Abstract
Kidney transplantation is a life-saving strategy for patients with end-stage renal disease. Although progress has been made in the field of transplantation medicine in recent decades in terms of surgical techniques and immunosuppression, long-term organ survival remains a challenge. Also, for reasons of organ shortage, there is an unmet need for new therapeutic approaches to improve the long-term survival of transplants. There is increasing evidence that the complement system plays a crucial role in various pathological events after transplantation, including ischemia/reperfusion injury as well as rejection episodes. The complement system is part of the innate immune system and plays a crucial role in the defense against pathogens but is also involved in tissue homeostasis. However, the tightly regulated complement system can become dysregulated or activated by non-infectious stimuli, then targeting the organism’s own cells and leading to inflammatory tissue damage that exacerbates injury. In this review, we will highlight the role of the complement system after transplantation and discuss ongoing and potential therapeutic approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Kidney transplantation is the best life-saving strategy for patients with end-stage renal disease. Achieving long-term graft survival is still challenging and requires new therapies. |
Complement-mediated injury is central in renal transplantation and occurs early during ischemia/reperfusion injury but is also involved in transplant rejection. |
The complement system was targeted in clinical studies at different levels of the complement cascade to prevent delayed graft function (DGF) and antibody-mediated rejection (ABMR). |
1 Introduction
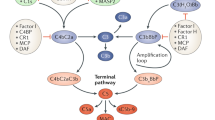
The complement system is an essential component of the innate immune system, involved in (i) opsonization, (ii) stimulation of different inflammatory pathways and (iii) osmolytic lysis of pathogens and damaged cells in numerous diseases, especially in inflammatory kidney disease [1]. This highly regulated system consists of >40 fluid-phase and surface-bound factors including activating proteases, regulating inhibitors, pore-forming proteins and complement receptors (Fig. 1). Three distinct activation pathways are known: (i) the classical pathway, activated by any structure that is recognized by C1q [2], (ii) the lectin pathway, activated when saccharide patterns are recognized by pattern recognition complexes [3] and (iii) the alternative pathway, activated through spontaneous hydrolysis of C3 [4].
For the activation of the classical pathway, the initiator molecule C1q recognizes a big variety of target molecules including immunoglobulin (Ig)G and IgM, C-reactive proteins, bacterial and viral proteins, apoptotic cells and others [2, 5]. A tetramer of the two serine proteases C1r and C1s binds to C1q and thereby forms the C1 complex [6]. The lectin pathway can be initiated by either mannose-binding-lectin (MBL), ficolin 1-3 or collectin 10 and 11, which recognize saccharides on the surface of pathogen- or danger-associated molecular patterns (PAMPs, DAMPs) [3, 7]. Upon binding of the recognition molecules to the specific carbohydrates, serine protease MASP-2 is activated [8]. In the classical as well as in the lectin pathway, the activation of the serine proteases leads to the cleavage of C4 and C2 into C4a and b and C2a and b [6, 8]. C2a attaches to C4b, whereby the complex becomes enzymatically active and forms the C3 convertase [9]. The alternative pathway has two different functions: it amplifies the C3 level activated by the other two pathways and it induces an independent activation [8]. C3 is constantly hydrolyzed and the resulting C3b binds to target molecules like foreign cells and bacteria. Factor B binds to C3b and is thereafter cleaved by Factor D, forming the C3 convertase C3bBb [10].
At this point, the three pathways merge. Both C3 convertases, the C4b2a of the classical and the lectin pathway and the C3bBb of the alternative pathway cleave C3 and release C3a and C3b [11]. C3a is an anaphylatoxin, and as such a pro-inflammatory chemoattractant that activates and recruits inflammatory cells including neutrophils and mast cells [12]. C3b can on the one hand opsonize target cells and on the other hand bind to the C3 convertase. Upon binding of C3b, the C3 convertase switches its specificity to the binding of C5 and becomes a C5 convertase [13]. The C5 convertase cleaves C5 and releases C5a and C5b. While C5a is an anaphylatoxin similar to C3a, C5b recruits the complement factors C6, C7, C8 and C9 to form the membrane-attack-complex (MAC) [14]. The MAC forms pores in the membrane of target cells disturbing calcium passage and thereby leading to apoptosis of these respective cells. The pore size is determined by the number of C9 molecules assembling in the MAC, which can vary from 2 up to 18 C9 molecules [15]. To prevent overactivation of the complement system, it is regulated by soluble (e.g. complement factor H [CFH]) and membrane bound (e.g. CD46) endogenous inhibitors that act at different levels of the complement cascade. Early activation of the classical and lectin-mediated pathways can be inhibited by the C1-esterase inhibitor SERPING1. CD35, also known as CR1, and CD55 can act as decoy receptors, limiting the activation of complement convertases. The formation of the C5 convertases C3bBbC3b in the alternative pathway is inhibited by soluble CFH and C4b2aC3b of the other two activation pathways by the surface molecule CD46. In the terminal complement cascade, CD59 inhibits the formation of the MAC. Of these endogenous complement inhibitors, SERPING1 is being tested therapeutically as a C1 inhibitor [16] and double transgenic pigs expressing human CD55 and CD59 were generated to be used in the future in xenotransplantation [17].
2 Involvement of Complement in Adverse Outcome of Renal Transplantation
2.1 Ischemia/Reperfusion Injury and Delayed Graft Function
During transplantation, complement is involved at different time points. Relevant factors are the donor type (deceased or living donor), ischemia/reperfusion (cold and warm ischemia time) but also antigen mismatch, the occurrence of donor-specific antibodies and rejection events. Many components of the complement cascade are primarily formed in the liver, but can also be produced locally in response to a damaging stimulus [18]. The relevance of locally produced complement has been demonstrated in a mouse transplantation model using C3-deficient isografts showing only mild reperfusion injury compared with wildtype grafts when transplanted in a C3-positive recipient [19]. Gene expression analyses in biopsies taken before transplantation revealed significantly higher expression of various complement genes in kidneys from deceased donors compared with living donors [20]. Furthermore, complement factors are expressed at significantly higher levels in deceased donors at later time points after renal transplantation [20, 21], and correlate significantly with cold ischemia time [21].
The importance of complement activation in mediating ischemia/reperfusion (I/R)-induced tissue damage has been demonstrated in animal models using mice deficient for a particular complement factor or by using different complement inhibitors [22,23,24,25,26]. In this regard, complement activation does not appear to be restricted to one pathway, as both inhibition of the alternative pathway by factor B deficiency [24] or anti-factor B antibodies [27], and C1-inhibitor therapy were successful in reducing I/R damage [28]. The C1-inhibitor SERPING1 is a serine esterase inhibitor that blocks C1s and C1r proteases of the classical pathway and MASP2 of the lectin pathway [29].There is an increasing body of evidence that the lectin pathway is of particular importance in mediating I/R injury [30,31,32,33]. The pattern recognition molecule collectin-11 recognizes hypoxia-induced fucosylated ligands, which allow the formation of a complex with MASP2 and subsequent activation of the lectin pathway. Consequently, deficiency for collectin-11 but also treatment with l-fucose prevented I/R injury in mice [34, 35]. Delayed graft function (DGF) is a major consequence of a profound kidney injury mediated by different factors resulting from unstable hemodynamics, impaired homeostasis and circulating DAMPs from injured cells due to brain death, hypoxia or related to the primary disease. Reperfusion of the donor organ in the recipient exacerbates organ damage. Complement activation was shown to be observed in brain death donors before I/R as demonstrated by increased expression of complement factors in donor organs [36] and systemic complement activation [37]. Accordingly, sC5b-9 levels can be used as a sensitive marker to predict DGF [38] and donor treatment or ex vivo complement inhibition is thus a promising way to prevent the earliest effects of complement activation. This concept was successfully tested in a rat model of kidney transplantation [39] and was also used in the EMPIRIKAL study using the C3 inhibitor mirococept [40] (Table 1). Complement-mediated injury can be a direct consequence of the formation of the MAC or indirectly caused by enhancement of the inflammatory response by anaphylatoxins C3a and C5a. The anaphylatoxins are involved in leukocyte chemotaxis and activation as well as inducers of the production of pro-inflammatory mediators like cytokines and chemokines [41]. The C5a/C5aR1 axis has been shown to be critically involved in mediating I/R injury [42, 43]. Moreover, C5a/C5aR2 signaling in renal I/R is involved in activation of inflammatory cells but not in chemotaxis [44]. Thus, the C5a/C5aR axis represents a possible target for treatment of I/R injury.
2.2 Antibody-Mediated Rejection
Antibody-mediated rejection (ABMR) is the leading cause of subsequent kidney transplant failure [45], but efficient treatment options are lacking. Components of the complement system are involved in both regulation of the humoral response and ABMR-mediated allograft injury. In ABMR, HLA IgG alloantibodies produced by plasma cells bind to the donor antigens on graft microvasculature, leading to complement activation, margination and activation of inflammatory cells, and endothelial cell injury, sometimes with intimal arteritis. Donor-specific antibodies (DSA) play a key role in mediating ABMR pathology and are present at the time of transplantation in sensitized patients who have developed DSA due to a previous transplant, but may also be formed by the recipient later after transplantation. When DSAs bind to surface antigens, such as HLA antigens on endothelial cells, C1q recognize these immune complexes and can initiate the complement cascade via the classical pathway, ultimately leading to MAC formation and endothelial cell lysis [46]. This process is known as complement-dependent cytotoxicity. Additional to activation via the classic pathway, activation can also occur via the lectin pathway; in this case, sugar residues on IgM and IgG antibodies are recognized by mannose-binding lectin (MBL), for example, and initiate the activation cascade [47].
Not all antibodies have the ability to bind complement. In a study with more than 1000 transplanted patients on the presence of complement fixing antibodies it was shown that complement-binding donor-specific anti-HLA antibodies are helpful for the diagnosis and risk assessment of transplant rejection. The presence of C1q-fixing DSAs was associated with an increased rate of ABMR, a more severe graft injury phenotype with more extensive microvascular inflammation, and increased deposition of complement fraction C4d within graft capillaries [48]. Detection of complement-fixing DSAs allowed detection of ABMR also in C4d-negative cases [48]. Capillary C4d deposits have been established as a marker for ABMR and have been included in the Banff classification for ABMR diagnosis [49]. However, C4d deposits are not ABMR-specific and have been observed in other renal diseases [50], and the absence of C4d deposits is by no means an exclusion criterion for ABMR [51].
Complement is involved not only in antibody-mediated injury but also in the regulation of antibody production [52]. Binding of C3d-opsonized antigen from injured cells to CR2 on B cells promotes B-cell activation and antibody production by lowering the activation threshold [53]. Memory B-cell maintenance is achieved by binding of C3d-fixed antigens to CR2 on the surface of follicular dendritic cells [54]. B cells in the marginal zone can acquire intact major histocompatibility complexes from dendritic cells by complement-dependent trogocytosis for presentation to T cells [55]. Activated B cells in germinal centers receive co-stimulatory signals from T helper cells. In these stimulated germinal center B cells, the expression of complement regulators on the surface shifts, enabling activation of complement receptors on germinal center B cells, which is required for affinity maturation [56]. Positively selected B cells with high affinity can then differentiate into plasma cells that produce antibodies reactive with the donor HLA. Subsequent sublytic complement attack may stimulate endothelial cells to activate CD4+ and CD8+ T cells, promoting cellular and humoral rejection [57]. Anaphylatoxins C3a and C5a also play a role in ABMR by indirectly participating in the activation of B cells and polarization of T cells [58].
3 Drugable Complement Targets in Kidney Transplantation
The complement cascade can be inhibited at different levels. The activation of the different complement pathways can either be inhibited separately early in the cascades, or the common final pathway can be targeted further downstream. The inhibition of complement activation at the beginning of a pathway has the advantage that all downstream components of a specific pathway can be blocked, preventing the formation of reactive cleavage products such as anaphylatoxins. In addition, by inhibition of one selected complement pathway, patients could retain complement-mediated defense against infection, a consideration that might be important in immunosuppressed transplant recipients, by sparing the other pathways. However, assuming at least two, maybe even three complement pathways are involved in the pathogenesis of transplant-related injury [59], complement blockade is incomplete and possibly not effective enough if only one pathway is inhibited. As all complement pathways terminate in a common pathway, other approaches use the inhibition of downstream complement factors such as C3 or C5.
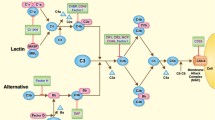
In clinical trials for kidney transplantation, C1 and C5 inhibitors and a C3 inhibitor have been tested so far. The goal was to prevent early graft failure by DGF (Table 1) or early and late rejection, especially ABMR (Table 2). These studies will be discussed in detail later. In addition to these inhibitors, which have already been tested in the setting of renal transplantation, others are available that have previously been applied in other complement-mediated diseases. Complement inhibitors not previously used in the transplant setting target the initiators of the lectin pathway (MASP-2 [60]) and the alternative pathway (FD [61,62,63], FB [64]), the activation of C3 [65, 66], the activity of C3/C5 convertases [67, 68], the amplification of the alternative pathway [69], or prevent signaling through the C5aR1 [70, 71]. An overview of these therapeutics is given by Mastellos et al. (2019) [72]. Since these therapeutics can potentially be used in transplantation, we have summarized the targets and therapeutics in Figure 2.
Overview of drugable complement targets. A Target: C1s, Drug(s): Sutimlimab, Mode of action: Inhibition of C1s protease. B Target: C1s/r, MASPs, Drug(s): C1-INH, CYNRIZE, berinert, ruconest, Mode of action: CP/LP inhibition, other serine proteases. C Target: Collectin-11, Drug(s): l-Fucose, Mode of action: Saturation of Collectin-11 binding. D Target: MASP-2, Drug(s): OMS721, Mode of action: Inhibition of MASP-2. E Target: FB, Drug(s): LNP023, Mode of action: Inhibition of AP C3 convertase. F Target: FD, Drug(s): ACH-4471, lampalizumab, Mode of action: Inhibition of AP C3 convertase. G Target: C3/C5 convertases, Drug(s): Mirococept, Mode of action: Inhibition of all C3/C5 convertases. H Target: C3, Drug(s): AMY-101, APL-2, Mode of action: Inhibition of C3 activation. I Target: Properdin, Drug(s): CLG561, Mode of action: Inhibition of AP amplification. J Target: C5, Drug(s): Eculizumab, ravulizumab, SKY59/RO7112689, tesidolumab; pozelimab, ABP959, SB12, Mode of action: Blockage of C5 activation; Drug(s): Coversin, Mode of action: Inhibition of C5 activation, Drug(s): Zilucoplan, Mode of action: Allosteric inhib. of C5 activation. K Target: C5a, Drug(s): IFX-1, Mode of action: Blocks binding of C5a to C5aR1. L Target: C5aR1, Drug(s): Avacopan, Mode of action: Antagonist of C5aR1 receptor; Drug(s): IPH5, Blockade of C5aR1 signaling. C1-INH C1 esterase inhibitor, MAC membrane-attack-complex, MASP mannose-binding lectin-associated serine protease
4 Past and Ongoing Studies on Complement Inhibition in Renal Transplantation
4.1 Complement Inhibition to Prevent Delayed Graft Function in Clinical Trials
In clinical trials, two main inhibitors have been tested to prevent DGF after renal transplantation: (i) eculizumab, a recombinant humanized monoclonal antibody targeting C5, a key molecule of terminal complement activation, and (ii) C1-esterase inhibitors, also known as SERPING1, either purified from plasma or recombinantly produced and provided by different companies, inhibiting early activation of the classical and lectin-mediated pathway. An overview of all clinical trials investigating complement inhibition in early I/R injury to prevent DGF after transplantation is shown in Table 1.
Studies investigating the efficacy of eculizumab in preventing DGF initially used a single dose of eculizumab 1200 mg in a pilot study (ClinicalTrials.gov identifier: NCT01403389), which was administered before reperfusion. However, this study was stopped after an interim evaluation and the treatment was changed to another pilot study that used an additional eculizumab dose 12–24 h after transplantation (NCT0191934). Eculizumab did not significantly reduce the number of patients with DGF in either of these two pilot studies or in the PROTECT trial (NCT02145182), which included a total of 288 patients [73]. Therefore, the second pilot study was also terminated before planned enrollment. In contrast, pediatric kidney transplant patients who received eculizumab showed better early graft function, less arterial hyalinosis and chronic glomerulopathy in protocol biopsies taken at day 30, and after 1 and 3 years. However, four children in the eculizumab group lost their graft during a flu-like infection, while none of the children in the control group lost their graft (NCT01756508) [74].
Other studies have investigated the efficacy of a C1 inhibitor, which inhibits activation of the classical and lectin pathways, in preventing DGF (Table 1). At the moment, only one phase I/II study using the C1-estase inhibitor (n = 35) compared with placebo (n = 35) has been completed (NCT02134314). Similar to the C5 inhibitor studies, therapy was given directly on the day of transplantation and 24 hours after surgery. Regarding the primary outcome, the occurrence of DGF, defined as the need for dialysis within the first week after transplantation, no difference was observed between the groups (C1-esterase inhibitor 44% vs placebo 60%) [75]. However, the duration of dialysis was significantly shortened in the C1-esterase inhibitor group [75]. In the follow-up of this study, 3.5 years after transplantation, a significantly better eGFR was determined in the C1-esterase inhibitor group (56 mL/min per 1.73 m2 vs 35 mL/min per 1.73 m2) [76].
Further studies with C1 esterase inhibitors to reduce the incidence of DGF with higher (NCT04696146, NCT02435732) and more numerous doses (NCT03791476) of the inhibitor are planned or currently have an unclear status (Table 1). Inhibition of the complement cascade at the level of C3 was investigated in another study perfusing the grafts with the CR1 analog mirococept ex vivo instead of the standard cold perfusion fluid (Soltran®) (ISRCTN49958194) [77]. Primarily, seven study arms with different inhibitor concentrations were planned; however, the study was stopped after planned interim evaluation with the first dose of 10 mg (n = 53) versus placebo (n = 30), because the treatment did not prevent DGF. Instead of continuing the study, a re-dosing study was performed using pig kidneys to determine the saturation range for the inhibitor mirococept. The optimal dose for the pig kidney was determined to be 80 mg of mirococept, which is equivalent to a dose of 120 mg for the human kidney [40]. Ex vivo administration of mirococept at this dose was safe and feasible and provides the basis for future new studies on the treatment of DGF in deceased donor kidney transplants.
4.2 Complement Inhibition to Prevent Antibody-Mediated Rejection and Chronic Complement-Mediated Injury in Clinical Trials
Similar to the studies focusing on prevention of DGF, mainly eculizumab and C1-esterase inhibitors have been used in clinical trials for the prevention of ABMR, but they were administered over a longer period of time and more frequently (Table 2). In addition, these studies primarily treated patients who were at particularly high risk of developing ABMR due to an unfavorable cross match or pre-existing donor-specific antibodies. In some cases, however, the study design was so specific that the studies were terminated because patients meeting the inclusion criteria were lacking (NCT01106027; NCT01095887; NCT03221842). A group of 26 highly sensitized recipients of living donor renal transplants, who received eculizumab post-transplant, was compared with a historical control group of 51 sensitized patients treated with a similar plasma exchange protocol without eculizumab to test the incidence of biopsy-proven ABMR in the first 3 months post-transplant. The incidence of ABMR was significantly lower in the eculizumab group compared with the control group (7.7% [2/26] vs 41.2% [21/51]; NCT00670774) [78]. In addition to decreased ABMR, chronic transplant glomerulopathy was detected after 1 year in 6.7% (1/15) of graft biopsies from eculizumab-treated recipients and in 35.7% (15/42) of control patients (p = 0.044) [78]. However, a later phase II, randomized, multicenter, open label, two-arm, parallel group study including 51 patients per arm at first glance showed no protective effect of eculizumab treatment for the primary endpoint of treatment failure rate and was therefore terminated. Only a reassessment of the study including grade I ABMR finally showed a slightly significant difference between the groups, indicating a potential benefit of eculizumab compared with standard operation of care to prevent active ABMR in sensitized recipients [79]. In a single-arm study of recipients who received a deceased donor transplant and had preformed donor-specific antibodies, the primary endpoint (a composite of biopsy‐proven grade II/III ABMR, graft loss, death, or loss to follow‐up, within 9 weeks post-transplant) was observed in 8.8% of eculizumab-treated patients and was thereby lower than expected for standard care (40%) [80].
While the studies described above have investigated whether ABMR after transplantation can be prevented by eculizumab, smaller studies have also investigated whether the course of already developed ABMR can be favorably influenced. Patients were treated with eculizumab immediately after diagnosis of biopsy-proven ABMR, followed by five additional weekly doses (n = 7) and compared with standard therapy (n = 4). Because eculizumab given as monotherapy did not significantly improve eGFR within 3 months, the study was terminated (NCT01895127) and a planned study of complement inhibition for treatment of subclinical ABMR was withdrawn (NCT02113891). However, in the terminated study, the timing of complement inhibitory therapy after transplantation was not determined, but may be important for success, as suggested by a retrospective observational study reporting effective treatment of ABMR in the first month after transplantation [81]. For the treatment of chronic complement-mediated renal injury after kidney transplantation, 11 patients were treated with slightly lower single doses of eculizumab (900 mg) for 6 months. Compared with the control group (n = 5), eGFR tended to be improved, but endothelial cell damage was not reduced (NCT01327573) [82].
In contrast to the studies with the C5 inhibitor eculizumab, of which many were designed to prevent ABMR, there is only one pilot study with a C1 esterase inhibitor (C1-INH). In this study with highly HLA-sensitized patients, none of the ten study participants developed DGF or ABMR in the C1-INH group (n = 10), while in the control group, four out of ten developed DGF and one developed ABMR. However, in further follow-up, two cases of ABMR occurred in each of the two groups [83]. The combination of standard therapy with antibody reduction and C1-INH may be useful for the prevention of ABMR, but further controlled studies are needed. Although a C1-INH trial to treat ABMR was withdrawn in 2012 due to reduced incidence of ABMR by recent improvements in clinical practice (NCT01035593), several new trials have still been initiated. The largest study with a total of 39 participants, 19 of whom received standard therapy for ABMR and 20 of whom also received C1-INH, showed in an interim evaluation no improvement in protection against transplant glomerulopathy and met the pre-specified criteria for futility (NCT02547220). Previously, a pilot study with the same C1-INH had also shown no differences in the primary endpoint, meaning no difference at day 20 after therapy initiation with respect to pathology or graft survival. However, no transplant glomerulopathy and a trend toward improved renal function in the C1-INH group was found at the 6-month biopsy (NCT01147302) [84]. In a first study with sutimlimab, which exclusively inhibits the classical pathway, five of eight C4d-positive recipients turned C4d negative in follow-up biopsies, while another two recipients showed a substantial decrease in C4d scores. There was, however, no change in renal inflammation, gene expression patterns, donor-specific antibody levels, or kidney function [85].
4.3 Side Effects of Complement-Targeted Therapies and Combination With Other Drugs
In the treatment of transplant patients, complement inhibitors are only used as monotherapy when given before transplantation (e.g. to avoid DGF). As with any other immunosuppressive therapy, it is a balancing act to, on the one hand, protect the graft from detrimental allogenic immune reactions and consequent graft loss, but, on the other hand, to avoid side effects, including primarily infections that also endanger the transplant and the patient. The complement system plays an important role in the opsonization of pathogens. One threatening side effect of eculizumab therapy is, therefore, the occurrence of meningococcal infections. As a result, appropriate vaccination is a critical prerequisite before treatment. However, vaccination of immunosuppressed patients is challenging and sometimes fails [86]. One study reported graft loss in four eculizumab-treated patients due to flu-like infection [74]. So far, drug-related serious adverse events associated with complement-inhibiting therapies have been observed only rarely, so at least the C1 and C5 inhibitors studied in several clinical trials can be considered safe. This is probably due to the fact that there are different activation pathways of the complement system and that the available complement inhibitors always block only one part of the cascade. Complement-inhibiting therapies aim to suppress the innate immune defense during transplantation and are used in addition to standard therapy including tacrolimus, mycophenolate mofetil and prednisolone, if appropriate, which suppress cell-mediated immune defense. Since complement factors also stimulate immune cells, it may be possible to lower the doses of standard therapy and thereby minimize side effects when combining them with complement inhibitors. Monitoring of complement activation in the plasma may help to determine an optimal and individual treatment of patients, allowing a tailored immune suppression [87].
5 Conclusion
In the field of transplantation medicine, several studies have already been conducted assessing the blockade of the complement system. The aim of these studies was to prevent I/R and DGF in the early phase or ABMR. Eculizumab, a C5 inhibitor, and C1 esterase inhibitor were most commonly used. A number of different complement inhibitors have already been tested in clinical trials in the context of other diseases and could therefore also be a treatment option in the future, both concomitantly and after transplantation. Since only a few studies with larger numbers of cases exist, studies with well-defined study arms and larger numbers of patients are needed to investigate which patient groups can particularly benefit from complement inhibition therapy. As complement therapies are very expensive, future studies must confirm that they have a significant benefit on graft survival compared with established treatments to justify the costs. Moreover, in some settings the high expense may prevent the use of the drugs when resources are limited. Hopefully, in the future the detrimental effects of complement activation in the transplantation process can be reduced by targeted, organ or cell-specific complement therapies, without fearing loss of the desirable systemic defense mediated by the complement system.
References
Couser WG. Basic and translational concepts of immune-mediated glomerular diseases. J Am Soc Nephrol. 2012;23:381–99. https://doi.org/10.1681/asn.2011030304.
Kojouharova M, Reid K, Gadjeva M. New insights into the molecular mechanisms of classical complement activation. Mol Immunol. 2010;47:2154–60. https://doi.org/10.1016/j.molimm.2010.05.011.
Dobo J, Pal G, Cervenak L, Gal P. The emerging roles of mannose-binding lectin-associated serine proteases (MASPs) in the lectin pathway of complement and beyond. Immunol Rev. 2016;274:98–111. https://doi.org/10.1111/imr.12460.
Pangburn MK, Schreiber RD, Muller-Eberhard HJ. Formation of the initial C3 convertase of the alternative complement pathway. Acquisition of C3b-like activities by spontaneous hydrolysis of the putative thioester in native C3. J Exp Med. 1981;154:856–67.
Kishore U, Ghai R, Greenhough TJ, Shrive AK, Bonifati DM, Gadjeva MG, Waters P, Kojouharova MS, Chakraborty T, Agrawal A. Structural and functional anatomy of the globular domain of complement protein C1q. Immunol Lett. 2004;95:113–28. https://doi.org/10.1016/j.imlet.2004.06.015.
Bally I, Rossi V, Lunardi T, Thielens NM, Gaboriaud C, Arlaud GJ. Identification of the C1q-binding Sites of Human C1r and C1s: a refined three-dimensional model of the C1 complex of complement. J Biol Chem. 2009;284:19340–8. https://doi.org/10.1074/jbc.M109.004473.
Matsushita M, Endo Y, Fujita T. Structural and functional overview of the lectin complement pathway: its molecular basis and physiological implication. Arch Immunol Ther Exp. 2013;61:273–83. https://doi.org/10.1007/s00005-013-0229-y.
Hein E, Garred P. The lectin pathway of complement and biocompatibility. Adv Exp Med Biol. 2015;865:77–92. https://doi.org/10.1007/978-3-319-18603-0_5.
Gaya da Costa M, Poppelaars F, Berger SP, Daha MR, Seelen MA. The lectin pathway in renal disease: old concept and new insights. Nephrol Dial Transplant. 2018. https://doi.org/10.1093/ndt/gfy073.
Sarma JV, Ward PA. The complement system. Cell Tissue Res. 2011;343:227–35. https://doi.org/10.1007/s00441-010-1034-0.
Sahu A, Lambris JD. Structure and biology of complement protein C3, a connecting link between innate and acquired immunity. Immunol Rev. 2001;180:35–48. https://doi.org/10.1034/j.1600-065x.2001.1800103.x.
Verschoor A, Karsten CM, Broadley SP, Laumonnier Y, Köhl J. Old dogs-new tricks: immunoregulatory properties of C3 and C5 cleavage fragments. Immunol Rev. 2016;274:112–26. https://doi.org/10.1111/imr.12473.
Kinoshita T, Takata Y, Kozono H, Takeda J, Hong KS, Inoue K. C5 convertase of the alternative complement pathway: covalent linkage between two C3b molecules within the trimolecular complex enzyme. J Immunol (Baltimore, Md: 1950). 1988;141:3895–901.
Merle NS, Church SE, Fremeaux-Bacchi V, Roumenina LT. Complement system part I—molecular mechanisms of activation and regulation. Front Immunol. 2015;6:262. https://doi.org/10.3389/fimmu.2015.00262.
Cole DS, Morgan BP. Beyond lysis: how complement influences cell fate. Clin Sci (Lond). 2003;104:455–66. https://doi.org/10.1042/cs20020362.
Berger M, Lefaucheur C, Jordan SC. Update on C1 esterase inhibitor in human solid organ transplantation. Transplantation. 2019;103:1763–75. https://doi.org/10.1097/tp.0000000000002717.
Ménoret S, Plat M, Blancho G, Martinat-Botté F, Bernard P, Karam G, Tesson L, Renaudin K, Guillouet P, Weill B, Chéreau C, Houdebine LM, Soulillou JP, Terqui M, Anegon I. Characterization of human CD55 and CD59 transgenic pigs and kidney xenotransplantation in the pig-to-baboon combination. Transplantation. 2004;77:1468–71. https://doi.org/10.1097/01.tp.0000111758.35048.ea.
Li K, Sacks SH, Zhou W. The relative importance of local and systemic complement production in ischaemia, transplantation and other pathologies. Mol Immunol. 2007;44:3866–74. https://doi.org/10.1016/j.molimm.2007.06.006.
Farrar CA, Zhou W, Lin T, Sacks SH. Local extravascular pool of C3 is a determinant of postischemic acute renal failure. FASEB J. 2006;20:217–26. https://doi.org/10.1096/fj.05-4747com.
Naesens M, Li L, Ying L, Sansanwal P, Sigdel TK, Hsieh SC, Kambham N, Lerut E, Salvatierra O, Butte AJ, Sarwal MM. Expression of complement components differs between kidney allografts from living and deceased donors. J Am Soc Nephrol. 2009;20:1839–51. https://doi.org/10.1681/asn.2008111145.
Vonbrunn E, Ries T, Söllner S, Müller-Deile J, Büttner-Herold M, Amann K, Daniel C. Multiplex gene analysis reveals T-cell and antibody-mediated rejection-specific upregulation of complement in renal transplants. Sci Rep. 2021;11:15464. https://doi.org/10.1038/s41598-021-94954-3.
Howard MC, Nauser CL, Farrar CA, Sacks SH. Complement in ischaemia-reperfusion injury and transplantation. Semin Immunopathol. 2021;43:789–97. https://doi.org/10.1007/s00281-021-00896-3.
Zhou W, Farrar CA, Abe K, Pratt JR, Marsh JE, Wang Y, Stahl GL, Sacks SH. Predominant role for C5b–9 in renal ischemia/reperfusion injury. J Clin Investig. 2000;105:1363–71. https://doi.org/10.1172/jci8621.
Thurman JM, Ljubanovic D, Edelstein CL, Gilkeson GS, Holers VM. Lack of a functional alternative complement pathway ameliorates ischemic acute renal failure in mice. J Immunol (Baltimore, Md: 1950). 2003;170:1517–23. https://doi.org/10.4049/jimmunol.170.3.1517.
Lien YH, Lai LW, Silva AL. Pathogenesis of renal ischemia/reperfusion injury: lessons from knockout mice. Life Sci. 2003;74:543–52. https://doi.org/10.1016/j.lfs.2003.08.001.
Zheng X, Feng B, Chen G, Zhang X, Li M, Sun H, Liu W, Vladau C, Liu R, Jevnikar AM, Garcia B, Zhong R, Min WP. Preventing renal ischemia-reperfusion injury using small interfering RNA by targeting complement 3 gene. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2006;6:2099–108. https://doi.org/10.1111/j.1600-6143.2006.01427.x.
Thurman JM, Royer PA, Ljubanovic D, Dursun B, Lenderink AM, Edelstein CL, Holers VM. Treatment with an inhibitory monoclonal antibody to mouse factor B protects mice from induction of apoptosis and renal ischemia/reperfusion injury. J Am Soc Nephrol. 2006;17:707–15. https://doi.org/10.1681/asn.2005070698.
Castellano G, Melchiorre R, Loverre A, Ditonno P, Montinaro V, Rossini M, Divella C, Battaglia M, Lucarelli G, Annunziata G, Palazzo S, Selvaggi FP, Staffieri F, Crovace A, Daha MR, Mannesse M, van Wetering S, Paolo Schena F, Grandaliano G. Therapeutic targeting of classical and lectin pathways of complement protects from ischemia-reperfusion-induced renal damage. Am J Pathol. 2010;176:1648–59. https://doi.org/10.2353/ajpath.2010.090276.
Kerr FK, Thomas AR, Wijeyewickrema LC, Whisstock JC, Boyd SE, Kaiserman D, Matthews AY, Bird PI, Thielens NM, Rossi V, Pike RN. Elucidation of the substrate specificity of the MASP-2 protease of the lectin complement pathway and identification of the enzyme as a major physiological target of the serpin, C1-inhibitor. Mol Immunol. 2008;45:670–7. https://doi.org/10.1016/j.molimm.2007.07.008.
Farrar CA, Zhou W, Sacks SH. Role of the lectin complement pathway in kidney transplantation. Immunobiology. 2016;221:1068–72. https://doi.org/10.1016/j.imbio.2016.05.004.
Asgari E, Farrar CA, Lynch N, Ali YM, Roscher S, Stover C, Zhou W, Schwaeble WJ, Sacks SH. Mannan-binding lectin-associated serine protease 2 is critical for the development of renal ischemia reperfusion injury and mediates tissue injury in the absence of complement C4. FASEB J. 2014;28:3996–4003. https://doi.org/10.1096/fj.13-246306.
Berger SP, Daha MR. Emerging role of the mannose-binding lectin-dependent pathway of complement activation in clinical organ transplantation. Curr Opin Organ Transplant. 2011;16:28–33. https://doi.org/10.1097/MOT.0b013e3283425509.
Berger SP, Roos A, Mallat MJ, Fujita T, de Fijter JW, Daha MR. Association between mannose-binding lectin levels and graft survival in kidney transplantation. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2005;5:1361–6. https://doi.org/10.1111/j.1600-6143.2005.00841.x.
Farrar CA, Tran D, Li K, Wu W, Peng Q, Schwaeble W, Zhou W, Sacks SH. Collectin-11 detects stress-induced L-fucose pattern to trigger renal epithelial injury. J Clin Investig. 2016;126:1911–25. https://doi.org/10.1172/jci83000.
Howard MC, Nauser CL, Farrar CA, Wallis R, Sacks SH. l-Fucose prevention of renal ischaemia/reperfusion injury in Mice. FASEB J. 2020;34:822–34. https://doi.org/10.1096/fj.201901582R.
Damman J, Bloks VW, Daha MR, van der Most PJ, Sanjabi B, van der Vlies P, Snieder H, Ploeg RJ, Krikke C, Leuvenink HG, Seelen MA. Hypoxia and complement-and-coagulation pathways in the deceased organ donor as the major target for intervention to improve renal allograft outcome. Transplantation. 2015;99:1293–300. https://doi.org/10.1097/tp.0000000000000500.
Damman J, Seelen MA, Moers C, Daha MR, Rahmel A, Leuvenink HG, Paul A, Pirenne J, Ploeg RJ. Systemic complement activation in deceased donors is associated with acute rejection after renal transplantation in the recipient. Transplantation. 2011;92:163–9. https://doi.org/10.1097/TP.0b013e318222c9a0.
Arias-Cabrales CE, Riera M, Pérez-Sáez MJ, Gimeno J, Benito D, Redondo D, Burballa C, Crespo M, Pascual J, Rodríguez E. Activation of final complement components after kidney transplantation as a marker of delayed graft function severity. Clin Kidney J. 2021;14:1190–6. https://doi.org/10.1093/ckj/sfaa147.
Yu ZX, Qi S, Lasaro MA, Bouchard K, Dow C, Moore K, Wu Z, Barama A, Xu J, Johnson K, Marozsan AJ, Wang Y. Targeting complement pathways during cold ischemia and reperfusion prevents delayed graft function. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2016;16:2589–97. https://doi.org/10.1111/ajt.13797.
Kassimatis T, Greenlaw R, Hunter JP, Douiri A, Flach C, Rebollo-Mesa I, Nichols LL, Qasem A, Danzi G, Olsburgh J, Drage M, Friend PJ, Neri F, Karegli J, Horsfield C, Smith RA, Sacks SH. Ex vivo delivery of Mirococept: A dose-finding study in pig kidney after showing a low dose is insufficient to reduce delayed graft function in human kidney. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2021;21:1012–26. https://doi.org/10.1111/ajt.16265.
Klos A, Tenner AJ, Johswich KO, Ager RR, Reis ES, Köhl J. The role of the anaphylatoxins in health and disease. Mol Immunol. 2009;46:2753–66. https://doi.org/10.1016/j.molimm.2009.04.027.
Peng Q, Li K, Smyth LA, Xing G, Wang N, Meader L, Lu B, Sacks SH, Zhou W. C3a and C5a promote renal ischemia-reperfusion injury. J Am Soc Nephrol. 2012;23:1474–85. https://doi.org/10.1681/asn.2011111072.
Peng Q, Wu W, Wu KY, Cao B, Qiang C, Li K, Sacks SH, Zhou W. The C5a/C5aR1 axis promotes progression of renal tubulointerstitial fibrosis in a mouse model of renal ischemia/reperfusion injury. Kidney Int. 2019;96:117–28. https://doi.org/10.1016/j.kint.2019.01.039.
Poppelaars F, van Werkhoven MB, Kotimaa J, Veldhuis ZJ, Ausema A, Broeren SGM, Damman J, Hempel JC, Leuvenink HGD, Daha MR, van Son WJ, van Kooten C, van Os RP, Hillebrands JL, Seelen MA. Critical role for complement receptor C5aR2 in the pathogenesis of renal ischemia-reperfusion injury. FASEB J. 2017;31:3193–204. https://doi.org/10.1096/fj.201601218R.
Einecke G, Sis B, Reeve J, Mengel M, Campbell PM, Hidalgo LG, Kaplan B, Halloran PF. Antibody-mediated microcirculation injury is the major cause of late kidney transplant failure. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2009;9:2520–31. https://doi.org/10.1111/j.1600-6143.2009.02799.x.
Charreau B. Cellular and molecular crosstalk of graft endothelial cells during AMR: effector functions and mechanisms. Transplantation. 2021;105:e156–67. https://doi.org/10.1097/tp.0000000000003741.
McMullen ME, Hart ML, Walsh MC, Buras J, Takahashi K, Stahl GL. Mannose-binding lectin binds IgM to activate the lectin complement pathway in vitro and in vivo. Immunobiology. 2006;211:759–66. https://doi.org/10.1016/j.imbio.2006.06.011.
Loupy A, Lefaucheur C, Vernerey D, Prugger C, Duong van Huyen JP, Mooney N, Suberbielle C, Frémeaux-Bacchi V, Méjean A, Desgrandchamps F, Anglicheau D, Nochy D, Charron D, Empana JP, Delahousse M, Legendre C, Glotz D, Hill GS, Zeevi A, Jouven X. Complement-binding anti-HLA antibodies and kidney-allograft survival. N Engl J Med. 2013;369:1215–26. https://doi.org/10.1056/NEJMoa1302506.
Al-Aly Z, Reddivari V, Moiz A, Balasubramanian G, Cortese CM, Salinas-Madrigal L, Bastani B. Renal allograft biopsies in the era of C4d staining: the need for change in the Banff classification system. Transplant Int. 2008;21:268–75. https://doi.org/10.1111/j.1432-2277.2007.00604.x.
Chandra P. C4d in native glomerular diseases. Am J Nephrol. 2019;49:81–92. https://doi.org/10.1159/000496059.
Haas M, Sis B, Racusen LC, Solez K, Glotz D, Colvin RB, Castro MC, David DS, David-Neto E, Bagnasco SM, Cendales LC, Cornell LD, Demetris AJ, Drachenberg CB, Farver CF, Farris AB 3rd, Gibson IW, Kraus E, Liapis H, Loupy A, Nickeleit V, Randhawa P, Rodriguez ER, Rush D, Smith RN, Tan CD, Wallace WD, Mengel M. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2014;14:272–83. https://doi.org/10.1111/ajt.12590.
Cumpelik A, Heeger PS. Effects of the complement system on antibody formation and function: implications for transplantation. Curr Opin Organ Transplant. 2022. https://doi.org/10.1097/mot.0000000000001002.
Gonzalez SF, Lukacs-Kornek V, Kuligowski MP, Pitcher LA, Degn SE, Turley SJ, Carroll MC. Complement-dependent transport of antigen into B cell follicles. J Immunol (Baltimore, Md: 1950). 2010;185:2659–64. https://doi.org/10.4049/jimmunol.1000522.
Anania JC, Westin A, Adler J, Heyman B. A novel image analysis approach reveals a role for complement receptors 1 and 2 in follicular dendritic cell organization in germinal centers. Front Immunol. 2021;12: 655753. https://doi.org/10.3389/fimmu.2021.655753.
Schriek P, Ching AC, Moily NS, Moffat J, Beattie L, Steiner TM, Hosking LM, Thurman JM, Holers VM, Ishido S, Lahoud MH, Caminschi I, Heath WR, Mintern JD, Villadangos JA. Marginal zone B cells acquire dendritic cell functions by trogocytosis. Science (New York, NY). 2022;375:eabf7470. https://doi.org/10.1126/science.abf7470.
Cumpelik A, Heja D, Hu Y, Varano G, Ordikhani F, Roberto MP, He Z, Homann D, Lira SA, Dominguez-Sola D, Heeger PS. Dynamic regulation of B cell complement signaling is integral to germinal center responses. Nat Immunol. 2021;22:757–68. https://doi.org/10.1038/s41590-021-00926-0.
Liu L, Fang C, Fu W, Jiang B, Li G, Qin L, Rosenbluth J, Gong G, Xie CB, Yoo P, Tellides G, Pober JS, Jane-Wit D. endothelial cell-derived interleukin-18 released during ischemia reperfusion injury selectively expands T peripheral helper cells to promote alloantibody production. Circulation. 2020;141:464–78. https://doi.org/10.1161/circulationaha.119.042501.
Shivshankar P, Li YD, Mueller-Ortiz SL, Wetsel RA. In response to complement anaphylatoxin peptides C3a and C5a, human vascular endothelial cells migrate and mediate the activation of B-cells and polarization of T-cells. FASEB J. 2020;34:7540–60. https://doi.org/10.1096/fj.201902397R.
Bobka S, Ebert N, Koertvely E, Jacobi J, Wiesener M, Buttner-Herold M, Amann K, Daniel C. Is Early complement activation in renal transplantation associated with later graft outcome? Kidney Blood Press Res. 2018;43:1488–504. https://doi.org/10.1159/000494014.
Lafayette RA, Rovin BH, Reich HN, Tumlin JA, Floege J, Barratt J. Safety, tolerability and efficacy of narsoplimab, a Novel MASP-2 inhibitor for the treatment of IgA nephropathy. Kidney Int Rep. 2020;5:2032–41. https://doi.org/10.1016/j.ekir.2020.08.003.
Aradottir SS, Kristoffersson AC, Roumenina LT, Bjerre A, Kashioulis P, Palsson R, Karpman D. Factor D inhibition blocks complement activation induced by mutant factor B associated with atypical hemolytic uremic syndrome and membranoproliferative glomerulonephritis. Front Immunol. 2021;12: 690821. https://doi.org/10.3389/fimmu.2021.690821.
Kulasekararaj AG, Risitano AM, Maciejewski JP, Notaro R, Browett P, Lee JW, Huang M, Geffner M, Brodsky RA. Phase 2 study of danicopan in patients with paroxysmal nocturnal hemoglobinuria with an inadequate response to eculizumab. Blood. 2021;138:1928–38. https://doi.org/10.1182/blood.2021011388.
Wiles JA, Galvan MD, Podos SD, Geffner M, Huang M. Discovery and development of the oral complement factor D inhibitor danicopan (ACH-4471). Curr Med Chem. 2020;27:4165–80. https://doi.org/10.2174/0929867326666191001130342.
Mainolfi N, Ehara T, Karki RG, Anderson K, Mac Sweeney A, Liao SM, Argikar UA, Jendza K, Zhang C, Powers J, Klosowski DW, Crowley M, Kawanami T, Ding J, April M, Forster C, Serrano-Wu M, Capparelli M, Ramqaj R, Solovay C, Cumin F, Smith TM, Ferrara L, Lee W, Long D, Prentiss M, De Erkenez A, Yang L, Liu F, Sellner H, Sirockin F, Valeur E, Erbel P, Ostermeier D, Ramage P, Gerhartz B, Schubart A, Flohr S, Gradoux N, Feifel R, Vogg B, Wiesmann C, Maibaum J, Eder J, Sedrani R, Harrison RA, Mogi M, Jaffee BD, Adams CM. Discovery of 4-((2S,4S)-4-Ethoxy-1-((5-methoxy-7-methyl-1H-indol-4-yl)methyl)piperidin-2-yl)benzoic Acid (LNP023), a factor B inhibitor specifically designed to be applicable to treating a diverse array of complement mediated diseases. J Med Chem. 2020;63:5697–722. https://doi.org/10.1021/acs.jmedchem.9b01870.
Hasturk H, Hajishengallis G, Lambris JD, Mastellos DC, Yancopoulou D. Phase IIa clinical trial of complement C3 inhibitor AMY-101 in adults with periodontal inflammation. J Clin Investig. 2021. https://doi.org/10.1172/jci152973.
Lamers C, Mastellos DC, Ricklin D, Lambris JD. Compstatins: the dawn of clinical C3-targeted complement inhibition. Trends Pharmacol Sci. 2022;43:629–40. https://doi.org/10.1016/j.tips.2022.01.004.
Azimzadeh A, Zorn GL III, Blair KS, Zhang JP, Pfeiffer S, Harrison RA, Cozzi E, White DJ, Pierson RN III. Hyperacute lung rejection in the pig-to-human model. 2. Synergy between soluble and membrane complement inhibition. Xenotransplantation. 2003;10:120–31. https://doi.org/10.1034/j.1399-3089.2003.01102.x.
Li JS, Sanders SP, Perry AE, Stinnett SS, Jaggers J, Bokesch P, Reynolds L, Nassar R, Anderson PA. Pharmacokinetics and safety of TP10, soluble complement receptor 1, in infants undergoing cardiopulmonary bypass. Am Heart J. 2004;147:173–80. https://doi.org/10.1016/j.ahj.2003.07.004.
Pauly D, Nagel BM, Reinders J, Killian T, Wulf M, Ackermann S, Ehrenstein B, Zipfel PF, Skerka C, Weber BH. A novel antibody against human properdin inhibits the alternative complement system and specifically detects properdin from blood samples. PLoS ONE. 2014;9: e96371. https://doi.org/10.1371/journal.pone.0096371.
Marceau F, Petitclerc E. C5a receptor antagonism coming of age for vascular pathology. Int Immunopharmacol. 2022;110: 109042. https://doi.org/10.1016/j.intimp.2022.109042.
Bauer M, Weyland A, Marx G, Bloos F, Weber S, Weiler N, Kluge S, Diers A, Simon TP, Lautenschläger I, Gründling M, Jaschinski U, Simon P, Nierhaus A, Moerer O, Reill L, Jörres A, Guo R, Loeffler M, Reinhart K, Riedemann N. Efficacy and safety of vilobelimab (IFX-1), a novel monoclonal anti-C5a antibody, in patients with early severe sepsis or septic shock—a randomized, placebo-controlled, double-blind, multicenter, phase IIa trial (SCIENS Study). Crit Care Explor. 2021;3: e0577. https://doi.org/10.1097/cce.0000000000000577.
Mastellos DC, Ricklin D, Lambris JD. Clinical promise of next-generation complement therapeutics. Nat Rev Drug Discov. 2019;18:707–29. https://doi.org/10.1038/s41573-019-0031-6.
Schroppel B, Akalin E, Baweja M, Bloom RD, Florman S, Goldstein M, Haydel B, Hricik DE, Kulkarni S, Levine M, Mehrotra A, Patel A, Poggio ED, Ratner L, Shapiro R, Heeger PS. Peritransplant eculizumab does not prevent delayed graft function in deceased donor kidney transplant recipients: results of two randomized controlled pilot trials. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2019. https://doi.org/10.1111/ajt.15580.
Kaabak M, Babenko N, Shapiro R, Zokoyev A, Dymova O, Kim E. A prospective randomized, controlled trial of eculizumab to prevent ischemia-reperfusion injury in pediatric kidney transplantation. Pediatric Transplant. 2018. https://doi.org/10.1111/petr.13129.
Jordan SC, Choi J, Aubert O, Haas M, Loupy A, Huang E, Peng A, Kim I, Louie S, Ammerman N, Najjar R, Puliyanda D, Vo A. A phase I/II, double-blind, placebo-controlled study assessing safety and efficacy of C1 esterase inhibitor for prevention of delayed graft function in deceased donor kidney transplant recipients. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2018;18:2955–64. https://doi.org/10.1111/ajt.14767.
Huang E, Vo A, Choi J, Ammerman N, Lim K, Sethi S, Kim I, Kumar S, Najjar R, Peng A, Jordan SC. Three-year outcomes of a randomized, double-blind, placebo-controlled study assessing safety and efficacy of C1 esterase inhibitor for prevention of delayed graft function in deceased donor kidney transplant recipients. Clin J Am Soc Nephrol CJASN. 2020;15:109–16. https://doi.org/10.2215/cjn.04840419.
Kassimatis T, Qasem A, Douiri A, Ryan EG, Rebollo-Mesa I, Nichols LL, Greenlaw R, Olsburgh J, Smith RA, Sacks SH, Drage M. A double-blind randomised controlled investigation into the efficacy of Mirococept (APT070) for preventing ischaemia reperfusion injury in the kidney allograft (EMPIRIKAL): study protocol for a randomised controlled trial. Trials. 2017;18:255. https://doi.org/10.1186/s13063-017-1972-x.
Stegall MD, Diwan T, Raghavaiah S, Cornell LD, Burns J, Dean PG, Cosio FG, Gandhi MJ, Kremers W, Gloor JM. Terminal complement inhibition decreases antibody-mediated rejection in sensitized renal transplant recipients. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2011;11:2405–13. https://doi.org/10.1111/j.1600-6143.2011.03757.x.
Marks WH, Mamode N, Montgomery RA, Stegall MD, Ratner LE, Cornell LD, Rowshani AT, Colvin RB, Dain B, Boice JA, Glotz D. Safety and efficacy of eculizumab in the prevention of antibody-mediated rejection in living-donor kidney transplant recipients requiring desensitization therapy: a randomized trial. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2019;19:2876–88. https://doi.org/10.1111/ajt.15364.
Glotz D, Russ G, Rostaing L, Legendre C, Tufveson G, Chadban S, Grinyo J, Mamode N, Rigotti P, Couzi L, Buchler M, Sandrini S, Dain B, Garfield M, Ogawa M, Richard T, Marks WH. Safety and efficacy of eculizumab for the prevention of antibody-mediated rejection after deceased-donor kidney transplantation in patients with preformed donor-specific antibodies. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2019;19:2865–75. https://doi.org/10.1111/ajt.15397.
Tan EK, Bentall A, Dean PG, Shaheen MF, Stegall MD, Schinstock CA. Use of eculizumab for active antibody-mediated rejection that occurs early post-kidney transplantation: a consecutive series of 15 cases. Transplantation. 2019;103:2397–404. https://doi.org/10.1097/tp.0000000000002639.
Kulkarni S, Kirkiles-Smith NC, Deng YH, Formica RN, Moeckel G, Broecker V, Bow L, Tomlin R, Pober JS. Eculizumab therapy for chronic antibody-mediated injury in kidney transplant recipients: a pilot randomized controlled trial. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2017;17:682–91. https://doi.org/10.1111/ajt.14001.
Vo AA, Zeevi A, Choi J, Cisneros K, Toyoda M, Kahwaji J, Peng A, Villicana R, Puliyanda D, Reinsmoen N, Haas M, Jordan SC. A phase I/II placebo-controlled trial of C1-inhibitor for prevention of antibody-mediated rejection in HLA sensitized patients. Transplantation. 2015;99:299–308. https://doi.org/10.1097/tp.0000000000000592.
Montgomery RA, Orandi BJ, Racusen L, Jackson AM, Garonzik-Wang JM, Shah T, Woodle ES, Sommerer C, Fitts D, Rockich K, Zhang P, Uknis ME. Plasma-derived C1 esterase inhibitor for acute antibody-mediated rejection following kidney transplantation: results of a randomized double-blind placebo-controlled pilot study. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2016;16:3468–78. https://doi.org/10.1111/ajt.13871.
Eskandary F, Jilma B, Mühlbacher J, Wahrmann M, Regele H, Kozakowski N, Firbas C, Panicker S, Parry GC, Gilbert JC, Halloran PF, Böhmig GA. Anti-C1s monoclonal antibody BIVV009 in late antibody-mediated kidney allograft rejection-results from a first-in-patient phase 1 trial. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2018;18:916–26. https://doi.org/10.1111/ajt.14528.
Struijk GH, Bouts AH, Rijkers GT, Kuin EA, ten Berge IJ, Bemelman FJ. Meningococcal sepsis complicating eculizumab treatment despite prior vaccination. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2013;13:819–20. https://doi.org/10.1111/ajt.12032.
Ma Y, Liu Y, Zhang Z, Yang GY. Significance of complement system in ischemic stroke: a comprehensive review. Aging Dis. 2019;10:429–62. https://doi.org/10.14336/ad.2019.0119.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation), project number 387509280, SFB 1350 TP C2.
Conflict of interest
Maike Büttner-Herold reports lecture fees from Alexion Pharma Germany GmbH, Novartis Pharma GmbH, Sanofi and Pfizer Pharma GmbH and Kerstin Amann reports consulting fees from Novartis Pharma GmbH and Otsuka Pharmaceutical Co. and lecture fees from Alexion Pharma Germany GmbH, Shire Pharmaceuticals, AstraZeneca, Novartis Pharma GmbH. All other authors report no potential conflicts of interest or financial disclosures that are pertinent to the article.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data availability statement
This article is a review article that does not include original data.
Code availability
Not applicable.
Author contributions
EV and CD provided general information on the complement system, clinical interpretation, created figures and drafted and reviewed all versions of the manuscript. MB-H created figures and drafted and reviewed all versions of the manuscript. KA drafted and reviewed all versions of the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Vonbrunn, E., Büttner-Herold, M., Amann, K. et al. Complement Inhibition in Kidney Transplantation: Where Are We Now?. BioDrugs 37, 5–19 (2023). https://doi.org/10.1007/s40259-022-00567-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40259-022-00567-1