Abstract
Introduction
Cost-of-illness (COI) studies provide policy-relevant information for cross-country, longitudinal, and other cost comparisons. Prior studies have called for standardization in COI methods. We investigated trends, identified factors associated with variation in COI estimation methods, and characterized reporting of heterogeneity in COI estimates.
Methods
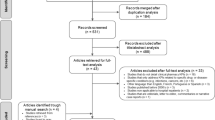
The review of COI studies was implemented following (i) a structured search of PubMed, SCOPUS and EMBASE; (ii) a review of abstracts; (iii) a full-text review; and (iv) classification of articles according to six COI estimation methods: Sum_All Medical, Sum_Diagnosis Specific, Matched, Regression, Other_Total and Other_Incremental. Descriptive and multivariable regression analyses were conducted.
Results
Of the 993 studies included in the full-text review, 186 (18.7 %) were Sum_All Medical, 458 (46.1 %) were Sum_Diagnosis Specific, 96 (9.7 %) were Matched, 97 (9.8 %) were Regression, 70 (7.1 %) were Other_Incremental, and 68 (6.9 %) were Other_Total. Compared with the early period, publications in the middle and late period were associated with lower odds of using Sum_All Medical compared with Sum_Diagnosis Specific (adjusted odds ratio [AOR]middle 0.14; 95 % CI 0.07–0.28; AORlate 0.44; 95 % CI 0.29–0.67). Overall, 640 articles (64 %) reported COI estimates across patient groups defined by patient-level factors, while 247 articles (25 %) reported COI estimates across patient groups defined by non-patient-level factors.
Conclusion
The disease-specific total costing method (Sum_Diagnosis Specific) was most commonly used and its use increased over the time period covered by this review. The investigation of subgroup heterogeneity in COI estimates represents an area for future research.




Similar content being viewed by others
References
Tarricone R. Cost-of-illness analysis. What room in health economics? Health Policy. 2006;77(1):51–63.
Akobundu E, Ju J, Blatt L, Mullins CD. Cost-of-illness studies: a review of current methods. Pharmacoeconomics. 2006;24(9):869–90.
Larg A, Moss JR. Cost-of-illness studies: a guide to critical evaluation. Pharmacoeconomics. 2011;29(8):653–71.
Mauskopf J, Mucha L. A review of the methods used to estimate the cost of Alzheimer’s disease in the United States. Am J Alzheimers Dis Other Dement. 2011;26(4):298–309.
Dee A, Kearns K, O’Neill C, Sharp L, Staines A, O’Dwyer V, et al. The direct and indirect costs of both overweight and obesity: a systematic review. BMC Res Notes. 2014;7:242.
Ettaro L, Songer TJ, Zhang P, Engelgau MM. Cost-of-illness studies in diabetes mellitus. Pharmacoeconomics. 2004;22(3):149–64.
Molinier L, Bauvin E, Combescure C, Castelli C, Rebillard X, Soulie M, et al. Methodological considerations in cost of prostate cancer studies: a systematic review. Value Health. 2008;11(5):878–85.
Molinier L, Combescure C, Chouaid C, Daures JP, Housset B, Fabre D, et al. Cost of lung cancer: a methodological review. Pharmacoeconomics. 2006;24(7):651–9.
Rice DP. Cost-of-illness studies: fact or fiction? Lancet. 1994;344(8936):1519–20.
Shiell A, Gerard K, Donaldson C. Cost of illness studies: an aid to decision making. Health Policy. 1987;8(3):317–23.
Kymes S. “Can we declare victory and move on?” The case against funding burden-of-disease studies. Pharmacoeconomics. 2014;32(12):1153–5.
Byford S, Torgerson DJ, Raftery J. Economic note: cost of illness studies. BMJ. 2000;320(7245):1335.
Currie G, Kerfoot KD, Donaldson C, Macarthur C. Are cost of injury studies useful? Inj Prev. 2000;6(3):175–6.
Rice DP. Cost of illness studies: what is good about them? Inj Prev. 2000;6(3):177–9.
Varmus H. Disease-specific estimates of direct and indirect costs of illness and NIH support. Washington, DC: National Institutes of Health; 2000.
Pisu M, James N, Sampsel S, Saag KG. The cost of glucocorticoid-associated adverse events in rheumatoid arthritis. Rheumatology (Oxford). 2005;44(6):781–8.
Brown P, Ki M, Foxman B. Acute pyelonephritis among adults: cost of illness and considerations for the economic evaluation of therapy. Pharmacoeconomics. 2005;23(11):1123–42.
Cisternas MG, Blanc PD, Yen IH, Katz PP, Earnest G, Eisner MD, et al. A comprehensive study of the direct and indirect costs of adult asthma. J Allergy Clin Immunol. 2003;111(6):1212–8.
Gerber AU, Torre AH, Buscher G, Stock SA, Graf C, Schickendantz S, et al. Direct non-medical and indirect costs for families with children with congenital cardiac defects in Germany: a survey from a university centre. Cardiol Young. 2010;20(2):178–85.
Zhou ZY, Koerper MA, Johnson KA, Riske B, Baker JR, Ullman M, et al. Burden of illness: direct and indirect costs among persons with hemophilia A in the United States. J Med Econ. 2015;18(6):457–65.
Depont F, Hunsche E, Abouelfath A, Diatta T, Addra I, Grelaud A, et al. Medical and non-medical direct costs of chronic low back pain in patients consulting primary care physicians in France. Fundam Clin Pharmacol. 2010;24(1):101–8.
Wallace PJ, Shah ND, Dennen T, Bleicher PA, Crown WH. Optum Labs: building a novel node in the learning health care system. Health Aff (Millwood). 2014;33(7):1187–94.
Institute of Medicine. Variation in health care spending: target decision making, not geography. Washington, DC: Institute of Medicine; 2013.
Gawande A. The hot spotters. The New Yorker. New York: Conde Nast; 2011.
Gawande A. The cost conundrum. The New Yorker. New York: Conde Nast; 2009.
Acknowledgments
The authors wish to thank Sohani Patel, Amy Howard, Jacinda Tran, and Mercedes Wilkes for their assistance with the review of article abstracts and data cleaning. Any errors in the interpretation and reporting of the results are the sole responsibility of the authors.
Author contributions
The interpretation and reporting of these data are the sole responsibility of the authors. Eberechukwu Onukwugha contributed to the study design, data collection, conduct and interpretation of the statistical analysis, and drafted and revised the manuscript with input from all co-authors. Jacquelyn McRae contributed to the study design, data collection, interpretation of the analysis, and drafted, revised and commented on/edited all drafts of the manuscript. Alex Kravetz and Stefan Varga contributed to the data collection, interpretation of the analysis, and reviewed and commented on/edited all drafts of the manuscript. Rahul Khairnar contributed to the data collection, data cleaning, conduct and interpretation of the analysis, and reviewed and commented on/edited all drafts of the manuscript. Daniel Mullins contributed to the study design and interpretation of the analysis, and reviewed and commented on/edited all drafts of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for the conduct of this study.
Conflict of interest
Dr. Onukwugha has received consulting income from AstraZeneca and Janssen Analytics, and Dr. Mullins has received consulting income from Regeneron, Novartis, Daiichi-Sankyo, Bayer, Bristol-Myers Squibb, Mundi Pharma, and Pfizer. Jacquelyn McRae, Alex Kravetz, Stefan Varga, and Rahul Khairnar have no conflicts of interest to declare.
Appendix
Rights and permissions
About this article
Cite this article
Onukwugha, E., McRae, J., Kravetz, A. et al. Cost-of-Illness Studies: An Updated Review of Current Methods. PharmacoEconomics 34, 43–58 (2016). https://doi.org/10.1007/s40273-015-0325-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-015-0325-4