Abstract
Background and Objectives
Although therapeutic drug monitoring of clozapine is recommended, its optimisation is often adjusted only on the basis of dosage. The aim of this study was to assess the link between clozapine plasma concentrations and clinical response by a meta-analysis of published studies and by an individual participant data meta-analysis.
Methods
We conducted a computerised search of bibliographic databases (EMBASE, PubMed, Clinical Trials, and Web of Science) to identify studies that assessed the relationship between clozapine serum or plasma concentrations and clinical efficacy. Using pooled data, we investigated the association between improvement of clinical outcome and clozapine or norclozapine plasma concentrations, the sum of clozapine and norclozapine plasma concentrations, and the coefficient of variation of clozapine plasma concentrations. Using available individual data, we assessed the relationship between clozapine plasma concentrations and clinical response (changes in the Brief Psychiatric Rating Scale score) and identified a threshold level for a favourable clinical response.
Results
Fifteen studies satisfied inclusion criteria. Our meta-analysis showed that responders had clozapine plasma concentrations that were, on average, 117 ng/mL higher than non-responders. The patients with plasma clozapine concentrations above the thresholds identified in each study had a higher likelihood of responding (odds ratio = 2.94, p < 0.001). Norclozapine plasma concentrations were not associated with a clinical response. The meta-analysis of individual data supported this result and confirmed the link between clozapine concentrations and a change in the Brief Psychiatric Rating Scale score and/or the probability of clinical response. Finally, with the analysis of the coefficient of variation of clozapine plasma concentrations, we found that a greater inter-individual fluctuation in plasma concentrations was associated with a loss of clinical response.
Conclusions
Our work confirmed that, in contrast to clozapine doses, clozapine plasma concentrations were related to a favourable clinical response, with a mean difference between responders and non-responders of 117 ng/mL. A threshold for a treatment response of 407 ng/mL was determined, with a high discriminatory capacity, and a sensitivity and specificity of 71% and 89.1%, respectively.




Similar content being viewed by others
References
World Health Organization (WHO), editor. Schizophrenia. WHO, 10 January 2022. Available from: https://www.who.int/news-room/fact-sheets/detail/schizophrenia. Accessed 21 Apr 2023.
Chong HY, Teoh SL, Wu DB-C, Kotirum S, Chiou C-F, Chaiyakunapruk N. Global economic burden of schizophrenia: a systematic review. Neuropsychiatr Dis Treat. 2016;12:357–73.
Kim D. Indicators of response to clozapine treatment. Mental Health Clin. 2015;5:74–7.
Nucifora FC, Woznica E, Lee BJ, Cascella N, Sawa A. Treatment resistant schizophrenia: clinical, biological, and therapeutic perspectives. Neurobiol Dis. 2019;131: 104257.
Kane J, Honigfeld G, Singer J, Meltzer H. Clozapine for the treatment-resistant schizophrenic: a double-blind comparison with chlorpromazine. Arch Gen Psychiatry. 1988;45:789–96.
Correll CU, Howes OD. Treatment-resistant schizophrenia: definition, predictors, and therapy options. J Clin Psychiatry. 2021;82.
Kerwin R. When should clozapine be initiated in schizophrenia? CNS Drugs. 2007;21:267–78.
Chakos M, Lieberman J, Hoffman E, Bradford D, Sheitman B. Effectiveness of second-generation antipsychotics in patients with treatment-resistant schizophrenia: a review and meta-analysis of randomized trials. Am J Psychiatry. 2001;158:518–26.
Lewis SW, Barnes TRE, Davies L, Murray RM, Dunn G, Hayhurst KP, et al. Randomized controlled trial of effect of prescription of clozapine versus other second-generation antipsychotic drugs in resistant schizophrenia. Schizophr Bull. 2006;32:715–23.
McEvoy JP, Lieberman JA, Stroup TS, Davis SM, Meltzer HY, Rosenheck RA, et al. Effectiveness of clozapine versus olanzapine, quetiapine, and risperidone in patients with chronic schizophrenia who did not respond to prior atypical antipsychotic treatment. Am J Psychiatry. 2006;163:600–10.
Schatzberg AF, DeBattista C. Manual of clinical psychopharmacology, 8th edn. Arlington, VA, US: American Psychiatric Publishing, Inc.; 2015. p. xxiii, 117.
Crilly J. The history of clozapine and its emergence in the US market: a review and analysis. Hist Psychiatry. 2007;18:39–60.
Wenthur CJ, Lindsley CW. Classics in chemical neuroscience: clozapine. ACS Chem Neurosci. 2013;4:1018–25.
Honigfield G. Effects of the clozapine national registry system on incidence of deaths related to agranulocytosis. Psychiatr Serv. 1996;47:52–6.
Warnez S, Alessi-Severini S. Clozapine: a review of clinical practice guidelines and prescribing trends. BMC Psychiatry. 2014;14:102.
Chakrabarti S. Clozapine resistant schizophrenia: newer avenues of management. World J Psychiatry. 2021;11:429–48.
Aissa A, Jouini R, Ouali U, Zgueb Y, Nacef F, El Hechmi Z. Clinical predictors of response to clozapine in Tunisian patients with treatment resistant schizophrenia. Compr Psychiatry. 2022;112: 152280.
Bersani FS, Capra E, Minichino A, Pannese R, Girardi N, Marini I, et al. Factors affecting interindividual differences in clozapine response: a review and case report. Hum Psychopharmacol. 2011;26:177–87.
Lane HY, Chang YC, Chang WH, Lin SK, Tseng YT, Jann MW. Effects of gender and age on plasma levels of clozapine and its metabolites: analyzed by critical statistics. J Clin Psychiatry. 1999;60:36–40.
de Leon J, Ruan C-J, Schoretsanitis G, De las Cuevas C. A rational use of clozapine based on adverse drug reactions, pharmacokinetics, and clinical pharmacopsychology. Psychother Psychosom. 2020;89:200–14.
Ruan C-J, Zang Y-N, Cheng Y-H, Wang C-Y, de Leon J. Around 3% of 1,300 levels were elevated during infections in a retrospective review of 131 Beijing hospital in-patients with more than 24,000 days of clozapine treatment. Psychother Psychosom. 2020;89:255–7.
Jerling M, Lindström L, Bondesson U, Bertilsson L. Fluvoxamine inhibition and carbamazepine induction of the metabolism of clozapine: evidence from a therapeutic drug monitoring service. Ther Drug Monit. 1994;16:368–74.
Spina E, de Leon J. Metabolic drug interactions with newer antipsychotics: a comparative review. Basic Clin Pharmacol Toxicol. 2007;100:4–22.
Hägg S, Spigset O, Mjörndal T, Dahlqvist R. Effect of caffeine on clozapine pharmacokinetics in healthy volunteers. Br J Clin Pharmacol. 2000;49:59–63.
Hasegawa M, Gutierrez-Esteinou R, Way L, Meltzer HY. Relationship between clinical efficacy and clozapine concentrations in plasma in schizophrenia: effect of smoking. J Clin Psychopharmacol. 1993;13:383–90.
Bondolfi G, Morel F, Crettol S, Rachid F, Baumann P, Eap CB. Increased clozapine plasma concentrations and side effects induced by smoking cessation in 2 CYP1A2 genotyped patients. Ther Drug Monit. 2005;27:539–43.
Facciolà G, Avenoso A, Spina E, Perucca E. Inducing effect of phenobarbital on clozapine metabolism in patients with chronic schizophrenia. Ther Drug Monit. 1998;20:628–30.
Djerada Z, Daviet F, Llorca P-M, Eschalier A, Saint-Marcoux F, Bentué-Ferrer D, et al. Suivi thérapeutique pharmacologique de la clozapine. Thérapie [Internet]. 2016 [cited 2023 Apr 24]. https://www.sciencedirect.com/science/article/pii/S0040595716311325.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–12.
Eriksen MB, Frandsen TF. The impact of Patient, Intervention, Comparison, Outcome (PICO) as a search strategy tool on literature search quality: a systematic review. J Med Libr Assoc. 2018;106:420–31.
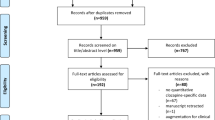
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. Open Med. 2009;3:e123–30.
Wong JO-Y, Leung S, Mak T, Ng RM, Chan K, Hon-Kee Cheung H, et al. Plasma clozapine levels and clinical response in treatment-refractory Chinese schizophrenic patients. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30:251–64.
Dettling M, Sachse C, Brockmöller J, Schley J, Müller-Oerlinghausen B, Pickersgill I, et al. Long-term therapeutic drug monitoring of clozapine and metabolites in psychiatric in- and outpatients. Psychopharmacology. 2000;152:80–6.
Hussein R, Gad A, Raines DA, Chaleby K, Al-Rawithi S, El-Yazigi A. Steady-state pharmacokinetics of clozapine in refractory schizophrenic Saudi Arabian patients. Pharm Pharmacol Commun. 1999;5:473–8.
Chong S-A, Tan C-H, Khoo Y-M, Lee H-S, Wong K-E, Ngui F, et al. Clinical evaluation and plasma clozapine concentrations in Chinese patients with schizophrenia. Ther Drug Monit. 1997;19:219–23.
Potkin SG, Bera R, Gulasekaram B, Costa J, Hayes S, Jin Y, et al. Plasma clozapine concentrations predict clinical response in treatment-resistant schizophrenia. J Clin Psychiatry. 1994;55 Suppl B:133–6.
Spina E, Avenoso A, Facciolà G, Scordo MG, Ancione M, Madia AG, et al. Relationship between plasma concentrations of clozapine and norclozapine and therapeutic response in patients with schizophrenia resistant to conventional neuroleptics. Psychopharmacology. 2000;148:83–9.
Perry PJ, Miller DD, Arndt SV, Cadoret RJ. Clozapine and norclozapine plasma concentrations and clinical response of treatment-refractory schizophrenic patients. Am J Psychiatry. 1991;148:231–5.
Siskind D, Sharma M, Pawar M, Pearson E, Wagner E, Warren N, Kisely S. Clozapine levels as a predictor for therapeutic response: a systematic review and meta-analysis. Acta Psychiatr Scand. 2021;144(5):422–32.
Overall JE, Gorham DR. The Brief Psychiatric Rating Scale. Psychol Rep. 1962;10:799–812.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36:1–48.
Nakagawa S, Poulin R, Mengersen K, Reinhold K, Engqvist L, Lagisz M, et al. Meta-analysis of variation: ecological and evolutionary applications and beyond. Method Ecol Evol. 2015;6:143–52.
Stewart GB, Altman DG, Askie LM, Duley L, Simmonds MC, Stewart LA. Statistical analysis of individual participant data meta-analyses: a comparison of methods and recommendations for practice. PLoS ONE. 2012;7: e46042.
Riley RD, Lambert PC, Abo-Zaid G. Meta-analysis of individual participant data: rationale, conduct, and reporting. BMJ. 2010;340: c221.
Zheng Y-P, Lin K-M, Zhao J-P, Zhang M-Y, Yong D. Comparative study of diagnostic systems: Chinese classification of Mental Disorders-Second Edition versus DSM-III-R. Compr Psychiatry. 1994;35:441–9.
Yuanguang C, Zhao J, Xie G. A study on serum concentration and clinical response to clozapine with different dose administration for treatment of schizophrenia. Chinese J Psychiatry. 1998;31(2):104–7.
VanderZwaag C, McGee M, McEvoy JP, Freudenreich O, Wilson WH, Cooper TB. Response of patients with treatment-refractory schizophrenia to clozapine within three serum level ranges. Am J Psychiatry. 1996;153:1579–84.
Kaladjian A, Bery B, Deturmeny E, Bruguerolle B. Clozapine monitoring: plasma or serum levels? Ther Drug Monit. 1999;21:327–9.
Hermida J, Paz E, Tutor JC. Clozapine and norclozapine concentrations in serum and plasma samples from schizophrenic patients. Ther Drug Monit. 2008;30:41–5.
Kronig MH, Munne RA, Szymanski S, Safferman AZ, Pollack S, Cooper T, et al. Plasma clozapine levels and clinical response for treatment-refractory schizophrenic patients. Am J Psychiatry. 1995;152:179–82.
Hiemke C, Bergemann N, Clement HW, Conca A, Deckert J, Domschke K, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51:9–62.
Schoretsanitis G, Kane JM, Correll CU, Marder SR, Citrome L, Newcomer JW, et al. Blood levels to optimize antipsychotic treatment in clinical practice: a joint consensus statement of the American Society of Clinical Psychopharmacology and the Therapeutic Drug Monitoring Task Force of the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie. J Clin Psychiatry. 2020;81:3649.
Llorca P-M, Lancon C, Disdier B, Farisse J, Sapin C, Auquier P. Effectiveness of clozapine in neuroleptic-resistant schizophrenia: clinical response and plasma concentrations. J Psychiatry Neurosci. 2002;27:30–7.
Miller DD, Fleming F, Holman TL, Perry PJ. Plasma clozapine concentrations as a predictor of clinical response: a follow-up study. J Clin Psychiatry. 1994;55 Suppl B:117–21.
Fabrazzo M, La Pia S, Monteleone P, Esposito G, Pinto A, De Simone L, et al. Is the time course of clozapine response correlated to the time course of clozapine plasma levels? A one-year prospective study in drug-resistant patients with schizophrenia. Neuropsychopharmacology. 2002;27:1050–5.
Yada Y, Kitagawa K, Sakamoto S, Ozawa A, Nakada A, Kashiwagi H, et al. The relationship between plasma clozapine concentration and clinical outcome: a cross-sectional study. Acta Psychiatr Scand. 2021;143:227–37.
Mauri M, Volonteri LS, Fiorentini A, Invernizzi G, Nerini T, Baldi M, et al. Clinical outcome and plasma levels of clozapine and norclozapine in drug-resistant schizophrenic patients. Schizophr Res. 2004;66:197–8.
PHD AK MD. Clozapine fixed dose steady state plasma levels and the relationship to the polymorphism of CYP1A2, CYP3A4, CYP3A5 and CYP2D6 in clinically stable schizophrenic adult patients. 2017 Oct. Report No.: NCT01663077. Available from: https://clinicaltrials.gov/ct2/show/NCT01663077. Accessed 21 Apr 2023.
Gaertner I, Gaertner HJ, Vonthein R, Dietz K. Therapeutic drug monitoring of clozapine in relapse prevention: a five-year prospective study. J Clin Psychopharmacol. 2001;21:305–10.
Stieffenhofer V, Saglam H, Schmidtmann I, Silver H, Hiemke C, Konrad A. Clozapine plasma level monitoring for prediction of rehospitalization schizophrenic outpatients. Pharmacopsychiatry. 2011;44:55–9.
Konecki C, Feliu C, Cazaubon Y, Giusti D, Tonye-Libyh M, Brixi H, et al. External evaluation of population pharmacokinetic models and Bayes-based dosing of infliximab. Pharmaceutics. 2021;13:1191.
Cazaubon Y, Talineau Y, Feliu C, Konecki C, Russello J, Mathieu O, et al. Population pharmacokinetics modelling and simulation of mitotane in patients with adrenocortical carcinoma: an individualized dose regimen to target all patients at three months? Pharmaceutics. 2019;11:E566.
Jerling M, Merlé Y, Mentré F, Mallet A. Population pharmacokinetics of clozapine evaluated with the nonparametric maximum likelihood method. Br J Clin Pharmacol. 1997;44:447–53.
Bhugra D, Bhui K. Ethnic and cultural factors in psychopharmacology. Adv Psychiatr Treat. 1999;5:89–95.
Lin KM, Finder E. Neuroleptic dosage for Asians. Am J Psychiatry. 1983;140:490–1.
Farooq S. Ethnicity and clozapine metabolism. Br J Psychiatry. 1998;173:87–87.
Matsuda KT, Cho MC, Lin KM, Smith MW, Young AS, Adams JA. Clozapine dosage, serum levels, efficacy, and side-effect profiles: a comparison of Korean-American and Caucasian patients. Psychopharmacol Bull. 1996;32:253–7.
Tan MSA, Honarparvar F, Falconer JR, Parekh HS, Pandey P, Siskind DJ. A systematic review and meta-analysis of the association between clozapine and norclozapine serum levels and peripheral adverse drug reactions. Psychopharmacology. 2021;238:615–37.
Skokou M, Karavia EA, Drakou Z, Konstantinopoulou V, Kavakioti C-A, Gourzis P, et al. Adverse drug reactions in relation to clozapine plasma levels: a systematic review. Pharmaceuticals (Basel). 2022;15:817.
Siskind D, Sharma M, Pawar M, Pearson E, Wagner E, Warren N, et al. Clozapine levels as a predictor for therapeutic response: a systematic review and meta-analysis. Acta Psychiatr Scand. 2021;144:422–32.
Acknowledgements
The authors thank Dr. Claire GOZALO for her proofreading and suggestions concerning the writing of the manuscript.
Funding
No funding was received for the preparation of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for the preparation of this article.
Conflicts of Interest/Competing Interests
Federica Tralongo, Céline Konecki, Catherine Feliu, Arthur Kaladjian and Zoubir Djerada have no conflicts of interest that are directly relevant to the content of this article.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of data and Material
The datasets generated and/oranalyzed during the current study are available from the corresponding authoron reasonable request.
Code Availability
Not applicable.
Authors’ Contributions
FT, ZD and AK wrote the manuscript; ZD designed the research; FT and ZD performed the research; FT, ZD, CK and AK analysed the data; FT, ZD, CK and CF contributed new reagents/analytical tools; and FT, ZD, AK, CK and CF validated the manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tralongo, F., Konecki, C., Feliu, C. et al. Association Between Clozapine Plasma Concentrations and Treatment Response: A Systematic Review, Meta-analysis and Individual Participant Data Meta-analysis. Clin Pharmacokinet 62, 807–818 (2023). https://doi.org/10.1007/s40262-023-01247-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-023-01247-1