Abstract
Objective
This research assesses the cost effectiveness of Axicabtagene ciloleucel (Axi-cel), Tisagenlecleucel (Tis-cel), Relmacabtagene autoleucel (Rel-cel) and Lisocabtagene maraleucel (Lis-cel) against standard of care (SOC) for patients with diffuse large B-cell lymphoma (DLBCL) in the first-line setting (1L), second-line setting (2L) and third-line or later setting (3L+).
Methods
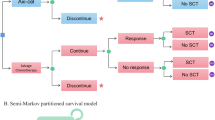
Markov modelling based on a flexible survival model was adopted to evaluate four chimeric antigen receptor T-cell (CAR-T) therapies compared with SOC for patients with diffuse large B-cell lymphoma (DLBCL). The clinical inputs and utility values of the model were derived from the most recent clinical trials and the health care costs from a Chinese provincial clinical center. Costs and quality-adjusted life years (QALYs) were used to derive incremental cost-effectiveness ratios (ICERs) from the Chinese health care system perspective.
Results
The ICER of Axi-cel (1L) versus SOC was approximately Chinese Yuan (CNY) 2,125,311 per QALY. The ICER for Axi-cel (2L), Tis-cel (2L) and Liso-cel (2L)) versus SOC in transplant-eligible patients were approximately CNY363,977, CNY32,066,781 and CNY347,746 per quality-adjusted life year (QALY), respectively. The ICER for Liso-cel (2L) versus SOC in transplant-ineligible patients was approximately CNY1,233,972 per QALY. The ICERs for Axi-cel (3L+), Tis-cel (3L+), Rel-cel (3L+) and Liso-cel (3L+) versus SOC were approximately CNY346,009, CNY654,344, CNY280,964 and CNY436,858 per QALY, respectively. In the scenario analysis using mixture cure models, the long-term survival benefit for CAR-T and SOC groups was found higher, and only Rel-cel (3L+) was found to be cost effective.
Conclusion
Our results demonstrated that CAR-T treatments are not cost effective in any-line settings for DLBCL patients at the WHO-recommended willingness-to-pay threshold (CNY257,241 per QALY) in the base-case analysis. Price reduction of CAR-T therapies is the main approach for lowering ICERs and ensuring that the drug costs are proportional to patient health benefits.

Similar content being viewed by others
References
Sehn LH, Donaldson J, Chhanabhai M, et al. Introduction of combined CHOP plus rituximab therapy dramatically improved outcome of diffuse large B-cell lymphoma in British Columbia. J Clin Oncol. 2005;23(22):5027–33. https://doi.org/10.1200/JCO.2005.09.137.
Liu W, Liu J, Song Y, et al. Burden of lymphoma in China, 1990–2019: an analysis of the global burden of diseases, injuries, and risk factors study 2019. Aging (Albany NY). 2022;14(7):3175–90. https://doi.org/10.18632/aging.204006.
Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346(4):235–42. https://doi.org/10.1056/NEJMoa011795.
Pfreundschuh M, Trümper L, Osterborg A, et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomized controlled trial by the MabThera International Trial (MinT) Group. Lancet Oncol. 2006;7(5):379–91. https://doi.org/10.1016/S1470-2045(06)70664-7.
Crump M, Neelapu SS, Farooq U, et al. Outcomes in refractory diffuse large B-cell lymphoma: results from the international SCHOLAR-1 study [published correction appears in Blood. 2018 Feb 1;131(5):587–588]. Blood. 2017;130(16):1800–8. https://doi.org/10.1182/blood-2017-03-769620.
Locke FL, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel as second-line therapy for large B-cell lymphoma. N Engl J Med. 2022;386(7):640–54. https://doi.org/10.1056/NEJMoa2116133.
Kamdar M, Solomon SR, Arnason J, et al. Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomized, phase 3 trial [published correction appears in Lancet. 2022 Jul 16;400(10347):160]. Lancet. 2022;399(10343):2294–308. https://doi.org/10.1016/S0140-6736(22)00662-6.
Bishop MR, Dickinson M, Purtill D, et al. Second-line tisagenlecleucel or standard care in aggressive B-cell lymphoma. N Engl J Med. 2022;386(7):629–39. https://doi.org/10.1056/NEJMoa211659.
IBM Micromedex Red Book. 2021. www.micromedex.com.
Lin JK, Muffly LS, Spinner MA, Barnes JI, Owens DK, Goldhaber-Fiebert JD. Cost Effectiveness of chimeric antigen receptor T-cell therapy in multiply relapsed or refractory adult large B-cell lymphoma. J Clin Oncol. 2019;37(24):2105–19. https://doi.org/10.1200/JCO.18.02079.
Furzer J, Gupta S, Nathan PC, et al. Cost-effectiveness of tisagenlecleucel vs standard care in high-risk relapsed pediatric acute lymphoblastic leukemia in Canada. JAMA Oncol. 2020;6(3):393–401. https://doi.org/10.1001/jamaoncol.2019.5909.
Wakase S, Teshima T, Zhang J, et al. Cost effectiveness analysis of tisagenlecleucel for the treatment of adult patients with relapsed or refractory diffuse large B cell lymphoma in Japan. Transplant Cell Ther. 2021;27(6):506.e1-506.e10. https://doi.org/10.1016/j.jtct.2021.03.005.
Cher BP, Gan KY, Aziz MIA, et al. Cost utility analysis of tisagenlecleucel vs salvage chemotherapy in the treatment of relapsed/refractory diffuse large B-cell lymphoma from Singapore’s healthcare system perspective. J Med Econ. 2020;23(11):1321–9. https://doi.org/10.1080/13696998.2020.1808981.
Choe JH, Abdel-Azim H, Padula WV, Abou-El-Enein M. Cost-effectiveness of axicabtagene ciloleucel and tisagenlecleucel as second-line or later therapy in relapsed or refractory diffuse large B-cell lymphoma. JAMA Netw Open. 2022;5(12): e2245956. https://doi.org/10.1001/jamanetworkopen.2022.45956.
Hu Y, Feng J, Gu T, et al. CAR T-cell therapies in China: rapid evolution and a bright future. Lancet Haematol. 2022;9(12):e930–41. https://doi.org/10.1016/S2352-3026(22)00291-5.
Neelapu SS, Dickinson M, Munoz J, et al. Axicabtagene ciloleucel as first-line therapy in high-risk large B-cell lymphoma: the phase 2 ZUMA-12 trial. Nat Med. 2022;28(4):735–42. https://doi.org/10.1038/s41591-022-01731-4.
Locke FL, Ghobadi A, Jacobson CA, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicenter, phase 1–2 trial. Lancet Oncol. 2019;20(1):31–42. https://doi.org/10.1016/S1470-2045(18)30864-7.
Sehgal A, Hoda D, Riedell PA, et al. Lisocabtagene maraleucel as second-line therapy in adults with relapsed or refractory large B-cell lymphoma who were not intended for haematopoietic stem cell transplantation (PILOT): an open-label, phase 2 study. Lancet Oncol. 2022;23(8):1066–77. https://doi.org/10.1016/S1470-2045(22)00339-4.
Abramson JS, Palomba ML, Gordon LI, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396(10254):839–52. https://doi.org/10.1016/S0140-6736(20)31366-0.
Ying Z, Yang H, Guo Y, et al. Relmacabtagene autoleucel (relma-cel) CD19 CAR-T therapy for adults with heavily pretreated relapsed/refractory large B-cell lymphoma in China. Cancer Med. 2021;10(3):999–1011. https://doi.org/10.1002/cam4.3686.
Schuster SJ, Tam CS, Borchmann P, et al. Long-term clinical outcomes of tisagenlecleucel in patients with relapsed or refractory aggressive B-cell lymphomas (JULIET): a multicentre, open-label, single-arm, phase 2 study. Lancet Oncol. 2021;22(10):1403–15. https://doi.org/10.1016/S1470-2045(21)00375-2.
Dunleavy K, Fanale MA, Abramson JS, et al. Dose-adjusted EPOCH-R (etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab) in untreated aggressive diffuse large B-cell lymphoma with MYC rearrangement: a prospective, multicentre, single-arm phase 2 study. Lancet Haematol. 2018;5(12):e609–17. https://doi.org/10.1016/S2352-3026(18)30177-7.
Byrd RH, Lu P, Nocedal J, et al. A limited memory algorithm for bound constrained optimization. SIAM J Sci Comput. 1995;16:1190–208.
Zhu J, et al. “Chinese Society of Clinical Oncology (CSCO) diagnosis and treatment guidelines for malignant lymphoma 2021 (English version).” Chin J Cancer Res. 2021;33(3):289–301. https://doi.org/10.21147/j.issn.1000-9604.2021.03.01.
Zhejiang Provincial Center for Drug & Medical Device Procurement. https://www.zjyxcg.cn/?pageNow=88.
Maziarz RT, Waller EK, Jaeger U, et al. Patient-reported long-term quality of life after tisagenlecleucel in relapsed/refractory diffuse large B-cell lymphoma. Blood Adv. 2020;4(4):629–37. https://doi.org/10.1182/bloodadvances.2019001026.
Yang Z, Busschbach J, Liu G, Luo N. EQ-5D-5L norms for the urban Chinese population in China. Health Qual Life Outcomes. 2018;16(1):210. https://doi.org/10.1186/s12955-018-1036-2.
Li Z, Pan Z, Zhang L, et al. End-of-life cost and its determinants for cancer patients in urban China: a population-based retrospective study. BMJ Open. 2019;9(3): e026309.
Vijenthira A, Kuruvilla J, Crump M, Jain M, Prica A. Cost-effectiveness analysis of frontline polatuzumab-rituximab, cyclophosphamide, doxorubicin, and prednisone and/or second-line chimeric antigen receptor T-cell therapy versus standard of care for treatment of patients with intermediate- to high-risk diffuse large B-cell lymphoma. J Clin Oncol. 2023;41(8):1577–89. https://doi.org/10.1200/JCO.22.00478.
Moradi-Lakeh M, Yaghoubi M, Seitz P, Javanbakht M, Brock E. Cost-effectiveness of tisagenlecleucel in paediatric acute lymphoblastic leukaemia (pALL) and adult diffuse large B-cell lymphoma (DLBCL) in Switzerland. Adv Ther. 2021;38(6):3427–43. https://doi.org/10.1007/s12325-021-01767-x.
Wang N, Yang Y, Xu L, Mao Z, Cui D. Influence of Chinese National Centralized Drug Procurement on the price of policy-related drugs: an interrupted time series analysis. BMC Public Health. 2021;21(1):1883.
Cai L, Tao T, Li H, Zhang Z, Zhang L, Li X. Impact of the national drug price negotiation policy on the utilization, cost, and accessibility of anticancer medicines in China: a controlled interrupted time series study. J Glob Health. 2022;12:11016. https://doi.org/10.7189/jogh.12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study is funded by China Scholarship Council (CSC, 2022062320370) and Zhejiang Pharmaceutical Association Economics and Health Technology Evaluation Special Scientific Research Funding Project (2022-SKY-A07054-0007).
Data availability
The data presented in this study are available on request from the corresponding author.
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
This research was approved by the institutional review board of the First Affiliated Hospital, School of Medicine, Zhejiang University and performed according to the Declaration of Helsinki.
Author contributions
Wu, and Zhou developed the economic model, performed the analyses, collected and reviewed the data. Wang, Zhao, and Yang interpreted the results and drafted the manuscript. All authors reviewed the data, interpreted the result and revised the manuscript. Comans and Dong supervised the whole study.
Consent to participate
Not applicable.
Consent for publication from patients/participants
Not applicable.
Code availability
Available upon request.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, W., Zhou, Y., Wang, Y. et al. Value for Money of CAR-T Cell Therapy for Patients with Diffuse Large B-cell Lymphoma in China: Evidence from a Cost-Effectiveness Analysis. Appl Health Econ Health Policy 21, 773–783 (2023). https://doi.org/10.1007/s40258-023-00817-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40258-023-00817-5