Abstract
Introduction
Increasing availability of medico-administrative databases has prompted the development of automated pharmacovigilance signal detection methodologies. Self-controlled approaches have recently been proposed. They account for time-independent confounding factors that may not be recorded. So far, large numbers of drugs have been screened either univariately or with LASSO penalized regressions.
Objective
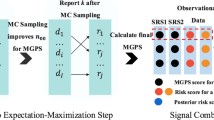
We propose and assess a new method that combines the case-crossover self-controlled design with propensity scores (propensity score-adjusted case-crossover) built from high-dimensional data-driven variable selection, to account for co-medications or possibly other measured confounders.
Methods
Comparison with the univariate and LASSO case-crossover was performed from simulations and a real-data study. Multiple regressions (LASSO, propensity score-adjusted case-crossover) accounted for co-medications and no other covariates. For the univariate and propensity score-adjusted case-crossover methods, the detection threshold was based on a false discovery rate procedure, while for LASSO, it relied on the Akaike Information Criterion. For the real-data study, two drug safety experts evaluated the signals generated from the analysis of 4099 patients with acute myocardial infarction from the French national health database.
Results
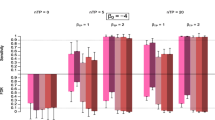
On simulations, our approach ranked the signals similarly to the LASSO and better than the univariate method while controlling the false discovery rate at the prespecified level, contrary to the univariate method. The LASSO provided the best sensitivity at the cost of larger false discovery rate estimates. On the application, our approach showed similar performances to the LASSO and better performances than the univariate method. It highlighted 43 signals out of 609 drug candidates: 22 (51%) were considered as potentially pharmacologically relevant, including seven (16%) regarded as highly relevant.
Conclusions
Our findings show the interest of a propensity score combined with a case-crossover for pharmacovigilance. They also confirm that indication bias remains a challenge when mining medico-administrative databases.




Similar content being viewed by others
References
Roux E, Thiessard F, Fourrier A, Bégaud B, Tubert-Bitter P. Evaluation of statistical association measures for the automatic signal generation in pharmacovigilance. IEEE Trans Inf Technol Biomed. 2005;9:518–27.
Tatonetti NP, Ye PP, Daneshjou R, Altman RB. Data-driven prediction of drug effects and interactions. Sci Transl Med. 2012;4:125ra31.
Harpaz R, DuMouchel W, Shah NH, Madigan D, Ryan P, Friedman C. Novel data mining methodologies for adverse drug event discovery and analysis. Clin Pharmacol Ther. 2012;91:1010–21.
Caster O, Norén GN, Madigan D, Bate A. Large-scale regression-based pattern discovery: the example of screening the WHO global drug safety database. Stat Anal Data Min ASA Data Sci J. 2010. https://doi.org/10.1002/sam.10078.
Ahmed I, Pariente A, Tubert-Bitter P. Class-imbalanced subsampling lasso algorithm for discovering adverse drug reactions. Stat Methods Med Res. 2018;27:785–97.
Pariente A, Gregoire F, Fourrier-Reglat A, Haramburu F, Moore N. Impact of safety alerts on measures of disproportionality in spontaneous reporting databases: the notoriety bias. Drug Saf. 2007;30:891–8.
Arnaud M, Salvo F, Ahmed I, Robinson P, Moore N, Bégaud B, et al. A method for the minimization of competition bias in signal detection from spontaneous reporting databases. Drug Saf. 2016;39:251–60.
Salvo F, Leborgne F, Thiessard F, Moore N, Bégaud B, Pariente A. A potential event-competition bias in safety signal detection: results from a spontaneous reporting research database in France. Drug Saf. 2013;36:565–72.
Arnaud M, Bégaud B, Thiessard F, Jarrion Q, Bezin J, Pariente A, et al. An automated system combining safety signal detection and prioritization from healthcare databases: a pilot study. Drug Saf. 2018;41:377–87.
Ryan PB, Stang PE, Overhage JM, Suchard MA, Hartzema AG, DuMouchel W, et al. A comparison of the empirical performance of methods for a risk identification system. Drug Saf. 2013;36(Suppl. 1):S143–58.
Farrington CP. Relative incidence estimation from case series for vaccine safety evaluation. Biometrics. 1995;51:228–35.
Ryan PB, Madigan D, Stang PE, Overhage JM, Racoosin JA, Hartzema AG. Empirical assessment of methods for risk identification in healthcare data: results from the experiments of the Observational Medical Outcomes Partnership. Stat Med. 2012;31:4401–15.
Maclure M. The case-crossover design: a method for studying transient effects on the risk of acute events. Am J Epidemiol. 1991;133:144–53.
Schneeweiss S, Rassen JA, Glynn RJ, Avorn J, Mogun H, Brookhart MA. High-dimensional propensity score adjustment in studies of treatment effects using health care claims data. Epidemiology. 2009;20:512–22.
Franklin JM, Eddings W, Glynn RJ, Schneeweiss S. Regularized regression versus the high-dimensional propensity score for confounding adjustment in secondary database analyses. Am J Epidemiol. 2015;182:651–9.
Courtois É, Pariente A, Salvo F, Volatier É, Tubert-Bitter P, Ahmed I. Propensity score-based approaches in high dimension for pharmacovigilance signal detection: an empirical comparison on the French Spontaneous Reporting Database. Front Pharmacol. 2018;9:1010.
Demailly R, Escolano S, Haramburu F, Tubert-Bitter P, Ahmed I. Identifying drugs inducing prematurity by mining claims data with high-dimensional confounder score strategies. Drug Saf. 2020;43:549–59.
Wang SV, Maro JC, Baro E, Izem R, Dashevsky I, Rogers JR, et al. Data mining for adverse drug events with a propensity score-matched tree-based scan statistic. Epidemiology. 2018;29:895–903.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol. 1995;57:289–300.
Simpson SE, Madigan D, Zorych I, Schuemie MJ, Ryan PB, Suchard MA. Multiple self-controlled case series for large-scale longitudinal observational databases. Biometrics. 2013;69:893–902.
Mittleman MA, Mostofsky E. Exchangeability in the case-crossover design. Int J Epidemiol. 2014;43:1645–55.
Hardin JW. Generalized estimating equations (GEE). In: Everitt BS, Howell DC, editors. Encyclopedia of statistics in behavioral science. Wiley; 2005. pp. 39–42. https://doi.org/10.1002/0470013192.bsa250.
Avalos M, Grandvalet Y, Adroher ND, Orriols L, Lagarde E. Analysis of multiple exposures in the case-crossover design via sparse conditional likelihood. Stat Med. 2012;31:2290–302.
Ahmed I, Dalmasso C, Haramburu F, Thiessard F, Broët P, Tubert-Bitter P. False discovery rate estimation for frequentist pharmacovigilance signal detection methods. Biometrics. 2010;66:301–9.
Månsson R, Joffe MM, Sun W, Hennessy S. On the estimation and use of propensity scores in case-control and case-cohort studies. Am J Epidemiol. 2007;166:332–9.
Chen T, Guestrin C. XGBoost: a scalable tree boosting system. In: Proceedings of the 22nd ACM SIGKDD international conference on knowledge discovery and data mining; 13–17 August, 2016; San Francisco (CA), pp. 785–94. https://doi.org/10.1145/2939672.2939785.
Kuhn M, Letunic I, Jensen LJ, Bork P. The SIDER database of drugs and side effects. Nucleic Acids Res. 2016;44:D1075–9.
WHO Collaborating Centre for Drug Statistics Methodology. ATC classification index with DDDs. Oslo, Norway 2018; 2019.
Zivich PN, Breskin A. Machine learning for causal inference: on the use of cross-fit estimators. Epidemiology. 2021;32:393–401.
Rose S, van der Laan MJ. Simple optimal weighting of cases and controls in case-control studies. Int J Biostat. 2008;4:Article 19.
Poluzzi E, Raschi E, Godman B, Koci A, Moretti U, Kalaba M, et al. Pro-arrhythmic potential of oral antihistamines (H1): combining adverse event reports with drug utilization data across Europe. PLoS ONE. 2015;10: e0119551.
Woosley RL, Heise CW, Romero KA. QTdrugs list. www.crediblemeds.org. Accessed 1 Feb 2022.
Stoetzer C, Voelker M, Doll T, Heineke J, Wegner F, Leffler A. Cardiotoxic antiemetics metoclopramide and domperidone block cardiac voltage-gated Na+ channels. Anesth Analg. 2017;124:52–60.
Coloma PM, Schuemie MJ, Trifirò G, Furlong L, van Mulligen E, Bauer-Mehren A, et al. Drug-induced acute myocardial infarction: identifying ‘prime suspects’ from electronic healthcare records-based surveillance system. PLoS One. 2013;8: e72148.
Rajpal S, Morris LA, Akkus NI. Non-ST-elevation myocardial infarction with the use of oxymetazoline nasal spray. Rev Port Cardiol. 2014;33(51):e1-4.
Oosterbaan R, Burns MJ. Myocardial infarction associated with phenylpropanolamine. J Emerg Med. 2000;18:55–9.
Akay S, Ozdemir M. Acute coronary syndrome presenting after pseudoephedrine use and regression with beta-blocker therapy. Can J Cardiol. 2008;24:e86–8.
Bally M, Dendukuri N, Rich B, Nadeau L, Helin-Salmivaara A, Garbe E, et al. Risk of acute myocardial infarction with NSAIDs in real world use: Bayesian meta-analysis of individual patient data. BMJ. 2017;357: j1909.
Bhala N, Emberson J, Merhi A, Abramson S, Arber N, et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet. 2013;382:769–79.
Varas-Lorenzo C, Riera-Guardia N, Calingaert B, Castellsague J, Salvo F, Nicotra F, et al. Myocardial infarction and individual nonsteroidal anti-inflammatory drugs meta-analysis of observational studies. Pharmacoepidemiol Drug Saf. 2013;22:559–70.
Suissa S. The case-time-control design. Epidemiology. 1995;6:248–53.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research was partially supported by the ANSM (Agence Nationale de Sécurité du Médicament et des Produits de Santé) as part of the 2014 ‘young researchers’ call for projects (AAP-2014-033).
Conflict of interest
The authors have declared no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
The data on which the findings are based cannot be made freely available because of legal restrictions. Data used for the present study come from the French National Health Insurance databases and include many variables that, when combined, can lead to reidentifying subjects and then collecting health information on these individuals. Therefore, the French Data Protection Authority (CNIL) forbids making such data freely available. Access to the raw data of the French National Health Insurance must be requested from the National Health Data System (https://www.snds.gouv.fr/).
Code availability
An R script is provided as Electronic Supplementary Material to illustrate implementation.
Author contributions
EV, EC, SE, PTB, and IA planned and designed the study. EV drafted the manuscript and performed the research. FS and AP conducted the pharmacological assessment. All authors reviewed the manuscript and approved the final version.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Volatier, E., Salvo, F., Pariente, A. et al. High-Dimensional Propensity Score-Adjusted Case-Crossover for Discovering Adverse Drug Reactions from Computerized Administrative Healthcare Databases. Drug Saf 45, 275–285 (2022). https://doi.org/10.1007/s40264-022-01148-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-022-01148-5