. In contrast, other studies found that PrEP is cost-effective among MSM in The Netherlands [43] and England [45] at efficacy levels between 65 and 80%; among MSM and transgender women in Brazil [38]; and among transgender FSWs and their clients in Lima, Peru [69].
Among serodiscordant couples, the general consensus in the literature is that due to the high cost of PrEP drugs, PrEP for the uninfected partner is generally not cost-effective compared with early ART for the HIV-infected partner [29, 72, 104], although a study from Kampala, Uganda, concluded that PrEP in serodiscordant couples as a bridge to sustained ART use for the HIV-infected partner is slightly more cost-effective (US$1340 per DALY averted) than scaling up ART for the whole HIV-infected population (US$1452 per DALY averted) [86]. Similarly, Schneider et al. found that oral PrEP is only cost-effective when targeting MSM in serodiscordant partnerships in Australia, compared with targeting 10–30% of all MSM or all high-risk MSM [53]. Other time-limited uses of PrEP, such as during pregnancy and breastfeeding [49] and sexual event-driven PrEP, were found to be cost-effective both in the general population and among MSM in most high- and upper middle-income countries, where PrEP prices are higher [27, 41, 43, 51], as well as among the partners of migrant miners during home visits in Mozambique [25]. No studies of PrEP in paediatric populations were retrieved.
3.3.2 Other Forms of Pre-Exposure Prophylaxis (PrEP) and Broadly Neutralizing HIV-1 Antibodies (bNABs)
Evidence on other forms of PrEP, including dapirivine-containing intravaginal rings, tenofovir-based vaginal microbicides, injectable long-acting PrEP (rilpirivine) and bNABs, also found that they are unlikely to be cost-effective outside of specific high-risk groups. A study by Smith et al. seeking to identify an optimal package of HIV prevention interventions for the South African population that includes both existing interventions (oral PrEP, condoms, voluntary medical male circumcision, early ART) and new interventions (vaginal microbicides/rings, bNABs, long-acting antiretrovirals, HIV vaccine) concluded that a cost-effective package would include all established technologies with the addition of a vaccine, while the newer PrEP technologies would be excluded unless their price could be lowered [82].
However, other studies from South Africa found that targeting specific risk groups could be cost-effective. Evidence on long-acting injectable PrEP in South Africa, for example, concludes that it would be cost-effective for young women and FSWs [80, 99, 105] but not for heterosexual men [102]. Another study assessing the use of injectable antiretrovirals among long-acting contraceptive users attending family planning clinics in Limpopo province, South Africa, found that despite the economic benefit of service integration, the intervention would not be cost-effective unless the price of PrEP decreased by 16% and coverage was sustained at 85% [100].
Early models exploring the cost-effectiveness of vaginal microbicide gels concluded that they had the potential to be cost-effective for women of reproductive age in a generalized epidemic setting such as in South Africa [97, 98], but not in the USA, even in areas with higher relative HIV prevalence such as Washington, DC [96]. Terris-Prestholt et al. calculated that tenofovir gel could be cost-effective for women in Gauteng Province, South Africa, at a maximum price of US$0.12 per dose, which could be increased to more realistic threshold prices (US$0.25 or US$0.33, respectively) if administered as a single dose or if highly effective [95]. Dapirivine-containing intravaginal rings could also be cost-effective among young women and other high-risk groups such as FSWs and women with multiple sexual partners in South Africa [81, 101].
3.3.3 HIV Vaccines
HIV vaccines are generally expected to achieve high impact even if imperfectly efficacious (i.e., allowing for breakthrough infections among vaccinated individuals), although much uncertainty remains around costs. The three retrieved studies focusing on an HIC (USA) found that the vaccine would be highly cost-effective among MSM compared with oral PrEP [62, 94], but even a single dose regimen would not be cost-effective compared with no intervention in the general population, and repeated booster doses every 3 years would make it no longer cost-effective even among MSM and PWIDs [93].
Six studies focused on vaccines in LMICs. Modelling evidence from these concluded that a vaccine administered to the reproductive-age population of LMICs would be cost-effective under most coverage, product profile and efficacy scenarios except for very high prices and a 3-year duration of protection [87]. A study from Thailand, modelling a prime-boost combination of the LVAC-HIV and AIDSVAX B/E vaccines, found that the vaccine would remain cost-effective at a high price per dose ($353) with 70% efficacy, lifetime protection and no change in post-vaccination risk behaviour, but would not be cost-effective at any price if efficacy was low (30%) and the vaccine induced even a relatively small increase (10%) in risk behaviour [37]. Evidence from South Africa suggested that a partially effective and rapidly waning vaccine could still be cost-effective under targeted delivery to age groups with the highest HIV incidence [90], and that campaign-based vaccination is consistently less costly and more effective than routine vaccine delivery [88, 89, 91].
4 Discussion
This systematic review summarized data from a large number of studies on the costs and cost-effectiveness of non-surgical, biomedical interventions to prevent HIV. Despite the substantial amount of literature retrieved, relatively few data were found on newer preventive technologies such as injectable long-acting PrEP, vaginal rings and bNABs. Moreover, the cost and cost-effectiveness evidence on these preventive technologies is limited in geographical scope, with most studies focusing on a single LMIC (South Africa), a consideration that also applies to the literature on HIV vaccines.
While the evidence seems to point towards high costs and limited efficacy in the general population, most forms of PrEP were demonstrated to be cost-effective in high-risk population subgroups and/or for specific use cases (e.g. sexual event-based use of oral PrEP, or as a bridge to sustained ART use in serodiscordant couples). These findings are in line with existing literature reviews which conclude that despite being cost-effective among selected key populations, oral PrEP could be less cost-effective than expanding existing prevention interventions [106, 107]. However, as the range of PrEP products with different durations of protection expands, greater analytical nuance will be required to understand how risk varies over time and its effect on cost- effectiveness. Different products may be more cost-effective at different points in an individual’s life.
For some of the technologies, the evidence is too scarce to draw firm conclusions across settings and populations. In particular, fewer than 10 studies each were retrieved on injectable PrEP, intravaginal rings and microbicide gels, and only one study on bNABs. More evidence should be generated comparing intervention strategies that include these modern technologies, focusing on head-to-head comparisons in different settings and for different delivery options. Furthermore, the gross domestic product-based cost-effectiveness thresholds used in the majority of published economic evaluation studies are outdated, as it is now recommended that analysts make use of country-specific opportuity cost-based thresholds [108]. More generally, cost-effectiveness thresholds, particuarly early in the life of a product, need to be interpreted with caution. Prices of active ingredients decrease as patents expire; cost and cost-effectiveness research should present findings in a disaggregated manner, allowing for the recalculation of costs and ICERs as prices change.
Substantial uncertainty remains around the price of preventive technologies, their efficacy in the event of less than optimal uptake and adherence (including vaccine hesitancy) or risk compensation in the form of lower condom use. Background rates of HIV prevalence and ART coverage, as well as duration of protection for HIV vaccines, were also found to drive uncertainty in cost-effectiveness estimates. While several model-based analyses conduct threshold analyses to define a price for the technologies to remain cost-effective, the majority of cost parameters in the models are based on secondary estimates or assumptions that are often not comprehensively listed, as revealed by the quality assessments of the included studies. The most frequently cited secondary sources of cost data on intervention delivery include PrEP demonstration projects conducted in South Africa, Kenya and Uganda, not all of them published and often extrapolated to other contexts without making the methods explicit, as well as a few other costing studies from the mid-2000s and early 2010s.
Our sample included relatively few primary costing studies or model-based analyses parameterized with primary data. Moreover, fewer than 15% of studies took a societal perspective and fewer than 30% mentioned the inclusion of above service-level costs. Collecting and reporting comprehensive cost data, including the costs of above service-level activities is particularly important in the case of interventions targeting specific population groups that are often harder to reach [109]. Excluding the additional spending that is necessary to expand coverage might lead to overestimating the cost-effectiveness of the intervention. Interestingly, the only study comparing existing and newer forms of PrEP and HIV vaccines in a range of populations, and also the only study that builds a cost function to model scale-up costs, concludes that while newer technologies might not be cost-effective in an ideal scale-up scenario, they might be included in the optimal package if health system constraints to the expansion of existing interventions (voluntary medical male circumcision, early ART) were taken into account [82]. In this case, for an equivalent benefit, the additional costs of increasing coverage of existing interventions among harder-to-reach groups may outweigh the costs of newer preventive technologies.
Only a small proportion of retrieved studies detailed the delivery mechanism of the interventions, specifying whether these were integrated with other services or stand-alone. Service integration is an area that deserves increased attention, given the emphasis on its potential for improving efficiency. Evidence from empirical studies of integration at the service delivery level is currently mixed [110], particularly from LMICs where health systems are overstretched, and adding new services onto existing platforms that are facing input constraints might actually reduce rather than boost efficiency [111]. A meta-analysis of the effects of HIV services integration concluded that uptake and retention improved with integration for both HIV and non-HIV services, although no difference in treatment outcomes was detected because of an insufficient number of eligible studies [112]. A recent study of oral PrEP in Zimbabwe further demonstrated that delivery modes that improve continuation rates achieve lower unit costs per person-year [113]. More updated evidence that also captures ‘real-world’ delivery modes and any health system constraints affecting the feasibility of implementation and scale-up is therefore essential for informing priority setting [106].
Modelling studies should also acknowledge the acceptability and equity implications of suggesting PrEP options be made available only to certain population groups. Beyond the equity implications of restricting access to prevention for at-risk individuals that fall outside defined risk groups, there is also evidence that expanding access may ultimately reduce costs and improve cost-effectiveness by substantially reducing new infections in HIV-endemic countries [114].
5 Conclusions
Evidence on the cost-effectiveness of biomedical, non-surgical HIV prevention interventions rests largely on transmission model-based analyses that to date have concentrated on oral PrEP while overlooking more modern prevention technologies, including long-acting injectable PrEP, intravaginal rings and bNABs. The evidence seems to suggest that given the relatively high anticipated costs of these drugs and devices compared with established interventions such as voluntary medical male circumcision and early ART, these interventions might be cost-effective only in higher-risk population subgroups and/or for specific time-limited use cases. However, the scenarios modelled do not include head-to-head comparisons of the more recently developed interventions, and studies are not representative of a wide variety of settings and are rarely explicit about delivery strategies.
More generally, the ‘real world’ mechanisms and costs of intervention delivery are rarely considered or made explicit in the papers analysed in this review. This is an important gap given that reaching some of the high-risk groups that made the delivery of different forms of PrEP cost-effective in model-based analyses may incur substantial additional costs in day-to-day implementation and may, in addition, be of limited practical feasibility in some settings. Similarly, the general lack of primary, detailed and updated cost data from a wider range of settings, including above service-level costs, makes it difficult to evaluate the cost-effectiveness of specific delivery modes at scale, as well as to evaluate strategies for services integration.
References
UNAIDS. UNAIDS data 2021. Available at: https://www.unaids.org/en/resources/documents/2021/2021_unaids_data.
WHO. HIV/AIDS Factsheet. Available at: https://www.who.int/en/news-room/fact-sheets/detail/hiv-aids.
EMA. Vaginal ring to reduce the risk of HIV infection for women in non-EU countries with high disease burden. 2020. Available at: https://www.ema.europa.eu/en/news/vaginal-ring-reduce-risk-hiv-infection-women-non-eu-countries-high-disease-burden.
WHO. WHO recommends the dapirivine vaginal ring as a new choice for HIV prevention for women at substantial risk of HIV infection. 2021. Available at: https://www.who.int/news/item/26-01-2021-who-recommends-the-dapivirine-vaginal-ring-as-a-new-choice-for-hiv-prevention-for-women-at-substantial-risk-of-hiv-infection.
Baeten JM, Palanee-Phillips T, Brown ER, Schwartz K, Soto-Torres LE, Govender V, et al. Use of a vaginal ring containing dapivirine for HIV-1 prevention in women. N Engl J Med. 2016;375(22):2121–32.
Delany-Moretlwe S, Hughes JP, Bock P, Ouma SG, Hunidzarira P, Kalonji D, et al. Cabotegravir for the prevention of HIV-1 in women: results from HPTN 084, a phase 3, randomised clinical trial. Lancet. 2022;399(10337):1779–89.
FDA. FDA Approves First Injectable Treatment for HIV Pre-Exposure Prevention. 2021. Available at: https://www.fda.gov/news-events/press-announcements/fda-approves-first-injectable-treatment-hiv-pre-exposure-prevention.
ClinicalTrials.gov. Evaluating the Safety and Efficacy of the VRC01 Antibody in Reducing Acquisition of HIV-1 Infection Among Men and Transgender Persons Who Have Sex With Men. Available at: https://clinicaltrials.gov/ct2/show/NCT02716675.
ClinicalTrials.gov. Evaluating the Safety and Efficacy of the VRC01 Antibody in Reducing Acquisition of HIV-1 Infection in Women. Available at: https://clinicaltrials.gov/ct2/show/NCT02568215.
Mgodi NM, Takuva S, Edupuganti S, Karuna S, Andrew P, Lazarus E, et al. A phase 2b study to evaluate the safety and efficacy of VRC01 broadly neutralizing monoclonal antibody in reducing acquisition of HIV-1 infection in women in sub-saharan africa: baseline findings. J Acquir Immune Defic Syndr. 2021;87(1):680–7.
AVAC. The rise of broadly neutralizing antibodies. 2018. Available at: https://www.avac.org/blog/rise-broadly-neutralizing-antibodies.
De Lay PR, Benkazen A, Abdool Karim Q, Aliyu A, Amole C, Ayala G, et al. Ending AIDS as a public health threat by 2030: time to reset targets for 2025. PLoS Med. 2021;18(6): e1003649.
UNAIDS. Delivering on SDG3: Strengthening and integrating comprehensive HIV responses into sustainable health systems for Universal Health Care. Geneva: UNAIDS; 13 June 2019.
CADTH. CADTH's database search filters 2019 [updated 29 April 2019]. Available at: https://www.cadth.ca/resources/finding-evidence/strings-attached-cadths-database-search-filters#health.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6(7): e1000100.
Vassall A, Sweeney S, Kahn J, Gomez GB, Bollinger L, Marseille E, et al. Reference case for estimating the costs of global health services and interventions. GHCC; 2017.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated health economic evaluation reporting standards (CHEERS) statement. Cost Eff Resour Alloc. 2013;11(1):6.
Eakle R, Gomez GB, Naicker N, Bothma R, Mbogua J, Escobar MAC, et al. HIV pre-exposure prophylaxis and early antiretroviral treatment among female sex workers in South Africa: Results from a prospective observational demonstration project. PLoS Med. 2017;14:11.
Phanitsiri S, Kiragu M, Nnakabonge I, Wachichi C, Tagar E, Kimani P, et al., editors. The unit cost of delivering oral PrEP as part of a combination HIV prevention package: results from the IPCP demonstration project in Kenya. 19th International Conference on AIDS and STIs in Africa; 2017; Abidjan, Cote d'Ivoire.
Akudibillah G, Pandey A, Medlock J. Maximizing the benefits of ART and PrEP in resource-limited settings. Epidemiol Infect. 2017;145(5):942–56.
Alistar SS, Grant PM, Bendavid E. Comparative effectiveness and cost-effectiveness of antiretroviral therapy and pre-exposure prophylaxis for HIV prevention in South Africa. BMC Med. 2014;12:11.
Cambiano V, Miners A, Dunn D, McCormack S, Ong KJ, Gill ON, et al. Cost-effectiveness of pre-exposure prophylaxis for HIV prevention in men who have sex with men in the UK: a modelling study and health economic evaluation. Lancet Infect Dis. 2018;18(1):85–94.
Chan SS, Chappel AR, Maddox KEJ, Hoover KW, Huang YLA, Zhu WM, et al. Pre-exposure prophylaxis for preventing acquisition of HIV: A cross-sectional study of patients, prescribers, uptake, and spending in the United States, 2015–2016. PLoS Med. 2020;17(4):17.
Chen A, Dowdy DW. Clinical effectiveness and cost-effectiveness of hiv preexposure prophylaxis in men who have sex with men: Risk calculators for real-world decision-making. PLoS ONE. 2014;9:10.
Cremin I, Morales F, Jewell BL, O’Reilly KR, Hallett TB. Seasonal PrEP for partners of migrant miners in southern Mozambique: A highly focused PrEP intervention. J Int AIDS Soc. 2015;18(Suppl):3.
Durand-Zaleski I, Mutuon P, Charreau I, Tremblay C, Rojas D, Pialoux G, et al. Costs and benefits of on-demand HIV preexposure prophylaxis in MSM. AIDS. 2018;32(1):95–102.
Ginsberg GM, Chemtob D. Cost utility analysis of HIV pre exposure prophylaxis among men who have sex with men in Israel. BMC Public Health. 2020;20(1):14.
Gomez GB, Borquez A, Caceres CF, Segura ER, Grant RM, Garnett GP, et al. The potential impact of pre-exposure prophylaxis for HIV prevention among men who have sex with men and transwomen in Lima, Peru: a mathematical modelling study. PLoS Med. 2012;9(10):15.
Hallett TB, Baeten JM, Heffron R, Barnabas R, de Bruyn G, Cremin I, et al. Optimal uses of antiretrovirals for prevention in HIV-1 serodiscordant heterosexual couples in South Africa: a modelling study. PLoS Med. 2011;8(11):12.
Horberg M, Raymond B. Financial policy issues for HIV pre-exposure prophylaxis cost and access to insurance. Am J Prev Med. 2013;44(1):S125–8.
Irungu EM, Sharma M, Maronga C, Mugo N, Ngure K, Celum C, et al. The incremental cost of delivering PrEP as a bridge to art for HIV serodiscordant couples in public HIV care clinics in Kenya. Aids Research and Treatment. 2019;2019:8.
Jamieson L, Gomez GB, Rebe K, Subedar H, Jenkins S, Shoko N, et al. The impact of self-selection based on HIV risk on the cost-effectiveness of pre-exposure prophylaxis in South Africa. AIDS. 2020;34(6):883–91.
Jewell BL, Cremin I, Pickles M, Celum C, Baeten JM, Delany-Moretlwe S, et al. Estimating the cost-effectiveness of pre-exposure prophylaxis to reduce HIV-1 and HSV-2 incidence in HIV-serodiscordant couples in South Africa. PLoS ONE. 2015;10(1): e0115511.
Juusola JL, Brandeau ML, Owens DK, Bendavid E. The cost-effectiveness of preexposure prophylaxis for HIV prevention in the united states in men who have sex with men. Ann Intern Med. 2012;156(8):541-U144.
Kazemian P, Costantini S, Kumarasamy N, Paltiel AD, Mayer KH, Chandhiok N, et al. The cost-effectiveness of human immunodeficiency virus (HIV) preexposure prophylaxis and HIV testing strategies in high-risk groups in India. Clin Infect Dis. 2020;70(4):633–42.
Kessler J, Myers JE, Nucifora KA, Mensah N, Toohey C, Khademi A, et al. Evaluating the impact of prioritization of antiretroviral pre-exposure prophylaxis in New York. AIDS. 2014;28(18):2683–91.
Leelahavarong P, Teerawattananon Y, Werayingyong P, Akaleephan C, Premsri N, Namwat C, et al. Is a HIV vaccine a viable option and at what price? An economic evaluation of adding HIV vaccination into existing prevention programs in Thailand. BMC Public Health. 2011;11:15.
Luz PM, Osher B, Grinsztejn B, Maclean RL, Losina E, Stern ME, et al. The cost-effectiveness of HIV pre-exposure prophylaxis in men who have sex with men and transgender women at high risk of HIV infection in Brazil. J Int Aids Soc. 2018;21:3.
MacFadden DR, Tan DH, Mishra S. Optimizing HIV pre-exposure prophylaxis implementation among men who have sex with men in a large urban centre: a dynamic modelling study. J Int AIDS Soc. 2016;19:1.
McKenney J, Chen A, Hoover KW, Kelly J, Dowdy D, Sharifi P, et al. Optimal costs of HIV pre-exposure prophylaxis for men who have sex with men. PLoS ONE. 2017;12(6): e0178170.
Mitchell KM, Dimitrov D, Hughes JP, Xia F, Donnell D, Amico KR, et al. In what circumstances could nondaily preexposure prophylaxis for HIV substantially reduce program costs? AIDS. 2018;32(6):809–18.
Nichols BE, Boucher CAB, Dijk JH, Thuma PE, Nouwen JL, Baltussen R, et al. Cost-effectiveness of pre-exposure prophylaxis (PrEP) in preventing HIV-1 infections in rural Zambia: a modeling study. PLoS ONE. 2013;8:3.
Nichols BE, Boucher CAB, van der Valk M, Rynders BJA, van de Inver D. Cost-effectiveness analysis of pre-exposure prophylaxis for HIV-1 prevention in the Netherlands: a mathematical modelling study. Lancet Infect Dis. 2016;16(12):1423–9.
O’Byrne P, Orser L, Jacob JD. The costs of HIV pre-exposure prophylaxis (PrEP) care delivery: comparing specialists, primary care, and PrEP-RN. Sex Res Soc Policy. 2020;17(2):326–33.
Ong KJ, Desai S, Field N, Desai M, Nardone A, van Hoek AJ, et al. Economic evaluation of HIV pre-exposure prophylaxis among men-who-have-sex-with-men in England in 2016. Euro Surveill. 2017;22:42.
Ouellet E, Durand M, Guertin JR, LeLorier J, Tremblay CL. Cost effectiveness of “on demand” HIV pre-exposure prophylaxis for non-injection drug-using men who have sex with men in Canada. Can J Infect Dis Med Microbiol. 2015;26(1):23–9.
Pretorius C, Schnure M, Dent J, Glaubius R, Mahiane G, Hamilton M, et al. Modelling impact and cost-effectiveness of oral pre-exposure prophylaxis in 13 low-resource countries. J Int AIDS Soc. 2020;23(2):13.
Pretorius C, Stover J, Bollinger L, Bacaer N, Williams B. Evaluating the cost-effectiveness of pre-exposure prophylaxis (PrEP) and its impact on HIV-1 transmission in South Africa. PLoS ONE. 2010;5(11): e13646.
Price JT, Wheeler SB, Stranix-Chibanda L, Hosek SG, Watts DH, Siberry GK, et al. Cost-effectiveness of pre-exposure HIV prophylaxis during pregnancy and breastfeeding in Sub-Saharan Africa. J Acquir Immune Defic Syndr. 2016;72(Suppl 2):S145–53.
Reitsema M, Hoek AJV, Van Der Loeff MS, Hoornenborg E, Van Sighem A, Wallinga J, et al. Preexposure prophylaxis for men who have sex with men in the Netherlands: impact on HIV and Neisseria gonorrhoeae transmission and cost-effectiveness. AIDS. 2020;34(4):621–30.
Reyes-Uruena J, Campbell C, Diez E, Ortun V, Casabona J. Can we afford to offer pre-exposure prophylaxis to MSM in Catalonia? Cost-effectiveness analysis and budget impact assessment. Aids Care-Psychol Socio-Med Aspects Aids/Hiv. 2018;30(6):784–92.
Roberts DA, Barnabas RV, Abuna F, Lagat H, Kinuthia J, Pintye J, et al. The role of costing in the introduction and scale-up of HIV pre-exposure prophylaxis: evidence from integrating PrEP into routine maternal and child health and family planning clinics in western Kenya. J Int AIDS Soc. 2019;22:7.
Schneider K, Gray RT, Wilson DP. A cost-effectiveness analysis of HIV preexposure prophylaxis for men who have sex with men in Australia. Clin Infect Dis. 2014;58(7):1027–34.
Shen MW, Xiao YN, Rong LB, Meyers LA, Bellan SE. The cost-effectiveness of oral HIV pre-exposure prophylaxis and early antiretroviral therapy in the presence of drug resistance among men who have sex with men in San Francisco. BMC Med. 2018;16:2.
Shen MW, Xiao YN, Rong LB, Zhuang GH. Global dynamics and cost-effectiveness analysis of HIV pre-exposure prophylaxis and structured treatment interruptions based on a multi-scale model. Appl Math Model. 2019;75:162–200.
van de Vijver DAMC, Richter AK, Boucher CAB, Gunsenheimer-Bartmeyer B, Kollan C, Nichols BE, et al. Cost-effectiveness and budget effect of pre-exposure prophylaxis for HIV-1 prevention in Germany from 2018 to 2058. Eurosurveillance. 2019;24:7.
Verguet S, Stalcup M, Walsh JA. Where to deploy pre-exposure prophylaxis (PrEP) in sub-Saharan Africa? Sex Transmit Infect. 2013;89(8):628–34.
Walensky RP, Horn T, McCann NC, Freedberg KA, Paltiel AD. Comparative pricing of branded tenofovir alafenamide-emtricitabine relative to generic tenofovir disoproxil fumarate-emtricitabine for hiv preexposure prophylaxis. Ann Intern Med. 2020;172(9):583.
Wang L, Hamilton DT, Rosenberg ES, Aslam MV, Sullivan PS, Katz DA, et al. Cost-effectiveness of pre-exposure prophylaxis among adolescent sexual minority males. J Adolesc Health. 2020;66(1):100–6.
Wong NS, Kwan TH, Tsang OTY, Lee MP, Yam WC, Lam W, et al. Pre-exposure prophylaxis (PrEP) for MSM in low HIV incidence places: should high risk individuals be targeted? Sci. 2018;8(1):11641.
Zhang L, Peng P, Wu YM, Ma XM, Soe NN, Huang XJ, et al. Modelling the epidemiological impact and cost-effectiveness of PrEP for HIV transmission in MSM in China. AIDS Behav. 2019;23(2):523–33.
Adamson BJS, Carlson JJ, Kublin JG, Garrison LP. The Potential cost-effectiveness of pre-exposure prophylaxis combined with HIV vaccines in the United States. Vaccines. 2017;5:2.
Alistar SS, Owens DK, Brandeau ML. Effectiveness and cost effectiveness of oral pre-exposure prophylaxis in a portfolio of prevention programs for injection drug users in mixed HIV epidemics. PLoS ONE. 2014;9(1): e86584.
Alsallaq RA, Buttolph J, Cleland CM, Hallett T, Inwani I, Agot K, et al. The potential impact and cost of focusing HIV prevention on young women and men: A modeling analysis in western Kenya. PLoS ONE. 2017;12(4): e0175447.
Anderson SJ, Cherutich P, Kilonzo N, Cremin I, Fecht D, Kimanga D, et al. Maximising the effect of combination HIV prevention through prioritisation of the people and places in greatest need: a modelling study. Lancet. 2014;384(9939):249–56.
Anderson SJ, Ghys PD, Ombam R, Hallett TB. Frontloading HIV financing maximizes the achievable impact of HIV prevention. J Int Aids Soc. 2018;21:3.
Bernard CL, Brandeau ML, Humphreys K, Bendavid E, Holodniy M, Weyant C, et al. Cost-effectiveness of HIV preexposure prophylaxis for people who inject drugs in the United States. Ann Intern Med. 2016;165(1):10.
Bernard CL, Owens DK, Goldhaber-Fiebert JD, Brandeau ML. Estimation of the cost-effectiveness of HIV prevention portfolios for people who inject drugs in the United States: a model-based analysis. Plos Med. 2017;14:5.
Borquez A, Guanira JV, Revill P, Caballero P, Silva-Santisteban A, Kelly S, et al. The impact and cost-effectiveness of combined HIV prevention scenarios among transgender women sex-workers in Lima, Peru: a mathematical modelling study. Lancet Public Health. 2019;4(3):E127–36.
Chiu C, Johnson LF, Jamieson L, Larson BA, Meyer-Rath G. Designing an optimal HIV programme for South Africa: Does the optimal package change when diminishing returns are considered? BMC Public Health. 2017;17:2.
Cremin I, McKinnon L, Kimani J, Cherutich P, Gakii G, Muriuki F, et al. PrEP for key populations in combination HIV prevention in Nairobi: a mathematical modelling study. Lancet Hiv. 2017;4(5):E214–22.
Hu QH, Meyers K, Xu JJ, Chu ZX, Zhang J, Ding HB, et al. Efficacy and cost-effectiveness of early antiretroviral therapy and partners’ pre-exposure prophylaxis among men who have sex with men in Shenyang, China: a prospective cohort and costing study. BMC Infect Dis. 2019;19:11.
Juusola JL, Brandeau ML. HIV treatment and prevention: a simple model to determine optimal investment. Med Decis Mak. 2016;36(3):391–409.
Krebs E, Zang X, Enns B, Min JE, Behrends CN, Del Rio C, et al. The impact of localized implementation: determining the cost-effectiveness of HIV prevention and care interventions across six United States cities. AIDS. 2020;34(3):447–58.
Li JH, Peng LP, Gilmour S, Gu J, Ruan YH, Zou HC, et al. A mathematical model of biomedical interventions for HIV prevention among men who have sex with men in China. BMC Infect Dis. 2018;18:9.
Long EF, Stavert RR. Portfolios of biomedical HIV interventions in South Africa: a cost-effectiveness analysis. J Gen Intern Med. 2013;28(10):1294–301.
Meyer-Rath G, van Rensburg C, Larson B, Jamieson L, Rosen S. Revealed willingness-to-pay versus standard cost-effectiveness thresholds: evidence from the South African HIV investment case. PLoS ONE. 2017;12(10): e0186496.
Mitchell KM, Lepine A, Terris-Prestholt F, Torpey K, Khamofu H, Folayan MO, et al. Modelling the impact and cost-effectiveness of combination prevention amongst HIV serodiscordant couples in Nigeria. AIDS. 2015;29(15):2035–44.
Nichols BE, Baltussen R, van Dijk JH, Thuma PE, Nouwen JL, Boucher CA, et al. Cost-effectiveness of PrEP in HIV/AIDS control in Zambia: a stochastic league approach. J Acquir Immune Defic Syndr. 2014;66(2):221–8.
Quaife M, Terris-Prestholt F, Eakle R, Escobar MAC, Kilbourne-Brook M, Mvundura M, et al. The cost-effectiveness of multi-purpose HIV and pregnancy prevention technologies in South Africa. J Int Aids Soc. 2018;21:2.
Reidy M, Gardiner E, Pretorius C, Glaubius R, Torjesen K, Kripke K. Evaluating the potential impact and cost-effectiveness of dapivirine vaginal ring pre-exposure prophylaxis for HIV prevention. PLoS ONE. 2019;14(6): e0218710.
Smith JA, Anderson SJ, Harris KL, McGillen JB, Lee E, Garnett GP, et al. Maximising HIV prevention by balancing the opportunities of today with the promises of tomorrow: a modelling study. Lancet Hiv. 2016;3(7):E289–96.
Stover J, Bollinger L, Izazola JA, Loures L, DeLay P, Ghys PD, et al. What is required to end the AIDS epidemic as a public health threat by 2030? The cost and impact of the fast-track approach. PLoS ONE. 2016;11(5):14.
Uyei J, Fiellin DA, Buchelli M, Rodriguez-Santana R, Braithwaite RS. Effects of naloxone distribution alone or in combination with addiction treatment with or without pre-exposure prophylaxis for HIV prevention in people who inject drugs: a cost-effectiveness modelling study. Lancet Public Health. 2017;2(3):E133–40.
Vermeersch S, Callens S, De Wit S, Goffard JC, Laga M, Van Beckhoven D, et al. Health and budget impact of combined HIV prevention—first results of the BELHIVPREV model. Acta Clin Belg. 2018;73(1):54–67.
Ying R, Sharma M, Heffron R, Celum CL, Baeten JM, Katabira E, et al. Cost-effectiveness of pre-exposure prophylaxis targeted to high-risk serodiscordant couples as a bridge to sustained ART use in Kampala, Uganda. J Int AIDS Soc. 2015;18:21–9.
Harmon TM, Fisher KA, McGlynn MG, Stover J, Warren MJ, Teng Y, et al. Exploring the potential health impact and cost-effectiveness of AIDS vaccine within a comprehensive HIV/AIDS response in low- and middle-income countries. PLoS ONE. 2016;11(1): e0146387.
Moodley N, Gray G, Bertram M. The case for adolescent HIV vaccination in South Africa: a cost-effectiveness analysis. Medicine (Baltimore). 2016;95(4): e2528.
Moodley N, Gray G, Bertram M. Projected economic evaluation of the national implementation of a hypothetical HIV vaccination program among adolescents in South Africa, 2012. BMC Public Health. 2016;16:13.
Selinger C, Bershteyn A, Dimitrov DT, Adamson BJS, Revill P, Hallett TB, et al. Targeting and vaccine durability are key for population-level impact and cost-effectiveness of a pox-protein HIV vaccine regimen in South Africa. Vaccine. 2019;37(16):2258–67.
de Montigny S, Adamson BJS, Masse BR, Garrison LP Jr, Kublin JG, Gilbert PB, et al. Projected effectiveness and added value of HIV vaccination campaigns in South Africa: a modeling study. Sci. 2018;8(1):6066.
Hontelez JAC, Nagelkerke N, Barnighausen T, Bakker R, Tanser F, Newell ML, et al. The potential impact of RV144-like vaccines in rural South Africa: a study using the STDSIM microsimulation model. Vaccine. 2011;29(36):6100–6.
Long EF, Owens DK. The cost-effectiveness of a modestly effective HIV vaccine in the United States. Vaccine. 2011;29(36):6113–24.
Adamson B, Garrison L, Barnabas RV, Carlson JJ, Kublin J, Dimitrov D. Competing biomedical HIV prevention strategies: potential cost-effectiveness of HIV vaccines and PrEP in Seattle, WA. J Int AIDS Soc. 2019;22(8):11.
Terris-Prestholt F, Foss AM, Cox AP, Heise L, Meyer-Rath G, Delany-Moretlwe S, et al. Cost-effectiveness of tenofovir gel in urban South Africa: model projections of HIV impact and threshold product prices. BMC Infect Dis. 2014;14:11.
Verguet S, Walsh JA. Vaginal microbicides save money: a model of cost-effectiveness in South Africa and the USA. Sexually Transmitted Infections. 2010;86(3):212–6.
Walensky RP, Park JE, Wood R, Freedberg KA, Scott CA, Bekker LG, et al. The cost-effectiveness of pre-exposure prophylaxis for HIV infection in South African women. Clin Infect Dis. 2012;54(10):1504–13.
Williams BG, Abdool Karim SS, Karim QA, Gouws E. Epidemiological impact of tenofovir gel on the HIV epidemic in South Africa. J Acquir Immune Defic Syndr. 2011;58(2):207–10.
Glaubius RL, Hood G, Penrose KJ, Parikh UM, Mellors JW, Bendavid E, et al. Cost-effectiveness of injectable preexposure prophylaxis for HIV prevention in South Africa. Clin Infect Dis. 2016;63(4):539–47.
van Vliet MM, Hendrickson C, Nichols BE, Boucher CAB, Peters RPH, van de Vijver D. Epidemiological impact and cost-effectiveness of providing long-acting pre-exposure prophylaxis to injectable contraceptive users for HIV prevention in South Africa: a modelling study. J Int AIDS Soc. 2019;22(12):9.
Glaubius R, Ding YJ, Penrose KJ, Hood G, Engquist E, Mellors JW, et al. Dapivirine vaginal ring for HIV prevention: modelling health outcomes, drug resistance and cost-effectiveness. J Int AIDS Soc. 2019;22(5):11.
Vogelzang M, Terris-Prestholt F, Vickerman P, Delany-Moretlwe S, Travill D, Quaife M. Cost-effectiveness of HIV pre-exposure prophylaxis among heterosexual men in South Africa: a cost-utility modeling analysis. J Acquir Immune Defic Syndr. 2020;84(2):173–81.
Awad SF, Sgaier SK, Tambatamba BC, Mohamoud YA, Lau FK, Reed JB, et al. Investigating voluntary medical male circumcision program efficiency gains through subpopulation prioritization: insights from application to Zambia. PLoS ONE. 2015;10(12): e0145729.
Leech AA, Burgess JF, Sullivan M, Kuohung W, Horny M, Drainoni ML, et al. Cost-effectiveness of preexposure prophylaxis for HIV prevention for conception in the United States. AIDS. 2018;32(18):2787–98.
Walensky RP, Jacobsen MM, Bekker LG, Parker RA, Wood R, Resch SC, et al. Potential clinical and economic value of long-acting preexposure prophylaxis for South African women at high-risk for HIV infection. J Infect Dis. 2016;213(10):1523–31.
Case KK, Gomez GB, Hallett TB. The impact, cost and cost-effectiveness of oral pre-exposure prophylaxis in sub-Saharan Africa: a scoping review of modelling contributions and way forward. J Int AIDS Soc. 2019;22(9):21.
Gomez GB, Borquez A, Case KK, Wheelock A, Vassall A, Hankins C. The cost and impact of scaling up pre-exposure prophylaxis for HIV prevention: a systematic review of cost-effectiveness modelling studies. PLoS Med. 2013;10(3):16.
Woods B, Revill P, Sculpher M, Claxton K. Country-level cost-effectiveness thersholds: initial estimates and the need for further research. Value in Health. 2016;19:929–35.
Hontelez JAC, Change AY, Ogbuoji O, de Vlas SJ, Barnighausen T, Atun R. Changing HIV treatment eligibility under health system constraints in sub-Saharan Africa: investment needs, population health gains, and cost-effectiveness. AIDS. 2016;30:2341–50.
UNAIDS. Technical consultation on HIV integration with other health services: 2025 target setting, and 2020-2030 resource needs and impact estimation. UNAIDS; 2020.
Obure CD, Sweeney S, Darsamo V, Michaels-Igbokwe C, Guinness L, Terris-Prestholt F, et al. The costs of delivering integrated HIV and sexual reproductive health services in limited resource settings. PLoS ONE. 2015;10(5): e0124476.
Bulstra CA, Hontelez JAC, Otto M, Stepanova A, Lamontagne E, Yakusik A, et al. Integrating HIV services and other health services: a systematic review and meta-analysis. PLoS Med. 2021;18(11): e1003836.
Mangenah C, Nhamo D, Gudukeya S, Gwavava E, Gavi C, Chiwawa P, et al. Efficiency in PrEP delivery: estimating the annual costs of Oral PrEP in Zimbabwe. AIDS Behav. 2022;26(1):161–70.
Grant H, Gomez GB, Kripke K, Barnabas RV, Watts C, Medley GF, et al. Time to scale up preexposure prophylaxis beyond the highest-risk populations? Modeling insights from high-risk women in Sub-Saharan Africa. Sex Transm Dis. 2020;47(11):767–77.
Acknowledgements
The authors would like to thank the stakeholders who participated in our virtual workshop in March 2021 and provided feedback to and advice on our preliminary findings
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was made possible by the support of the American people through the US President’s Emergency Plan for AIDS Relief (PEPFAR) through the United States Agency for International Development (USAID; Grant no: AID-OAA-A-16-00032). The contents of this paper are the sole responsibility of the co-authors and do not necessarily reflect the views of the PEPFAR, USAID, or the United States Government. The views expressed in this article do not necessarily represent the decisions, policy or views of UNAIDS.
Conflict of interests
Fiammetta M. Bozzani, Fern Terris-Prestholt, Matthew Quaife, Mitzy Gafos, Pitchaya P. Indravudh, Rebecca Giddings, Graham F. Medley, Shelly Malhotra and Sergio Torres-Rueda declare they have no conflicts of interest in relation to this article.
Ethics approval
Not applicable.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Code availability
Not applicable.
Authors’ Contributions
FTP, SM and MQ conceived of the study and reviewed the methods. FB co-designed the literature review with STR, collected and analyzed the data, and drafted the manuscript. All authors read and approved the final version of the manuscript
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Bozzani, F.M., Terris-Prestholt, F., Quaife, M. et al. Costs and Cost-Effectiveness of Biomedical, Non-Surgical HIV Prevention Interventions: A Systematic Literature Review. PharmacoEconomics 41, 467–480 (2023). https://doi.org/10.1007/s40273-022-01223-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-022-01223-w