Abstract
Background
There is evidence supporting the value of patient engagement (PE) in research to patients and researchers. However, there is little research evidence on the influence of PE throughout the entire research process as well as the outcomes of research engagement. The purpose of our study is to add to this evidence.
Methods
We used a convergent mixed method design to guide the integration of our survey data and observation data to assess the influence of PE in two groups, comprising patient research partners (PRPs), clinicians, and researchers. A PRP led one group (PLG) and an academic researcher led the other (RLG). Both groups were given the same research question and tasked to design and conduct an inflammatory bowel disease (IBD)-related patient preference study. We administered validated evaluation tools at three points and observed PE in the two groups conducting the IBD study.
Results
PRPs in both groups took on many operational roles and influenced all stages of the IBD-related qualitative study: launch, design, implementation, and knowledge translation. PRPs provided more clarity on the study design, target population, inclusion–exclusion criteria, data collection approach, and the results. PRPs helped operationalize the project question, develop study material and data collection instruments, collect data, and present the data in a relevant and understandable manner to the patient community. The synergy of collaborative partnership resulted in two projects that were patient-centered, meaningful, understandable, legitimate, rigorous, adaptable, feasible, ethical and transparent, timely, and sustainable.
Conclusion
Collaborative and meaningful engagement of patients and researchers can influence all stages of qualitative research including design and approach, and outputs.
Similar content being viewed by others
Although there is evidence that patient engagement in research has value, few studies assess the actual influence of patient engagement at all stages of research. |
By integrating qualitative and quantitative research findings, we identified patient research partner roles, and their influence on critical activities throughout the research spectrum. |
Collaborative and meaningful engagement of patients and researchers can influence the research design and approach, and outputs in qualitative research. |
1 Introduction
Harrington et al. define patient engagement (PE) in research as “the active, meaningful, and collaborative interaction between patients and researchers across all stages of the research process, where research decision making is guided by patients’ contributions as partners, recognizing their specific experiences, values, and expertise” [1]. How patients (including relatives, family caregivers, and public) operate with academic researchers in actual practice varies by the patients’ roles and level of power and decision-making authority [2]. The roles of patients as partners could include involvement in governance, priority setting, developing the research questions, sharing the results with the target audiences, or even performing aspects of the research to ensure that the research being conducted is relevant and valuable to the patients that it affects [3,4,5,6,7,8,9,10]. In this paper, we use the term ‘patient research partner’ (PRP) to define patients who operate as active project members on an equal basis with academic researchers [11].
Several frameworks, guidelines, and resources are available to guide PE activities in research and elucidate the different levels of PE [4, 12,13,14,15,16,17]. The International Association for Public Participation spectrum of public participation (IAP2) framework is an example of a framework that is used often to describe levels of partnership among researchers, patients, and clinicians [18]. However, it is not clear whether this spectrum accurately reflects patient experiences in research and is a desirable model of engagement in health research from the patient’s perspective [19]. There has also been an increasing number of evaluation frameworks to monitor and evaluate PE in research, such as the Community Engagement and Participation in Research measure, Patients as Partners in Research surveys, Patient-Centered Outcomes Research Institute (PCORI) engagement activity inventory (WE-ENACT), Public and Patient Engagement Evaluation Tool (PPEET) etc. [20], as well as a number of studies reporting the various ways in which patient involvement has made a difference, particularly the value of engagement to the patient or researcher [21, 22]. Fewer studies assess how PE influences the research during the different research stages [2, 23,24,25,26] from the perspectives of all members working together on projects using validated evaluation tools or whether there are different study designs, approaches, and outputs based on the roles of PRPs in the research. Few use a mixed method approach. No study uses three evaluation tools together to study PE. Our aim in this exploratory study was to investigate the influence of PE throughout the entire research process (from the launch stage to the knowledge translation stage) and identify the outcomes of research engagement.
2 Methods
We chose a convergent mixed method design [27] to gather a more comprehensive account of the influence of PE in research and outcomes on the research, drawing upon the strengths of both quantitative and qualitative approaches to address our research question. We selected this design because of the lack of rigorously developed and validated tools specifically designed to evaluate the impact of patient engagement on research, and that have involved patients in their development and reporting [28]. We ensured that our study design met all criteria in the Good Reporting of A Mixed Methods Study (GRAMMS) checklist. Our study aimed to assess the influence of PE throughout the entire research process. Our primary research question was “Do PRPs as study team members make a difference to a research study, and at what stages of the research?” Our quantitative approach looked at the self-perceived influence and process of PE on the research during the different stages while our qualitative approach looked at the critical outcomes of research engagement. We used Dillon’s Critical Outcomes of Research Engagement (CORE) framework during analysis to identify short- and long-term outcomes of engagement [23].
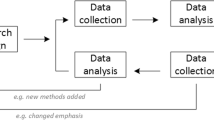
We approached the study in four phases (Fig. 1).
2.1 Phase 1: Co-Designing Research, Capacity and Study Team Building
In this phase, the overarching study team comprising researchers, institutional leaders, and a PRP finalized the study goals and objectives, design, and the data collection approach. We also developed a training package for our study participants containing information about patient preference studies (PPS), qualitative research, and basic information about the Phase 2 project group work and deliverables.
2.2 Phase 2: Establishing Project Groups and Execution of Group Projects
We established two project groups, a PRP-led group (PLG) and an academic researcher-led group (RLG). Each group had two PRPs, two researchers and two clinicians. Our reasoning for two groups was to acknowledge that both researchers and patients can lead research.
2.2.1 Recruitment of Group Members
Outreach to potential project group members occurred through our professional contacts, and through provincial and national networks such as the SPOR IMAGINE (Strategy for Patient-Oriented Research–Inflammation, Microbiome, and Alimentation: Gastro-Intestinal and Neuropsychiatric Effects) Network [29], the Alberta Health Services Digestive Health Strategic Clinical Network (DHSCN), and the Alberta and British Columbia SPOR SUPPORT (Support for People and Patient-Oriented Research and Trials) Units between Fall 2020 and Spring 2021. The study research coordinators sent study flyers to their contacts and mentioned networks requesting them to disseminate this information to potential participants through their organization’s website and by email. Interested patients then contacted the coordinators for more information about the study. We used a maximum variation purposive sampling strategy [30] to recruit PRPs and convenience sampling [30, 31] to recruit clinicians and researchers. PRP participants were purposively selected based on their qualitative research experience and knowledge and training in POR. Table 1 outlines the eligibility criteria for each type of group member. We provided all group members, including the two group leads, an honorarium as per the CIHR guidelines of $25 per hour [32].
2.2.2 Group Allocation
We administered a screening survey prior to the group work to purposively place the recruited members in a group to ensure that both groups were matched as much as possible. The screening survey contained items from the Patient Centered Outcome Research Institute’s Ways of Engaging–ENgagement ACtivity Tool (WE-ENACT) [33, 34]. WE-ENACT allows for modifications of questions and selection or deletion of items to capture stakeholder experience in the engagement. The items used for placing PRPs in their groups included years of qualitative experience and involvement in POR, experience leading POR projects, graduate of the Patient and Community Engagement Research (PaCER) program [35] and some demographic items. The PLG and RLG leads did not have access to the screening survey responses to avoid any unconscious bias that might affect PE during the group work.
2.2.3 Project Group Work
Each project group was tasked with conducting a qualitative patient preference research project independently within a period of 7 months. We selected inflammatory bowel disease (IBD) as the project topic as PRP involvement in qualitative research in this area is considered good research practice [36]. Both groups were provided with the same research question: “What factors or attributes are important to patients with IBD in considering treatment tapering of biologics?”, and were asked to design and conduct a study addressing this question.
2.2.4 Data Collection from Group Members
We collected quantitative data through surveys [41] and qualitative data through observation [40] to address the gaps in the survey data. Our observation data augmented our understanding of what the survey numbers truly mean, including their implications. Qualitative and quantitative data collection was carried out in a similar timeframe, but independent of each other. Equal importance was given to both types of data in answering our study objective.
Members from both groups completed surveys administered at two timepoints: 2 months into the project (early engagement) and at the end of the project (post-engagement). The early-engagement survey contained items from the WE-ENACT tool [33]. The post-engagement survey contained items from three tools: The Patient Engagement in Research Scale-22 (PEIRS-22) [28], the Public and Patient Engagement Evaluation Tool (PPEET-Version 2) [37], and the WE-ENACT. PIERS-22 is a scale that captures the quality or degree of meaningful patient engagement in a project while PPEET-2 captures the participants’ assessments of the key features of the engagement initiative.
Together, the three tools measured the roles of project group members and their perceived influence and impact on the critical activities—the processes and outputs of the qualitative IBD study, as well as the levels of meaningful engagement in the two groups. Participants provided information about their general experience (whether they felt trust, honesty, shared learning, etc.), how convenient it was for them to work on the project, the support they received, the benefits of their involvement, and how satisfied they were with the initiative. We complemented the survey data collection with observation [38,39,40,41] of PE in the two groups. Due to COVID-19 and the different locations of the project group members, observation was undertaken online and included the collection of descriptive information (factual data of online discussions and chats), and reflective information (thoughts, ideas, and questions). One qualified study team member was attached to each group to observe and document stakeholder roles and influence during the qualitative study. A third staff performed oversight of this work to ensure quality data collection. The two observers (NS and KB), one for each group, were trained prior to data collection by a staff member well versed in the observation technique. We used a semi-structured guide as a template for documenting the observations (refer to Marshall et al. [40] for the guide). The observation data enabled us to explore the critical outcomes of research engagement in the two groups. Together with the survey data, we were able to understand the influence of PE on the research design, process, approach, and outputs and the impact on the research.
We received ethics approval from the University of Calgary [REB20-1563] and the University of British Columbia [H20-03385] to conduct this study and for the groups to conduct their qualitative projects.
2.3 Phase 3: Data Analysis and Interpretation
Data analysis occurred after the data collection process was completed. The two forms of data were analyzed separately and then merged.
For each group, we completed the statistical and textual analysis of the survey data (datasets 1 and 2) and the textual analysis of the observation data (dataset 3), separately (Fig. 2). We calculated the frequencies for each level of the Likert scale items for the PPEET and the WE-ENACT items and the PEIRS-22 single construct called meaningful engagement in research [28]. We coded the open-ended questions in our survey data into themes using deductive and inductive codes.
We thematically coded the observation data using NVivo-12 [42] by project stages and activities and by the 11 CORE items [23] (patient-centered, meaningful, team collaboration, understandable, rigorous, integrity and adaptable, legitimate, feasible, ethical and transparent, timely, and sustainable). We created ‘journey maps’ for PRPs, clinicians, and researchers to see how they influenced the project at each project stage and activity [40]. The journey maps gave us an understanding of each participant’s role and their influence to accomplish the group project’s goal. We integrated the findings of the three datasets (dataset 4) by codes/identifiers such as roles of each group member at each project stage and activity. We compared and contrasted the results looking at the complementary and diverse aspects of the influence of PE at each research stage, specifically on the design, approach, and outputs in the two groups. We looked to see how the findings complemented, corroborated, and contradicted each other. We addressed any discordance in our findings during the member check-in exercise with both groups after the discussions. Our integrated approach thus provided a more holistic understanding of PE driven by a framework. The analysis was conducted by two study team members (NS, KB). A third team member (GM) checked the coding and analysis of the observation data.
2.4 Phase 4: Dissemination and Knowledge Translation
We created a research publication and presentation guideline (authorship, process etc.) and shared this document with the group members during a workshop. We also discussed opportunities for joint and individual presentations. This discussion resulted in one group agreeing to consider publishing their reflections about working on the project.
2.5 PRP Involvement
A PRP with IBD lived experience and an extensive background of training and involvement in POR [35, 43, 44] collaborated with the study team in multiple ways from developing the research question and study design through to reviewing this manuscript critically. We held an online meeting to discuss the results and outcomes of PE with members of both groups. The group PRPs were also involved in all the stages and critical tasks of their respective qualitative projects.
3 Results
3.1 Characteristics of Group Participants
Table 2 describes the characteristics of the group participants. From the 29 eligible participants, 14 consented (48% participation) and were placed in a group. Workload issues and health concerns were the most common reasons eligible participants declined participation. Both groups had participants who they had worked with earlier on other projects or were professionally acquainted. Our team felt that it was important to gather this information as group member familiarity could impact collaboration and teamwork which in turn could influence the results of our study. One PRP in the RLG withdrew during the project design stage (retention rate of 93%).
PRPs in both groups had either received PaCER training delivered by University of Calgary Continuing Education [35, 43, 44] or had gained qualitative research expertise through their education and previous jobs. Most of the researchers, including the academic researcher lead, had limited knowledge of IBD. Two researchers and two clinicians were involved in patient-oriented research (POR) for < 1 year.
3.2 Roles and Influence of PE by Project Stages and Critical Activities Including Design, Approach and Output
What PRPs, researchers, and clinicians did, and how they influenced the project is described below by the research stages, and summarized in Tables 3a, 3b, 3c, and 3d. The critical outcomes of research engagement are also highlighted in these tables for each stage. We present the collective results of both groups and individual group results where possible, protecting participants’ confidentiality.
There was active and meaningful interaction between members in both groups. PRPs in both groups were part of the decision-making process in many critical activities across all stages of the group projects. We identified discordance between the number of roles one PRP reported in the surveys compared with the number determined through observation. Two of the three PRPs, including the PLG lead, influenced many project activities “a great deal or moderately”. The PRP in the RLG influenced the question, design, literature review, and data collection a “great deal or moderately” and a “small amount” in the other critical tasks. Clinicians and researchers in the RLG influenced more activities “a great deal or moderately” than those in the PLG group. The researchers in both groups influenced the study design, literature review, and analysis. The clinicians provided their clinical expertise and influenced the literature review. Both the leads had a considerable influence on each critical activity except in capacity building of group members.
3.2.1 Launch Stage
The launch stage involved getting to know each other, working together, and sharing experiences to help the group understand what information patients need (Table 3a). The roles of the researchers and PRPs in both groups at this stage were mainly advisory. Group leads set up discussions to get insights into the group’s experiences, research interests, and skills. This provided a context to plan and discuss group member roles and develop a “working together” plan in the PLG. The RLG had a working plan, but roles were not formally assigned. Group members volunteered to participate in research tasks on an ad hoc basis, resulting in some members in this group feeling “less engaged” on the project. In contrast, knowing their roles and attending weekly meetings kept group members in the PLG accountable, created a transparent process, and proved to be an efficient way for continued participation, and making timely progress on the project. Other studies have also pointed out that a clear patient engagement plan is a successful engagement approach in health research [16, 45].
3.2.2 Design Stage
The design stage involved refining the research question, designing the study, conducting the literature review, and developing the study material (Table 3b). Both groups had productive conversations around the word ‘tapering’ within the research question. The PRPs used their lived experience to help operationalize this word for the qualitative projects. Collaboratively, all members participated in decisions regarding the project title, the target population, and the design and approach for the qualitative project. Not all member ideas were accepted if the group felt that the quality of the project would be compromised and/or if there was not a clear rationale and/or if the idea affected the project timelines. As an example, a group member proposed collecting additional data through patient blogs which was respectfully discussed but vetoed due to time and resource constraints.
There was a clear shared understanding and agreement of the final design and approach in the PLG, while opinions differed regarding the study design and approach in the RLG. The final decision was not attested by all PRPs in this group with the PRPs influencing the group to conduct a literature review first during this stage. The RLG study design was a rapid literature review followed by interviews of IBD patients residing in Canada who had been or were taking biologics, and clinicians who were practicing in Canada with an interest or specialization in IBD. The PLG study design was an unstructured focus group conducted simultaneously with a rapid literature review, followed by one-on-one interviews with IBD patients residing in Canada who were on biologics.
The PRP lead participated in the literature review in the PLG, provided search terms, and reviewed and extracted data, while a PRP in the RLG suggested, reviewed and extracted data from some papers. The researchers were active players in this critical activity. The clinicians provided directions on what literature to look for and where, and they also shared some papers. The final review results were described in simple, plain language by the PRP lead for the ease and understanding of all group members. Group members specified that the literature review reiterated what the PRPs discussed regarding biologic treatments and shed additional light on some of the nuances that patients may perceive when contemplating the tapering question. PRP participation also helped ensure the inclusion of the patient-elucidated terms in the search.
All the study materials were developed by the PRP lead and PRPs in the PLG, while some of the study materials such as the group project recruitment flyer, demographic questionnaire, and the clinician interview guide were developed by a PRP in the RLG, with the remaining materials developed by the RLG researchers. The written material shared at the meeting was presented in patient-friendly language and in a format that worked best for the PRPs in the groups. For example, PRPs in the PLG received lay summaries of DCEs, qualitative research, and results of the literature reviews to support better participation and decision making. Group members indicated that development of study materials by PRPs formed a roadmap to execute the project in alignment with the research question. PRPs were able to tailor the format and language of the study materials for the diverse group of qualitative study participants. They were also able to ensure that the materials were ethical, transparent, and easier to administer during data collection.
PLG group members indicated that PRP insights during this design phase provided their group more clarity on what is important to an IBD patient, the challenges of stopping biologics, and the pitfalls to avoid while designing the project. The group was able to decide on the inclusion–exclusion criteria, and how to approach data collection. One PRP indicated that these conversations helped them develop appropriate data collection guides and improve the quality of data collected.
3.2.3 Implementation Stage
The implementation stage involved recruitment, data collection, and analysis (Table 3c). PRPs in the PLG managed all recruitment and data collection. The PRP lead was the contact person for recruitment and consent of study participants. The structure of the focus group, the sample size, timing, and roles of each PRP during the interviews were decided by the PRPs and shared with other members in this group. The researchers shared the study flyers with their contacts. In the RLG, a PRP managed the recruitment, consent and interviews of clinician participants, while researchers managed the recruitment, consent and interviews of patient participants. A researcher in the group trained the PRP in data collection. PRPs from both groups provided insights into networks and leveraged established relationships with other patient and clinical groups in the system to help with recruitment. Reflexivity was part of the qualitative data collection and analysis process in both groups, with the interviewers debriefing to discuss their reflections. The PRPs continuously checked the assumptions of research team members during the data collection process.
The recruitment and data collection process of both groups incorporated strategies that showed respect for participant diversity. Both groups used purposive sampling and screened interested participants to achieve a diverse sample of research participants. Interviews were arranged on dates and times suitable to both the study participants and group members conducting the interviews. Consent was truly informed. Both groups followed data privacy guidelines and stored and worked on their project data through a secure folder hosted on a university server. Members in both groups supported and appreciated the patient partners in the groups. For example, one of the researchers offered to be a standby in case the patient partners conducting the interviews felt overwhelmed or burdened.
PE in this stage helped the PLG meet its recruitment target and complete data collection satisfying the requirements for diversity of perspective and data saturation. Eleven patients were recruited and interviewed. The RLG recruited and interviewed three clinicians and two patients. The patient sample size was not realized due to delays in “taking off” including ethics approval. PRPs in both groups indicated that they were able to use their skills and experience more meaningfully and ask relevant questions during the interviews. One felt that their role as a facilitator made project participants comfortable and at ease to share information.
Data analysis was conducted by the PRP lead and a researcher in the PLG. In the RLG, data analysis was carried out by the researchers from that group with input from the PRPs on the clinician data. The analyzed data were shared with all group members in both groups iteratively as it was being coded. PRPs’ involvement in data analysis and interpretation helped identify new codes, ensured the patient voice was not missed or mis-represented during coding, and helped clarify some of the responses that were not fully captured in the transcripts. The discussions also produced new topics for research. One member indicated that the discussions increased their understanding of qualitative research methods.
The PLG identified 11 candidate attributes (outputs of the project), while the RLG identified 21 candidate attributes. Both groups generated attributes related to both the process of treatment tapering and the outcomes of tapering. The PLG identified more outcome-oriented attributes and the RLG more process-oriented attributes. Some unique attributes were identified by each group; in the PLG these were impact on mental health and impact on pregnancy or fertility. In the RLG these were presence of antibodies against current biologic medication and type of IBD/location of disease. Similar attributes were side effects and financial cost of the medication. The lead in the PLG influenced the way the final attributes were presented, with the group describing their attributes using patient‐friendly language. The RLG presented their attributes using more research/clinical terms.
3.2.4 Knowledge Translation Stage
The knowledge translation stage involved explaining or applying results to a real-world setting and sharing study findings (Table 3d). Members from both groups participated in a knowledge translation meeting to discuss the research publication and presentation plan. Possible platforms for presenting the results, including journals, were discussed. The PLG offered to discuss publishing reflections on their engagement experience.
3.2.5 Group Member’s Overall Experience and Value of Engagement
Most members in the PLG (6/7) and the RLG (3/5) either “agreed” or “strongly agreed” that they were better informed about research as a result of their participation. All seven PLG members and three RLG members reported overall satisfaction with the engagement initiative and reported that the work was a good use of their time. Six PLG members and three RLG members also indicated that they felt trust, honesty, transparency, shared learning, and give and take relationships, “a great deal” or “somewhat” on their projects.
The PEIRS scale overall group score in the PLG was over 82.7, meaning that the quality of engagement of the members on this project was either “extremely meaningful” or “very meaningful”. The overall group score of meaningful engagement in the RLG was slightly over the cutoff point for deficient engagement (< 70.1) with only one member in this group indicating a “moderately meaningful” engagement. The experience of these participants was not as rewarding, as reflected by two domains on the scale: Team Environment and Interaction (6.0/6.4 cutoff) and Benefits (9.2/9.6 cutoff). Not having a clear idea of the research question; not having team members with clear responsibilities at the start; and failing to complete the group work on time due to many obstacles were some factors that contributed to the “unrewarding” experience in this group.
Both PRPs and researchers perceived an added value from the collaborative work increasing the likelihood of a change in perception of power dynamics in health care research and future collaboration. PRPs indicated that working on the group project provided them the opportunity to work with new colleagues from across the country, build new relationships and learn more about IBD and research. Clinicians and researchers also learned a lot from this experience and found the process “dynamic” and “valuable”. The biggest barrier encountered was the lack of “time” and “understanding” of the overall research objective.
4 Discussion
Over the last 10 years, researchers have been engaging with PRPs in different roles across the research spectrum, with roles generally limited to the preliminary research activities [45, 46]. The influence of the engagement has also been increasingly assessed with some studies presenting hypothesized impacts [47]. Even though our qualitative and quantitative data individually provided some understanding of the influence of PE at each research stage, our mixed methods approach [48, 49] helped us validate and complement our findings from both methods to facilitate a deeper, more comprehensive understanding of patient engagement in research.
PRPs alongside researchers were active members in both project groups. There was equity in the decisions made. PRPs in the two groups performed many parts of the project work, and/or provided insights that were used to shape the direction, methods, analysis, and/or outcomes of the two qualitative research projects. A few crucial activities of influence occurred: during operationalizing the research question to avoid any misinterpretation of the question; during the design stage to ensure that the patient lived experience was embedded in the process and approach and that the method was unbiased and ethical; during the development of the study materials to ensure that the content was clear, ethical and transparent; during the data collection stage to capture high quality data by promoting participant comfort and willingness to share information; and during data interpretation to ensure that the results were relevant and easily understandable. The process of meaningful engagement seen in the groups adds to the synthesis conducted by Greenhalgh et al., in which the authors emphasized the importance of involving patient partners across the research cycle/process and how this involvement gets tailored specifically to the study [15].
Similar results have been seen in other studies using frameworks and tools such as the PPEET, WE-ENACT, and or the PIERS to formally evaluate the impact and capture outcomes of engagement on research. Bhati et al. used the PPEET and the WE-ENACT to assess patient experience and areas of involvement and reported high involvement of PRPs in the initial stages and less involvement in operational activities [50]. Chudyk et al. identified seven activities that PRPs were engaged in across the research cycle, which included identifying and choosing the research topic or method, helping conduct the study, and presenting on behalf of the study team [26]. In the study by Morel et al., PRPs reported an extremely high meaningful engagement similar to the PLG group in our study using the PIERS-22 [51]. A recent work by Babatunde et al., using a mixed method approach, found that PRPs guided the project direction and process, and influenced data collection, analysis, and knowledge translation [52].
The researcher’s roles were operational and advisory; and the clinicians’ role was advisory throughout the project. Despite the differences in the group members’ roles in the two groups, the synergy of collaborative partnership resulted in two projects that were patient-centered, meaningful, understandable, legitimate, rigorous, adaptable, feasible, ethical and transparent, timely, and sustainable, valued by members in the two groups, and by the larger community impacted by this condition.
Formal training of PRP in qualitative methods through programs such as PaCER, and researcher training in POR, as well as resources (human and financial) made available for both groups facilitated meaningful and equitable engagement. The PRP lead specifically had the training and expertise to take on many operational roles such as in data analysis. Similar results are also seen with other educational initiatives such as the Partners in Research (PiR) 2-month online course [53], and the Foundations in Patient-Oriented Research curriculum [54], and in articles emphasizing the importance of thoughtful preparation [54, 55] and training for both researchers and patients for effective collaboration [43, 54,55,56,57,58,59]. However, regardless of training and experience, the team needs to decide which part of the project would benefit from PE, and not expect PRPs to ‘perform’ the research but participate in ways they desire to be involved [53]. Other known facilitators to PE, also seen in our study, were a flexible plan for engagement [45], recognizing that PRP involvement is an iterative and dynamic process, dependent on the PRP disease state throughout the project [59]. The researcher knowledge of the disease condition was also found to be helpful to promote collaborative relationships.
Equally important, as mentioned in PE literature and seen in our recruitment and retention, a minimum of two PRPs or more should be involved in a project due to project workload issues and PRP health concerns [60]. While previous studies have identified challenges to measuring the influence of patient engagement, including the lack of validated measurement tools, or evaluation methods and frameworks [20, 61, 62], a mixed methods approach, supported by a framework, provided an in-depth understanding of the value of collaborative interactions between researchers, healthcare providers and PRPs in research. The findings, however, may not be applicable in other contexts due to the “un-predictableness” of the stakeholders involved [63] in general, as would be the case with any other study involving small project teams. Partly as a consequence of the small size of the project teams, there was not a great deal of diversity (10/14 participants were white and 12/14 were highly educated) in our two groups. Different stakeholders bring different values, attitudes, and perspectives to projects that could affect PE.
Due to the focused nature of the evaluation and the small size of the two groups, we applied descriptive methodologies. The findings may provide insights to researchers engaging in POR with PRPs by reflecting an understanding of partnerships across all stages of research, and how they can change the way a study is designed, approached and conducted. A key insight, for example, is the role of discussions early in the project that will help guide PRPs and researchers in increasing engagement of research team members. Future exploratory research opportunities include studies with diverse types of PRPs not represented in our study and in different study questions and contexts.
5 Conclusion
Collaborative and meaningful engagement of patients and researchers can influence all stages of the qualitative research, including design and approach, and outputs. Supported by the SPOR IMAGINE Network and two provincial SPOR SUPPORT Units (AB and BC), this study provides valuable learnings on the influence of PE throughout the entire research process.
References
Harrington RL, Hanna ML, Oehrlein EM, et al. Defining patient engagement in research: results of a systematic review and analysis: report of the ISPOR Patient-Centered Special Interest Group. Value Health. 2020;23(6):677–88. https://doi.org/10.1016/j.jval.2020.01.019.
Merker VL, Hyde JK, Herbst A, et al. Evaluating the impacts of patient engagement on health services research teams: lessons from the veteran consulting network. J Gen Intern Med. 2022;37(1):33–41. https://doi.org/10.1007/s11606-021-06987-z.
Boaz A, Robert G, Locock L, et al. What patients do and their impact on implementation. J Health Organ Manag. 2016;30(2):258–78. https://doi.org/10.1108/JHOM-02-2015-0027.
Duffett L. Patient engagement: what partnering with patient in research is all about. Thromb Res. 2017;150:113–20. https://doi.org/10.1016/j.thromres.2016.10.029.
Canadian Institutes of Health Research. Patient Engagement in Research Resources; 2020. https://cihr-irsc.gc.ca/e/51916.html. Accessed July 7, 2022.
Hyde C, Dunn KM, Higginbottom A, Chew-Graham CA. Process and impact of patient involvement in a systematic review of shared decision making in primary care consultations. Health Expect. 2017;20(2):298–308. https://doi.org/10.1111/hex.12458.
McCarron TL, Clement F, Rasiah J, et al. Patients as partners in health research: a scoping review. Health Expect. 2021;24(4):1378–90. https://doi.org/10.1111/hex.13272.
Rhodes P, Nocon A, Booth M, et al. A service users’ research advisory group from the perspectives of both service users and researchers. Health Soc Care Community. 2002;10(5):402–9. https://doi.org/10.1046/j.1365-2524.2002.00376.x.
Tapp H, Derkowski D, Calvert M, Welch M, Spencer S. Patient perspectives on engagement in shared decision-making for asthma care. Family Pract. 2017;34(3):353–7. https://doi.org/10.1093/fampra/cmw122.
Wilson T, Miller J, Teare S, et al. Patient perspectives on engagement in decision-making in early management of non-ST elevation acute coronary syndrome: a qualitative study. BMC Med Inform Decis Mak. 2017;17(1):153. https://doi.org/10.1186/s12911-017-0555-9.
Zabalan C, Pennings P, Clausen J, et al. EULAR Guide: Starting a Patient Research Partner (PRP) Group on a National Level. September 2021:13. https://www.eular.org/myUploadData/files/2021_prp_guide_long.pdf.
Canadian Institute for Health Research. Strategy for patient-oriented research–patient engagement framework; 2020. https://cihr-irsc.gc.ca/e/48413.html. Accessed July 17, 2020.
Lavallee DC, Williams CJ, Tambor ES, Deverka PA. Stakeholder engagement in comparative effectiveness research: how will we measure success? J Comp Effect Res. 2012;1(5):397–407. https://doi.org/10.2217/cer.12.44.
Oliver SR, Rees RW, Clarke-Jones L, et al. A multidimensional conceptual framework for analysing public involvement in health services research. Health Expect. 2008;11(1):72–84. https://doi.org/10.1111/j.1369-7625.2007.00476.x.
Greenhalgh T, Hinton L, Finlay T, et al. Frameworks for supporting patient and public involvement in research: systematic review and co-design pilot. Health Expect. 2019;22(4):785–801. https://doi.org/10.1111/hex.12888.
Manafo E, Petermann L, Mason-Lai P, Vandall-Walker V. Patient engagement in Canada: a scoping review of the ‘how’ and ‘what’ of patient engagement in health research. Health Res Policy Syst. 2018;16(1):5. https://doi.org/10.1186/s12961-018-0282-4.
Vanderhout S, Nicholls S, Monfaredi Z, et al. Facilitating and supporting the engagement of patients, families and caregivers in research: the “Ottawa model” for patient engagement in research. Res Involv Engag. 2022;8(1):25. https://doi.org/10.1186/s40900-022-00350-0.
International Association for Public Participation. Core Values, Ethics, Spectrum – The 3 Pillars of Public Participation. https://www.iap2.org/page/pillars. Accessed Apr 25, 2022.
Johannesen J. Patient views on “ladders of engagement”; 2018. https://doi.org/10.13140/RG.2.2.29370.85444.
Boivin A, L’Espérance A, Gauvin FP, et al. Patient and public engagement in research and health system decision making: a systematic review of evaluation tools. Health Expect. 2018;21(6):1075–84. https://doi.org/10.1111/hex.12804.
Lauzon-Schnittka J, Audette-Chapdelaine S, Boutin D, et al. The experience of patient partners in research: a qualitative systematic review and thematic synthesis. Res Involv Engag. 2022;8(1):55. https://doi.org/10.1186/s40900-022-00388-0.
Brett J, Staniszewska S, Mockford C, et al. A systematic review of the impact of patient and public involvement on service users, researchers and communities. Patient. 2014;7(4):387–95. https://doi.org/10.1007/s40271-014-0065-0.
Dillon EC, Tuzzio L, Madrid S, et al. Measuring the impact of patient-engaged research: how a methods workshop identified critical outcomes of research engagement. J Patient-Centered Res Rev. 2017;4(4):237–46. https://doi.org/10.17294/2330-0698.1458.
Russell J, Fudge N, Greenhalgh T. The impact of public involvement in health research: what are we measuring? Why are we measuring it? Should we stop measuring it? Res Involv Engag. 2020;6(1):63. https://doi.org/10.1186/s40900-020-00239-w.
Mockford C, Staniszewska S, Griffiths F, Herron-Marx S. The impact of patient and public involvement on UK NHS health care: a systematic review. Int J Qual Health Care. 2012;24(1):28–38. https://doi.org/10.1093/intqhc/mzr066.
Chudyk AM, Stoddard R, McCleary N, et al. Activities and impacts of patient engagement in CIHR SPOR funded research: a cross-sectional survey of academic researcher and patient partner experiences. Res Involv Engag. 2022;8(1):44. https://doi.org/10.1186/s40900-022-00376-4.
Creswell J, Clark VP. Designing and conducting mixed methods research. 3rd ed. Thousand Oaks: Sage Publications; 2017.
Hamilton CB, Hoens AM, McKinnon AM, et al. Shortening and validation of the Patient Engagement In Research Scale (PEIRS) for measuring meaningful patient and family caregiver engagement. Health Expect. 2021;24(3):863–79. https://doi.org/10.1111/hex.13227.
Moayyedi P, MacQueen G, Bernstein CN, et al. IMAGINE Network’s Mind And Gut Interactions Cohort (MAGIC) Study: a protocol for a prospective observational multicentre cohort study in inflammatory bowel disease and irritable bowel syndrome. BMJ Open. 2020;10(10): e041733. https://doi.org/10.1136/bmjopen-2020-041733.
Patton MQ. Qualitative research and evaluation methods. 4th ed. Thousand Oaks: SAGE; 2014.
Palinkas LA, Horwitz SM, Green CA, et al. Purposeful sampling for qualitative data collection and analysis in mixed method implementation research. Admin Policy Ment Health Ment Health Serv Res. 2015;42(5):533–44. https://doi.org/10.1007/s10488-013-0528-y.
IMAGINE. Compensation and Guidelines. https://imaginespor.com/compensation-policy/. Accessed Apr 25, 2022.
PCORI. Engagement Activity Inventory (NET-ENACT AND WE-ENACT). https://ceppp.ca/en/evaluation-toolkit/pcori-engagement-activity-inventory-net-enact-and-we-enact/. Accessed Aug 10, 2022.
PCORI. Ways of Engaging—ENgagement ACtivity Tool (WE-ENACT)—Patients and Stakeholders 3.0 Item Pool. http://www.pcori.org/sites/default/files/PCORI-WE-ENACT-3-0-Patients-Stakeholders-Item-Pool-080916.pdf. Accessed Apr 25, 2022.
PaCER. Patient and Community Engagement Research. https://pacerinnovates.ca. Accessed Apr 25, 2022.
van Overbeeke E, Vanbinst I, Jimenez-Moreno AC, Huys I. Patient centricity in patient preference studies: the patient perspective. Front Med. 2020;7:93. https://doi.org/10.3389/fmed.2020.00093.
Abelson J, Tripp L, Kandasamy S, et al. Supporting the evaluation of public and patient engagement in health system organizations: results from an implementation research study. Health Expect. 2019;22(5):1132–43. https://doi.org/10.1111/hex.12949.
Berg BL, Lune H. Qualitative research methods for the social sciences. 9th ed. Boston: Pearson; 2017.
Kawulich B. Collecting data through observation. In: Doing social research: a global context. New York: McGraw-Hill Higher Education; 2012. p. 150–60.
Marshall DA, Suryaprakash N, Lavallee DC, et al. Exploring the outcomes of research engagement using the observation method in an online setting. BMJ Open. 2023;13: e073953. https://doi.org/10.1136/bmjopen-2023-073953.
Marshall DA, Suryaprakash N, Bryan S, et al. Measuring the impact of patient engagement in health research: an exploratory study using multiple survey tools. J Can Assoc Gastroenterol. 2023. https://doi.org/10.1093/jcag/gwad045.
Best Qualitative Data Analysis Software for Researchers | NVivo. https://www.qsrinternational.com/nvivo-qualitative-data-analysis-software/home. Accessed Apr 25, 2022.
Shklarov S, Marshall DA, Wasylak T, Marlett NJ. “Part of the Team”: mapping the outcomes of training patients for new roles in health research and planning. Health Expect. 2017;20(6):1428–36. https://doi.org/10.1111/hex.12591.
Marlett N, Shklarov S, Marshall D, et al. Building new roles and relationships in research: a model of patient engagement research. Qual Life Res. 2015;24(5):1057–67. https://doi.org/10.1007/s11136-014-0845-y.
Etchegary H, Pike A, Patey AM, et al. Operationalizing a patient engagement plan for health research: sharing a codesigned planning template from a national clinical trial. Health Expect. 2022;25(2):697–711. https://doi.org/10.1111/hex.13417.
Skovlund PC, Nielsen BK, Thaysen HV, et al. The impact of patient involvement in research: a case study of the planning, conduct and dissemination of a clinical, controlled trial. Res Involv Engag. 2020;6(1):43. https://doi.org/10.1186/s40900-020-00214-5.
Ball S, Harshfield A, Carpenter A, et al. Patient and public involvement in research: enabling meaningful contributions. RAND Corp. 2019. https://doi.org/10.7249/RR2678.
Fetters MD, Curry LA, Creswell JW. Achieving integration in mixed methods designs-principles and practices. Health Serv Res. 2013;48(6 Pt 2):2134–56. https://doi.org/10.1111/1475-6773.12117.
Wasti SP, Simkhada P, van Teijlingen ER, et al. The growing importance of mixed-methods research in health. Nepal J Epidemiol. 2022;12(1):1175–8. https://doi.org/10.3126/nje.v12i1.43633.
Bhati DK, Fitzgerald M, Kendall C, Dahrouge S. Patients’ engagement in primary care research: a case study in a Canadian context. Res Involv Engag. 2020;6(1):65. https://doi.org/10.1186/s40900-020-00238-x.
Morel T, Schroeder K, Cleanthous S, et al. The value of co-creating a clinical outcome assessment strategy for clinical trial research: process and lessons learnt. Res Involv Engag. 2023;9:98. https://doi.org/10.1186/s40900-023-00505-7.
Babatunde S, Ahmed S, Santana MJ, et al. Working together in health research: a mixed-methods patient engagement evaluation. Res Involv Engag. 2023;9:62. https://doi.org/10.1186/s40900-023-00475-w.
Courvoisier M, Baddeliyanage R, Wilhelm L, et al. Evaluation of the partners in research course: a patient and researcher co-created course to build capacity in patient-oriented research. Res Involv Engag. 2021;7(1):76. https://doi.org/10.1186/s40900-021-00316-8.
Bell T, Vat LE, McGavin C, et al. Co-building a patient-oriented research curriculum in Canada. Res Involv Engag. 2019;5(1):7. https://doi.org/10.1186/s40900-019-0141-7.
Bird M, Ouellette C, Whitmore C, et al. Preparing for patient partnership: a scoping review of patient partner engagement and evaluation in research. Health Expect. 2020;23(3):523–39. https://doi.org/10.1111/hex.13040.
Vat LE, Ryan D, Etchegary H. Recruiting patients as partners in health research: a qualitative descriptive study. Res Involv Engag. 2017;3(1):15. https://doi.org/10.1186/s40900-017-0067-x.
Harrison JD, Auerbach AD, Anderson W, et al. Patient stakeholder engagement in research: a narrative review to describe foundational principles and best practice activities. Health Expect. 2019;22(3):307. https://doi.org/10.1111/hex.12873.
Langlois S, Mehra K. Teaching about partnerships between patients and the team: exploring student perceptions. J Patient Exp. 2020;7(6):1589–94. https://doi.org/10.1177/2374373520933130.
Boaz A, Hanney S, Borst R, et al. How to engage stakeholders in research: design principles to support improvement. Health Res Policy Syst. 2018;16(1):60. https://doi.org/10.1186/s12961-018-0337-6.
Kirwan JR, de Wit M, Frank L, Haywood KL, Salek S, Brace-McDonnell S, Lyddiatt A, Barbic SP, Alonso J, Guillemin F, Bartlett SJ. Emerging guidelines for patient engagement in research. Value Health. 2017;20(3):481–6. https://doi.org/10.1016/j.jval.2016.10.003.
Vat LE, Finlay T, Jan Schuitmaker-Warnaar T, et al. Evaluating the “return on patient engagement initiatives” in medicines research and development: a literature review. Health Expect. 2020;23(1):5–18. https://doi.org/10.1111/hex.12951.
L’Espérance A, O’Brien N, Grégoire A, et al. Developing a Canadian evaluation framework for patient and public engagement in research: study protocol. Res Involv Engag. 2021;7(1):10. https://doi.org/10.1186/s40900-021-00255-4.
Staley K. ‘Is it worth doing?’ Measuring the impact of patient and public involvement in research. Res Involv Engag. 2015;1(1):6. https://doi.org/10.1186/s40900-015-0008-5.
Acknowledgements
The authors thank the 14 study participants from the two project groups who helped the study team carry out the research. They also acknowledge the support from study team members Aida Fernandes, Executive Director IMAGINE Network; Tracy Wasylak, Chief Program Officer, Strategic Clinical Networks™ with Alberta Health Services; Dr Gilaad Kaplan, Gastroenterologist and Professor in the Cumming School of Medicine at the University of Calgary; and Louise Morrin, Senior Provincial Director, Medicine Strategic Clinical Network at Alberta Health Services during the different study phases. The authors are also grateful to the funders of the study: Strategy for Patient-Oriented Research–Inflammation, Microbiome, and Alimentation: Gastro-Intestinal and Neuropsychiatric Effects (SPOR IMAGINE) Network, Canadian Institute of Health Research Network, McMaster University, University of Calgary, University of Alberta, Queen’s University, Dalhousie University, Montreal Heart Institute Research Centre, Takeda Pharmaceutical Company, Allergan Incorporated, Alberta Innovates, Research Manitoba, and Crohn’s and Colitis Canada.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Funding from the SPOR IMAGINE (Strategy for Patient-Oriented Research, Inflammation, Microbiome, and Alimentation: Gastro-Intestinal and Neuropsychiatric Effects) Network was used to conduct the study. The Network is supported by a grant from the Canadian Institute of Health Research (Funding Reference Number: 1715-000-001) with funding matched by McMaster University, University of Calgary, University of Alberta, Queen’s University, Dalhousie University, Montreal Heart Institute Research Centre, Takeda Pharmaceutical Company, Allergan Incorporated, Alberta Innovates, Research Manitoba, Crohn’s and Colitis Canada. The IMAGINE Network is sponsoring the open access fee.
Conflict of Interest
Deborah A. Marshall discloses consulting fees from the Office for Health Economics, Novartis, and Analytica during the conduct of this study. She also received support from Illumina for travel expenses to attend a meeting. Nitya Suryaprakash and Karis L. Barker received reimbursement of expenses related to conference attendance from the SPOR IMAGINE Chronic Disease Network. All other authors declare no conflicts of interest relevant to the content of this article.
Data Availability
The ethics approval for this study does not support the sharing of raw data.
Ethics Approval
The study was approved by the respective research ethics boards of the University of Calgary [REB20-1563] and the University of British Columbia [H20-03385]. The authors certify that the study was performed in accordance with the ethical standards detailed in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Code Availability
Not applicable.
Author Contributions
Deborah A. Marshall conceptualized the study and led the design, conduct, and analysis of this study and the drafting of and revising of the article. Danielle C. Lavelle and Stirling Bryan conceptualized the study and led the design, conduct, and analysis of this study and helped revise the manuscript. Nitya Suryaprakash and Karis L. Barker participated in the design, coordination, data collection, conduct, and analysis of the study and in drafting and revising the manuscript. Paul Moayyedi contributed to the acquisition and interpretation of data and reviewed the manuscript critically. All other authors participated in the design, conduct, and analysis of the study and reviewed the manuscript critically. All authors approved the final version to be published and agreed to be accountable for all aspects of the work.
Additional information
Sandra Zelinsky: Patient Author.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Marshall, D.A., Suryaprakash, N., Lavallee, D.C. et al. Studying How Patient Engagement Influences Research: A Mixed Methods Study. Patient (2024). https://doi.org/10.1007/s40271-024-00685-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s40271-024-00685-8