Abstract
Background and objectives
Ultra-long-acting insulin degludec (DEG) has a longer duration of action and less daily variability relative to other basal insulin (BI), and thus may benefit patients with type 1 diabetes mellitus (T1DM). We examined the impact of switching BI to DEG on glycemic control and insulin dose in T1DM.
Methods
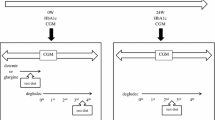
T1DM patients (n = 22; six male; mean age: 64.5 ± 12.6 years) receiving basal-bolus insulin therapy were included. Initially, the BI dose was replaced with DEG in a 1:1 ratio; 80–100% of the total dose was replaced with DEG for multiple basal insulin injections. DEG was titrated according to study protocol. Changes in HbA1c, daily insulin dose, glycemic self-monitored blood glucose variations, and hypoglycemia frequency were evaluated for 24 weeks.
Results
Once-daily DEG significantly decreased HbA1c levels when switched from once-daily BI (7.9 ± 0.8 vs. 7.5 ± 0.9%, p = 0.020) and maintained HbA1c when switched from twice-daily BI (8.5 ± 1.6 vs. 8.4 ± 1.2%, p = 0.457). The BI dose decreased by −7.8 ± 13.9% (p = 0.017) and −16.6 ± 16.9% (p = 0.050) when switched from once-daily BI and twice-daily BI, respectively. The total bolus insulin dose significantly decreased when switched from once-daily BI (21.7 ± 8.3 to 19.3 ± 8.8 U/day, p = 0.016) especially in the injection before breakfast and evening meal. Body weight and hypoglycemia frequency was not significantly different.
Conclusion
DEG improved glycemic control when switched from once-daily BI and maintained glycemic control when switched from twice-daily BI without increasing hypoglycemia.


Similar content being viewed by others
References
Jonassen I, Havelund S, Hoeg-Jensen T, Bjerre D, Ribel U, et al. Insulin degludec is a new generation ultra-long acting basal insulin with a unique mechanism of protraction based on multi-hexamer formation. Diabetes. 2010;59(suppl 1):A11.
Heise T, Hövelmann U, Nosek L, Hermanski L, Bøttcher SG, Haahr H. Comparison of the pharmacokinetic and pharmacodynamic profiles of insulin degludec and insulin glargine. Expert Opin Drug Metab Toxicol. 2015;11:1193–201.
Heise T, Hermanski L, Nosek L, Feldman A, Rasmussen S, Haahr H. Insulin degludec: four times lower pharmacodynamic variability than insulin glargine under steady-state conditions in type 1 diabetes. Diabetes Obes Metab. 2012;14:859–64.
Peyrot M, Rubin RR, Kruger DF, Travis LB. Correlates of insulin injection omission. Diabetes Care. 2010;33:240–5.
Ashwell SG, Gebbie J, Home PD. Twice-daily compared with once-daily insulin glargine in people with Type 1 diabetes using meal-time insulin aspart. Diabet Med. 2006;23:879–86.
Ohnishi Y, Iwamoto Y, Yoo SJ, Clauson P, Tamer SC, Park S. Insulin degludec compared with insulin glargine in insulin-naïve patients with type 2 diabetes: a 26-week, randomized, controlled, pan-Asian, treat-to-target trial. J Diabetes Investig. 2013;4:605–12.
Zinman B, Philis-Tsimikas A, Cariou B, Handelsman Y, Rodbard HW, Johansen T, et al. Insulin degludec versus insulin glargine in insulin-naive patients with type 2 diabetes: a 1-year, randomized, treat-to-target trial (BEGIN Once Long). Diabetes Care. 2012;35:2464–71.
Heller S, Buse J, Fisher M, Garg S, Marre M, Merker L, et al. Insulin degludec, an ultra-long acting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin as part in type 1 diabetes (BEGIN Basal-Bolus Type 1): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet. 2012;379:1489–97.
Kusunoki Y, Katsuno T, Miyakoshi K, Ikawa T, Nakae R, Ochi F, et al. Effects of switching from insulin glargine or detemir to insulin degludec in patients with type 1 diabetes mellitus. Diabetes Ther. 2013;4:461–72.
Nakamura T, Sakaguchi K, So A, Nakajima S, Takabe M, Komada H, et al. Effects of insulin degludec and insulin glargine on day-to-day fasting plasma glucose variability in individuals with type 1 diabetes: a multicentre, randomised, crossover study. Diabetologia. 2015;58:2013–9.
Thalange N, Deeb L, Iotova V, Kawamura T, Klingensmith G, Philotheou A, et al. Insulin degludec in combination with bolus insulin aspart is safe and effective in children and adolescents with type 1 diabetes. Pediatr Diabetes. 2015;16:164–76.
Garber AJ, King AB, Del Prato S, Sreenan S, Balci MK, Muñoz-Torres M, et al. Insulin degludec, an ultra-long acting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet. 2012;379:1498–507.
Bode BW, Buse JB, Fisher M, Garg SK, Marre M, Merker L, et al. Insulin degludec improves glycaemic control with lower nocturnal hypoglycaemia risk than insulin glargine in basal-bolus treatment with mealtime insulin aspart in Type 1 diabetes (BEGIN(®) Basal-Bolus Type 1): 2-year results of a randomized clinical trial. Diabet Med. 2013;30:1293–7.
Hollander P, King AB, Del Prato S, Sreenan S, Balci MK, Muñoz-Torres M, et al. Insulin degludec improves long-term glycaemic control similarly to insulin glargine but with fewer hypoglycaemic episodes in patients with advanced type 2 diabetes on basal-bolus insulin therapy. Diabetes Obes Metab. 2015;17:202–6.
Heller S, Mathieu C, Kapur R, Wolden ML, Zinman B. A meta-analysis of rate ratios for nocturnal confirmed hypoglycaemia with insulin degludec vs. insulin glargine using different definitions for hypoglycaemia. Diabet Med. 2016;33:478–87.
Abdelhafiz AH, Rodríguez-Mañas L, Morley JE, Sinclair AJ. Hypoglycemia in older people—a less well recognized risk factor for frailty. Aging Dis. 2015;6:156–67.
Li FF, Liu BL, Zhu HH, Li T, Zhang WL, Su XF, et al. Continuous glucose monitoring in newly diagnosed type 2 diabetes patients reveals a potential risk of hypoglycemia in older men. J Diabetes Res. 2017;2017:2740372.
Acknowledgements
The Center for Diabetes and Endocrinology, Kitano Hospital receives financial support from Novo Nordisk Pharma, MSD Pharmaceutical, Novartis Pharma, Daiichi-Sankyo, and Astellas Pharma. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Some of the results described herein were presented at the annual scientific meeting of the American Diabetes Association in 2014.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external source of funding was used to conduct this research.
Conflict of interest
YH has received speaker’s fees from Novo Nordisk Pharma. The other authors declare that they have no conflict of interests. No other potential conflicts of interest relevant to this study exist.
Ethical approval
This study was approved by the ethics committee of our institute and all procedures performed in studies involving human participants were in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
All participants provided informed consent upon study enrollment.
Rights and permissions
About this article
Cite this article
Hamamoto, Y., Honjo, S., Fujimoto, K. et al. Basal and Bolus Insulin Dose Changes after Switching Basal Insulin to Insulin Degludec in Patients with Type 1 Diabetes Mellitus: A Pilot Study. Clin Drug Investig 37, 845–852 (2017). https://doi.org/10.1007/s40261-017-0541-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-017-0541-0