Abstract
Hypertrophic cardiomyopathy (HCM) is a chronic, progressive disease of the cardiomyocyte with a diverse and heterogeneous clinical presentation and course. This diversity and heterogeneity have added to the complexity of modeling the pathophysiological pathways that contribute to the disease burden. The development of novel therapeutic approaches targeting precise mechanisms within the underlying biology of HCM provides a tool to model and test these pathways. Here, we integrate the results of clinical observations with mavacamten, an allosteric, selective, and reversible inhibitor of cardiac myosin, the motor unit of the sarcomere, to develop an integrated pathophysiological pathway model of HCM, confirming the key role of excess sarcomeric activity. This model may serve as a foundation to understand the role of HCM pathophysiological pathways in the clinical presentation of the disease, and how a targeted therapeutic intervention capable of normalizing sarcomeric activity and repopulating low-energy utilization states may reduce the impact of these pathways in HCM and potentially related disease states.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The pathophysiology of hypertrophic cardiomyopathy (HCM) is complex and not yet fully understood. |
Mavacamten is a first-in-class, selective, cardiac myosin inhibitor that improved function and symptoms in patients with HCM. |
We used established biological pathways and the results of clinical observations with mavacamten to build an integrated pathophysiological pathway model of HCM. |
1 Introduction
1.1 Genetics
Hypertrophic cardiomyopathy (HCM) is a chronic, progressive disease of the cardiomyocyte that leads to increased ventricular wall thickness, with a diverse and heterogeneous clinical presentation and course. Although the pathophysiology of HCM is complex and multifactorial [1], HCM is now generally recognized as a disease caused by dysfunction of the sarcomere, the basic contractile apparatus of the heart muscle. Indeed, approximately 40% of affected individuals, and a significant portion of those with a family history of clinical disease, have at least one mutation in one or more genes that encode sarcomere proteins [2,3,4,5]. Moreover, systolic hyperactivity and diastolic dysfunction in HCM have been reported regardless of etiology [6, 7], and often herald the clinical onset of the disease [8]. In most cases, myofibrillar dysfunction resulting from excess sarcomeric myosin–actin cross-bridge formation during systole and, more importantly, diastole, leads to hypertrophy, hypercontractility, and impaired relaxation as well as to myocardial inefficiency and unproductive energy consumption [9].
1.2 Pathophysiology
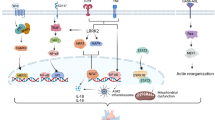
In the healthy heart, 40–50% of the myosin heads are in the ‘off state’ with negligible energy consumption, whereas in HCM there is a shift in this ratio, with only 15–20% of the myosin heads being in this ‘off state’ [10,11,12]. The extra myosin heads in the ‘on state’ not only consume more adenosine triphosphate (ATP) (i.e., energy), but are also primed to interact with actin. These effects combine to cause excess myosin–actin cross-bridges during both systole and diastole, leading to inefficient hyperdynamic contraction and diastolic dysfunction [13]. This sustained sarcomeric hyperactivity activates pro-hypertrophic, pro-inflammatory, and pro-fibrotic pathways, resulting in progressive myocardial remodeling, characterized by fibrosis, myofilament disarray, and elevated stresses [13,14,15].
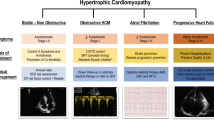
In a subset of patients with HCM, these functional derangements and their associated altered intraventricular hemodynamics can combine with septal hypertrophy, and/or morphological abnormalities of the mitral valvular apparatus, favoring anteriorly displacing forces which may lead to systolic anterior motion (SAM) of the mitral valve [16]. This combination triggers an obstruction to blood flow, creating a pressure gradient through the left ventricular outflow tract (LVOT), termed obstructive HCM (oHCM) [17]. In other cases, obstruction is due to dynamic narrowing within the left ventricular (LV) chamber driven by hypertrophy.
1.3 Current Management
Because the LVOT gradient is dynamic, reflecting increases in obstructing/anteriorly displacing forces that can occur with changes in heart rate, contractile force, and/or loading conditions, patients with oHCM are currently prescribed negative chronotropes and inotropes (non-vasodilating beta-blockers, non-dihydropyridine calcium channel blockers, and/or disopyramide). In cases refractory to pharmacological treatment, invasive therapies such as alcohol septal ablation or myectomy can relieve the structural obstruction. Conversely, in non-obstructive HCM (nHCM), the thickened heart muscle does not block blood outflow from the left ventricle, but does result in impaired relaxation and diastolic dysfunction, which is thought to play an important role in nHCM pathophysiology. Interestingly, impaired relaxation, associated with LV hypertrophy, is also observed in heart failure with preserved ejection fraction.
Responses to mavacamten, a first-in-class, precision intervention that directly targets the cardiac sarcomere and restores its organization and function to a natural state, allow us to confirm the role of the sarcomere and to refine our understanding of the pathology of HCM. Both preclinical (in vitro and in vivo) and clinical data begin to uncover the potential inter-relationship between HCM’s impacts on systolic and diastolic function and, potentially, extend a framework for studying mechanistically related disease states.
1.4 Mavacamten: A Cardiac Myosin Inhibitor
Mavacamten is an allosteric, selective, and reversible inhibitor of cardiac myosin, the motor unit of the sarcomere [18]. Mavacamten decreases the number of myosin heads that can enter the on-actin (power-generating) state, thus reducing the probability of cross-bridge formation in HCM, and shifts the overall myosin population towards the energy-sparing ‘off state’ (see the Electronic Supplementary Material, Fig. S1) [12]. Consequently, and as observed in preclinical studies, mavacamten decreases overall ATP turnover (utilization) at the sarcomere level, reduces diastolic tensions, and promotes relaxation. As a result of these direct, salutary diastolic, systolic, and energy-sparing attributes, mavacamten increases the ventricular chamber size and reduces the velocity of myocardial contraction. This reduces anteriorly displacing forces that favor SAM and, therefore, creates an optimal intraventricular mechanical environment to reduce LVOT obstruction [12, 18, 19].
Because mavacamten has been studied in patients with oHCM and in patients with nHCM, we have aimed to map the related findings to pathophysiological pathways, examining the effects of pharmacological therapy that may be directly related to the impact of HCM on systolic and diastolic function [20,21,22]. In compiling the pathways, we aimed to describe a potential interdependent systolic and diastolic pathophysiology of HCM, and the resultant potential impact of cardiac myosin-directed intervention.
2 Data Source
2.1 Studies
Data from five phase 2/3 clinical trials of mavacamten were examined; these studies were as follows: PIONEER-HCM (NCT02842242) [20]; PIONEER-open-label extension (OLE) ( NCT03496168; data cut-off June 4, 2020) [23]; MAVERICK-HCM (NCT03442764) [21]; EXPLORER-HCM (NCT03470545) [22] and its cardiac magnetic resonance (CMR) sub-study [24]; and MAVA-long-term extension (LTE) (NCT03723655; data cut-off October 30, 2020) [25]. Most patients enrolled in these studies were receiving background HCM treatments (beta-blockers and/or non-dihydropyridine calcium channel blockers). The designs and study populations are summarized in Table 1. Briefly, MAVA-LTE is an ongoing, extension study of mavacamten in patients who completed either EXPLORER-HCM or MAVERICK-HCM. Site personnel and patients remained blinded to treatment assignment and dosing in the respective parent studies.
2.2 Study Population
Overall, 331 unique patients were enrolled in the five clinical studies; the majority of patients participated in one of the extension studies. Each parent study included a post-treatment washout period of 4 weeks (PIONEER-HCM) or 8 weeks (MAVERICK-HCM and EXPLORER-HCM), and subsequent re-screening and enrolment into the respective extension studies. Eligible patients were 18 years old or older (and < 70 years for the PIONEER-HCM study only). All patients must have received a diagnosis of HCM (hypertrophied and non-dilated left ventricle in the absence of systemic or other known cause) with a maximum LV wall thickness of 15 mm or more at screening, or 13 mm or more with a positive family history of HCM. Patients in the MAVERICK-HCM study were required to have LVOT gradients under 30 mmHg at rest, during Valsalva maneuver, and post-exercise with no interventricular gradient (obstruction below the level of the LVOT [e.g., mid-cavity]). Eligible patients in the PIONEER-HCM study had a resting LVOT gradient of 30 mmHg or higher and a post-exercise LVOT gradient of 50 mmHg or higher, and these patients were eligible to enroll in the PIONEER-OLE study. Patients in the EXPLORER-HCM study had a Valsalva gradient of 30 mmHg or higher and at least one peak LVOT gradient of 50 mmHg or higher at rest, after Valsalva maneuver, or post-exercise. LV ejection fraction (LVEF) must have been 55% or more at baseline in all patients. The full eligibility criteria have been described previously [20,21,22]. Upon completion of the MAVERICK-HCM and EXPLORER-HCM studies, patients were eligible to enroll in the MAVA-LTE study.
2.3 Treatments
In total, 163 unique patients received mavacamten (PIONEER-HCM, n = 21; PIONEER-OLE, n = 13; MAVERICK-HCM, n = 40; EXPLORER-HCM, n = 123) and 147 received placebo (MAVERICK-HCM, n = 19; EXPLORER-HCM, n = 128). In addition, the 267 patients who enrolled in MAVA-LTE (data cut-off October 30, 2020) all received mavacamten. This includes 224 patients from EXPLORER-HCM (112 of whom previously received placebo) and 43 patients from MAVERICK-HCM (15 of whom previously received placebo). A summary of the dosage regimen used in each study is presented in the electronic supplementary material (Supplementary Table S1).
2.4 Outcomes
The outcomes of interest for the present article were as follows: LVOT gradient (for oHCM); maximum LV wall thickness; early diastolic mitral annular velocity (e´) and the ratio between early mitral inflow velocity (E) and e´; minute ventilation to carbon dioxide production slope (VE/VCO2); left atrial (LA) volume index (LAVI); N-terminal pro B-type natriuretic peptide (NT-proBNP) level; and cardiac troponin I (cTnI) level (Table 1).
3 Observations/Findings
3.1 Sarcomeric Hyperactivity and LVOT Obstruction
In oHCM, the asymmetrically hypertrophied septum can lead to a dynamic systolic obstruction, commonly at the LVOT, that requires increased cardiac work to maintain cardiac output (Fig. 1A). Because the dynamic obstruction is the result of HCM-mediated hypertrophy, sarcomeric hyperactivity, and hypercontractility, the need to increase work (contractile force) to sustain cardiac output can result in a maladaptive positive feedback loop that may further increase sarcomeric overdrive and the obstruction (Fig. 1A) [26, 27]. Therefore, the reduction in sarcomeric hyperactivity and hypercontractility observed with mavacamten, measured by a normalization in LVEF, would be expected to lead to a decrease in peak LVOT gradients.
A The relationship between hypercontractility of the heart, LVOT obstruction, and cardiac work. B The effect of mavacamten on post-exercise LVOT peak gradient (EXPLORER-HCM). Data from Olivotto et al. Lancet 2020;396:759–69. CI confidence interval, HCM hypertrophic cardiomyopathy, LVOT left ventricular outflow tract, LS least-squares, SAM septal anterior motion, SD standard deviation
Indeed, mavacamten lowered the LVEF in patients with HCM, bringing it within the normal range in PIONEER, MAVERICK-HCM, EXPLORER-HCM, and MAVA-LTE (see the electronic supplementary material, Supplementary Table S2). Additionally, mavacamten was shown to decrease peak LVOT gradients in patients with oHCM [20, 22]. In the EXPLORER-HCM study, the mean (standard deviation) changes in post-exercise and Valsalva LVOT peak gradient from baseline to week 30 were − 47 (40) mmHg and – 49 (34) mmHg, respectively, in the mavacamten group and − 10 (30) mmHg and − 12 (31) mmHg, respectively, in the placebo group (Fig. 1B) [22]. These findings were confirmed in the MAVA-LTE study at week 48. In addition, in EXPLORER-HCM, more patients receiving mavacamten (80.9%; n = 76/94) versus placebo (34.0%; n = 33/97) showed complete resolution of mitral valve SAM after 30 weeks (difference, 46.8%; p < 0.0001) [28]. The changes in LVOT peak gradients from baseline to end of treatment observed in the PIONEER-HCM, PIONEER-OLE, EXPLORER-HCM, and MAVA-LTE studies are presented in the electronic supplementary material (Supplementary Table S2). Notably and consistent with the normalization of sarcomeric activity, overall contractile state, and loading conditions, patients treated with mavacamten displayed greater improvements in peak oxygen consumption (pVO2), a multidimensional index dependent on cardiac output, than those receiving placebo [22].
3.2 Sarcomeric Hyperactivity and LV Hypertrophy
The sarcomeric hyperactivity that occurs in HCM and that leads to hypercontractility of the heart has also been linked to an increase in pro-hypertrophic signaling [29], which in turn results in LV hypertrophy (Fig. 2A). Maximum LV wall thickness is a commonly accepted indicator of LV hypertrophy.
A The relationship between hypercontractility of the heart, LV hypertrophy, and elevated wall stress. B NT-proBNP plasma levels (EXPLORER-HCM). (C) The effect of mavacamten on maximal LV wall thickness (EXPLORER-HCM CMR sub-study). Reduction in NT-proBNP level after mavacamten treatment was 80% greater than for placebo (proportion of geometric mean ratio between the two groups, 0.202; 95% CI 0.169–0.241). The LV wall thickness after reduction following mavacamten treatment was still in the hypertrophic range. Data from Olivotto et al. Lancet 2020;396:759–69 and from Saberi et al. Circulation 2020;142:e470–500, abstract 18654. CI confidence interval, CMR cardiac magnetic resonance, HCM hypertrophic cardiomyopathy, LV left ventricular, NT-proBNP N-terminal pro B-type natriuretic peptide, SD standard deviation
NT-proBNP, a biomarker of pro-hypertrophic signaling, is an inactive, stable peptide synthesized primarily in the left ventricle in response to increased wall tension, and plasma levels of NT-proBNP have been positively correlated with cardiac filling pressure [30, 31]. Owing to the mechanism of action of mavacamten and based on the relationships described above, a reduction in hypercontractility following treatment with mavacamten, as measured by change in LVEF, would be expected to lead to a decrease both in pro-hypertrophic signaling and in LV hypertrophy, as measured by plasma levels of NT-proBNP and maximal LV wall thickness, respectively.
Mavacamten treatment significantly reduced plasma NT-proBNP levels from baseline to end of treatment in both the oHCM and the nHCM studies, including PIONEER-HCM, PIONEER-OLE, MAVERICK-HCM, EXPLORER-HCM, and MAVA-LTE (Fig. 2B; Electronic Supplementary Material, Supplementary Table S2), and reductions in the cardiac gene expression of natriuretic peptide precursors following mavacamten treatment were observed in a mouse HCM model [19].
In the EXPLORER-HCM CMR sub-study, mavacamten was associated with a greater reduction in maximal LV wall thickness measured by CMR than placebo (Fig. 2C; Supplementary Table S2), although the LV wall thickness remained in the hypertrophic range [24, 32]. It should be noted that only a small number of patients were enrolled in the CMR sub-study, and that although the change in LV wall thickness associated with mavacamten was statistically significant, it was small and should therefore be interpreted with caution. However, a reduction in LV wall thickness with mavacamten treatment was also observed in a model of HCM in heterozygous mice harboring a human mutation in the myosin heavy chain [19].
3.3 LV Hypertrophy and Relaxation
LV diastolic dysfunction is another hallmark of HCM that can be characterized by impaired relaxation [33,34,35]. LV hypertrophy has been shown to be a major contributing factor to impaired relaxation (Fig. 3A) [36,37,38,39]. Avegliano et al. reported an association between LV hypertrophy and the degree of diastolic dysfunction using tissue Doppler echocardiography methods [40]. Moreover, Wigle et al. showed a direct correlation between LV end-diastolic pressure and the extent of LV hypertrophy, and Chacko et al. reported a correlation between maximum LV wall thickness and diastolic function using CMR [41, 42]. Finally, Finocchiaro et al. also demonstrated a positive correlation between diastolic dysfunction and the degree of hypertrophy [43]. In addition, there is evidence that LV hypertrophy is associated with diastolic dysfunction and impaired relaxation (Fig. 3A) [44, 45]. However, it should be noted that other factors such as abnormal calcium handling, structural remodeling, fibrosis, and/or inflammation can alter ventricular relaxation and filling.
A The relationship between elevated wall stress, LV hypertrophy, and impaired relaxation. B The effect of mavacamten on septal e´ (MAVERICK-HCM). Data from Ho et al. J Am Coll Cardiol 2020;75:2649–60. e´ early diastolic mitral annular velocity, HCM hypertrophic cardiomyopathy, LV left ventricular, SD standard deviation
Reduced and delayed e’ velocity is associated with impaired myocardial relaxation, while the E/e´ ratio measured by echocardiography is a commonly used surrogate of LV filling pressure, and both are central to the evaluation of diastolic function [46,47,48]. As such, an elevated E/e´ ratio is a good indicator of impaired relaxation of the heart, with healthy individuals presenting a ratio of less than 8, and a ratio over 14 suggesting elevated filling pressure and hindered relaxation [49]. Interestingly, Badran et al. demonstrated that the E/e´ ratio was a powerful independent predictor of cardiac events in patients with HCM [50]. Overall, the ability of mavacamten to increase e´ (Fig. 3B) and to reduce the E/e´ ratio provides further evidence to support the interrelationship between LV hypertrophy on one hand and impaired relaxation of the heart on the other (see the Electronic Supplementary Material, Supplementary Table S2) [21, 22].
3.4 Impaired Relaxation, Increased Preload and LA Volume, and Risk of Atrial Fibrillation and Elevated Pulmonary Pressure
Impaired relaxation increases ventricular filling pressure, as seen by the elevated measures of E/e´ described above and, therefore, diastolic stresses. Moreover, Appleton et al. have reported an association between increased LV filling pressure and LA enlargement in patients with cardiac disease [51]. It is well established that increased LAVI is associated with an increased risk of atrial fibrillation (AF) in patients with HCM (Fig. 4A) [52, 53]. Indeed, Tani et al. reported that LA volume was the most sensitive predictor for the occurrence of paroxysmal AF in patients with HCM [54]. In a retrospective longitudinal analysis, Debonnaire et al. showed that LA diameter and volume were positively correlated with new onset of AF in patients with HCM [55]. Interestingly, Costabel et al. demonstrated that the E/e´ ratio as well as LA enlargement were strong, independent predictors of AF [56]. Both elevated E/e´ and LA enlargement were observed at baseline in the patients with HCM enrolled in mavacamten clinical studies.
A The relationship between elevated wall stress, impaired relaxation, increased preload, and LA volume, and the risk of AF. B The effect of mavacamten on LA volume index (EXPLORER-HCM). Data from Hegde et al. J Am Coll Cardiol 2021;78:2518–32. AF atrial fibrillation, CI confidence interval, HCM hypertrophic cardiomyopathy, LA left atrial, LS least-squares, SD standard deviation
Consequently, mavacamten would be expected to decrease LA volume through its capacity to promote relaxation, among other potential mechanisms. Indeed, results from PIONEER-OLE, EXPLORER-HCM, and the EXPLORER-HCM CMR sub-study showed reductions in LAVI in patients with oHCM (Fig. 4B; Electronic Supplementary Material, Supplementary Table S2) [24, 28]. The effect on LAVI was not observed during the 16-week MAVERICK-HCM study in nHCM, possibly owing to the short duration of the study, but reductions have been observed in the MAVA-LTE MAVERICK cohort over longer periods of treatment (Supplementary Table S2) [21, 57]. In EXPLORER-HCM, the rates of AF were similar between the mavacamten and placebo groups [22]. The impact of mavacamten on the occurrence of AF is being investigated through ongoing observation of AF events in the MAVA-LTE study.
Furthermore, increased LV filling pressure in HCM could lead to elevated pulmonary pressures, which can manifest by an abnormal ventilatory response to exercise. The VE/VCO2 slope is a measure of ventilatory efficiency and can be used to identify an abnormal ventilatory response [58]. The reduction in VE/VCO2 slope observed in patients with symptomatic oHCM who were enrolled in the PIONEER-HCM and EXPLORER-HCM studies [20, 22] and who were treated with mavacamten may be indicative of reduced pulmonary congestion. However, VE/VCO2 slope is a complex biological parameter comprising several determinants which might also have influenced the improvements in ventilatory efficiency observed. Furthermore, improvements in the HCM Symptom Questionnaire (HCMSQ)-Shortness-of-Breath sub-score and in the numerical rating scale (NRS) dyspnea score have been reported in the EXPLORER-HCM and PIONEER studies, respectively, reinforcing the potential for a mechanistic correlation [20, 22].
3.5 Hypertrophy, Myocardial Ischemia/injury, and Fibrosis
The hypertrophied ventricular myocardium has an increased myocardial oxygen demand leading to abnormal myocardial blood flow, which can be regarded as a surrogate for myocardial ischemia in patients with HCM [59]. Because increases in troponin levels show high specificity for myocyte cell death, cTnI and cardiac troponin T (cTnT) are strong and specific indicators of myocardial injury, and are well-established biomarkers of ischemia and cardiac injury [60, 61]. Additionally, altered energetics observed in HCM are characterized by an increase in glucose uptake, which reflects an impaired metabolism and has been linked to troponin release in HCM [62]. In patients with HCM, altered energetics have also been proposed as a potential mechanism driving fibrosis [63], which is a histopathological hallmark of HCM and is regarded as a consequence of ischemic and hemodynamic stressors [64]. The non-invasive assessment of myocardial extracellular volume (ECV) fraction by CMR is an established measure of interstitial fibrosis [65, 66].
Although the exact cause of troponin release in HCM is still unresolved, owing to its beneficial effect on hypertrophy and in accordance with the aforementioned relationships (Fig. 5A), mavacamten would be expected to reduce the level of myocardial ischemia/injury and fibrosis, and therefore the levels of injury biomarkers (such as cTnI and cTnT) and ECV observed in patients with HCM. Indeed, reductions in cTnI level have been reported in the MAVERICK-HCM and EXPLORER-HCM studies (Fig. 5B; Electronic Supplementary Material, Supplementary Table S2).
A The relationship between elevated wall stress, myocardial ischemia/injury, fibrosis, impaired contractility, and increased preload. B The effect of mavacamten on cTnI levels (EXPLORER-HCM). Data from Olivotto et al. Lancet 2020;396:759–69. CI confidence interval, cTnI cardiac troponin I, ECV extracellular volume, HCM hypertrophic cardiomyopathy
3.6 Proposed Integrated Model of HCM Clinical Pathophysiology
Because HCM has an impact on multiple pathophysiological pathways, we aimed to organize the learnings from the mavacamten studies into an integrated model (Fig. 6).
A Proposed integrated model of HCM clinical pathophysiology. B The common mechanism with HFpEF. AF atrial fibrillation, HCM hypertrophic cardiomyopathy, HFpEF heart failure with preserved ejection fraction, LA left atrial, LV left ventricular, nHCM non-obstructive hypertrophic cardiomyopathy, oHCM obstructive hypertrophic cardiomyopathy
The proposed model places sarcomeric hyperactivity and hypercontractility at the center of the anatomical and pathophysiological pathways of HCM (Fig. 6A). By design, mavacamten is able to normalize the sarcomeric hyperactivity observed in HCM, and provides a mechanistically targeted therapeutic approach with downstream improvement in several of the various pathophysiological manifestations of HCM that extend well beyond LVOT obstruction reprieve and negative inotropy. The proposed pathophysiological model of HCM suggests a unifying mechanism for structural changes associated with HCM and other diseases of pathogenic hypertrophy such as heart failure with preserved ejection fraction (Fig. 6B) [67,68,69]. Figure 7 shows the various biomarkers used to assess the clinical course of the disease, and the effect of mavacamten on those biomarkers.
A The biomarkers of HCM. B The overall effect of mavacamten. AF atrial fibrillation, ECV extracellular volume, e´ early diastolic mitral annular velocity, HCM hypertrophic cardiomyopathy, LA left atrial, LV left ventricular, LVOT left ventricular outflow tract, nHCM non-obstructive hypertrophic cardiomyopathy, NT-proBNP N-terminal pro B-type natriuretic peptide
4 Discussion
The pathophysiology of HCM is complex, multifactorial, and not fully understood. Here, we propose an integrated model of HCM clinical pathophysiology based on established pathways of the disease and on the effect of mavacamten, the first-in-class, selective inhibitor of cardiac myosin. The pathway model highlights the proposed positive feedback loop in oHCM, placing sarcomeric overactivity as its main effector, and emphasizing the connection of this loop to the broader pathways impacted in HCM. It is not anticipated that all pathways are equal nor that the pathways are similarly induced in all patients with HCM at all times. Indeed, it is well established that the presentations of HCM are diverse with temporal variability. The results with mavacamten serve as a means to better understand HCM pathophysiology, the relationship between HCM’s impacts on systolic and diastolic function, and improvements that could address the underlying biology of HCM. The lack of effect in patients with nHCM on exercise performance, NT-proBNP levels, diastolic function or quality of life observed with ranolazine, a late sodium inhibitor, illustrates the specific action of mavacamten and strengthen the model hypothesized here [70].
Owing to its mechanism of action, mavacamten reduces hypercontractility by returning more myosin heads to the ‘off state.’ Multiple ex vivo and in vivo studies support the clinical observations summarized in this article. For instance, in vivo, mavacamten has been shown to blunt cardiac hypercontractility, alleviating LVOT obstruction and improving systemic hemodynamic. In cats with naturally occurring HCM [71] and in mice harboring HCM pathogenic mutation in myosin, mavacamten has been shown to not only prevent hypertrophy (i.e., reduce hypertrophic wall thickness), but also to prevent the activation of pro-inflammatory, pro-fibrotic, and pro-hypertrophic pathways, including the normalization of natriuretic peptide gene expressions [19]. Results from the studies of mavacamten enabled the development of the proposed model of HCM pathophysiology that is generalizable to both oHCM and nHCM, and illustrate the impact of normalizing the ratio of ‘on’ and ‘off’ myosin heads on the HCM heart. The model allows the integration of the diverse results of mavacamten treatment in oHCM and nHCM studies to illustrate the potential relationship between obstruction, LV hypertrophy, myocardial ischemia/injury and LA volume, and diastolic filling. This integrated model links the ability of mavacamten to reverse the pathophysiology of HCM to resolve clinical presentations and suggests possible extensions to other diseases caused by a similar underlying pathophysiology.
It should be noted that, although based on pre-clinical and clinical evidence, our model remains hypothetical and has the potential to serve as a blueprint for future studies. This model will most certainly require refinement as more evidence emerges from other studies. For instance, pre-clinical and clinical evidence from other cardiac myosin inhibitors such as aficamten, which has been reported to reduce LVOT gradients in a phase 2 trial, would help in refining and strengthening our model [72]. Additionally, it would be of great interest to assess the effect of cardiac myosin inhibitors in patients with heart failure with preserved ejection fraction. Another area of potential future research is the investigation of the pathophysiology of HCM and of the effects of cardiac myosin inhibitors in a pediatric patient population.
5 Conclusions
Building from well-known pathophysiological pathways of HCM and previously published data on the effects of mavacamten on accepted HCM biomarkers, we constructed an integrated model of HCM clinical pathophysiology that identifies sarcomeric hyperactivity as a key driver of disease.
References
Sequeira V, Bertero E, Maack C. Energetic drain driving hypertrophic cardiomyopathy. FEBS Lett. 2019;593(13):1616–26. https://doi.org/10.1002/1873-3468.13496.
Hershberger RE, Cowan J, Morales A, et al. Progress with genetic cardiomyopathies: screening, counseling, and testing in dilated, hypertrophic, and arrhythmogenic right ventricular dysplasia/cardiomyopathy. Circ Heart Fail. 2009;2(3):253–61. https://doi.org/10.1161/circheartfailure.108.817346.
Gersh BJ, Maron BJ, Bonow RO, et al. ACCF/AHA guideline for the diagnosis and treatment of hypertrophic cardiomyopathy: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. Developed in collaboration with the American Association for Thoracic Surgery, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2011;58(25):e212–60. https://doi.org/10.1016/j.jacc.2011.06.011.
Maron BJ, Maron MS, Semsarian C. Genetics of hypertrophic cardiomyopathy after 20 years: clinical perspectives. J Am Coll Cardiol. 2012;60(8):705–15. https://doi.org/10.1016/j.jacc.2012.02.068.
Alfares AA, Kelly MA, McDermott G, et al. Results of clinical genetic testing of 2,912 probands with hypertrophic cardiomyopathy: expanded panels offer limited additional sensitivity. Genet Med. 2015;17(11):880–8. https://doi.org/10.1038/gim.2014.205.
Hoskins AC, Jacques A, Bardswell SC, et al. Normal passive viscoelasticity but abnormal myofibrillar force generation in human hypertrophic cardiomyopathy. J Mol Cell Cardiol. 2010;49(5):737–45. https://doi.org/10.1016/j.yjmcc.2010.06.006.
van Dijk SJ, Paalberends ER, Najafi A, et al. Contractile dysfunction irrespective of the mutant protein in human hypertrophic cardiomyopathy with normal systolic function. Circ Heart Fail. 2012;5(1):36–46. https://doi.org/10.1161/circheartfailure.111.963702.
Ho CY. New paradigms in hypertrophic cardiomyopathy: insights from genetics. Prog Pediatr Cardiol. 2011;31(2):93–8. https://doi.org/10.1016/j.ppedcard.2011.02.005.
McNamara JW, Li A, Smith NJ, et al. Ablation of cardiac myosin binding protein-C disrupts the super-relaxed state of myosin in murine cardiomyocytes. J Mol Cell Cardiol. 2016;94:65–71. https://doi.org/10.1016/j.yjmcc.2016.03.009.
Alamo L, Ware JS, Pinto A, et al. Effects of myosin variants on interacting-heads motif explain distinct hypertrophic and dilated cardiomyopathy phenotypes. Elife. 2017;6: e24634. https://doi.org/10.7554/eLife.24634.
Trivedi DV, Adhikari AS, Sarkar SS, et al. Hypertrophic cardiomyopathy and the myosin mesa: viewing an old disease in a new light. Biophys Rev. 2018;10(1):27–48. https://doi.org/10.1007/s12551-017-0274-6.
Anderson RL, Trivedi DV, Sarkar SS, et al. Deciphering the super relaxed state of human β-cardiac myosin and the mode of action of mavacamten from myosin molecules to muscle fibers. Proc Natl Acad Sci USA. 2018;115(35):e8143–52. https://doi.org/10.1073/pnas.1809540115.
Spudich JA. Three perspectives on the molecular basis of hypercontractility caused by hypertrophic cardiomyopathy mutations. Pflugers Arch. 2019;471(5):701–17. https://doi.org/10.1007/s00424-019-02259-2.
Maron BJ. Clinical course and management of hypertrophic cardiomyopathy. N Engl J Med. 2018;379(7):655–68. https://doi.org/10.1056/NEJMra1710575.
Bonne G, Carrier L, Richard P, et al. Familial hypertrophic cardiomyopathy: from mutations to functional defects. Circ Res. 1998;83(6):580–93. https://doi.org/10.1161/01.res.83.6.580.
Sherrid MV, Gunsburg DZ, Moldenhauer S, et al. Systolic anterior motion begins at low left ventricular outflow tract velocity in obstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2000;36(4):1344–54. https://doi.org/10.1016/s0735-1097(00)00830-5.
Elliott PM, Anastasakis A, Borger MA, et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the task force for the diagnosis and management of hypertrophic cardiomyopathy of the European Society of Cardiology (ESC). Eur Heart J. 2014;35(39):2733–79. https://doi.org/10.1093/eurheartj/ehu284.
Kawas RF, Anderson RL, Ingle SRB, et al. A small-molecule modulator of cardiac myosin acts on multiple stages of the myosin chemomechanical cycle. J Biol Chem. 2017;292(40):16571–7. https://doi.org/10.1074/jbc.M117.776815.
Green EM, Wakimoto H, Anderson RL, et al. A small-molecule inhibitor of sarcomere contractility suppresses hypertrophic cardiomyopathy in mice. Science. 2016;351(6273):617–21. https://doi.org/10.1126/science.aad3456.
Heitner SB, Jacoby D, Lester SJ, et al. Mavacamten treatment for obstructive hypertrophic cardiomyopathy: a clinical trial. Ann Intern Med. 2019;170(11):741–8. https://doi.org/10.7326/m18-3016.
Ho CY, Mealiffe ME, Bach RG, et al. Evaluation of mavacamten in symptomatic patients with nonobstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2020;75(21):2649–60. https://doi.org/10.1016/j.jacc.2020.03.064.
Olivotto I, Oreziak A, Barriales-Villa R, et al. Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2020;396(10253):759–69. https://doi.org/10.1016/s0140-6736(20)31792-x.
Heitner SB, Lester S, Wang A, et al. Abstract 13962: precision pharmacological treatment for obstructive hypertrophic cardiomyopathy with mavacamten: one-year results from PIONEER-OLE. Circulation. 2019;140(Suppl_1):A13962-A. https://doi.org/10.1161/circ.140.suppl_1.13962.
Saberi S, Cardim N, Yamani MH, et al. Mavacamten favorably impacts cardiac structure in obstructive hypertrophic cardiomyopathy: EXPLORER-HCM CMR substudy analysis. Circulation. 2021;143:606–8. https://doi.org/10.1161/circulationaha.120.052359.
Rader F, Choudhury L, Saberi S, et al. Long-term safety of mavacamten in patients with obstructive hypertrophic cardiomyopathy: interim results of the MAVA-long term extension (LTE) study. J Am Coll Cardiol. 2021;77(18_Supplement_1):532. https://doi.org/10.1016/S0735-1097(21)01891-X.
Vilcant V, Hai O. Left ventricular outflow tract obstruction. StatPearls. Treasure Island (FL): StatPearls Publishing LLC; 2020.
De Vecchi A, Marlevi D, Nordsletten DA, et al. Left ventricular outflow obstruction predicts increase in systolic pressure gradients and blood residence time after transcatheter mitral valve replacement. Sci Rep. 2018;8(1):15540. https://doi.org/10.1038/s41598-018-33836-7.
Hegde S, Lester SJ, Solomon SD, et al. Effect of mavacamten on echocardiographic features in symptomatic patients with obstructive hypertrophic cardiomyopathy. J Am Coll Cardiol. 2021;78(25):2518–32.
Wijnker PJM, Sequeira V, Kuster DWD, et al. Hypertrophic cardiomyopathy: a vicious cycle triggered by sarcomere mutations and secondary disease hits. Antioxid Redox Signal. 2019;31(4):318–58. https://doi.org/10.1089/ars.2017.7236.
Brito D, Matias JS, Sargento L, et al. Plasma N-terminal pro-brain natriuretic peptide: a marker of left ventricular hypertrophy in hypertrophic cardiomyopathy. Rev Port Cardiol. 2004;23(12):1557–82.
Kim SW, Park SW, Lim SH, et al. Amount of left ventricular hypertrophy determines the plasma N-terminal pro-brain natriuretic peptide level in patients with hypertrophic cardiomyopathy and normal left ventricular ejection fraction. Clin Cardiol. 2006;29(4):155–60. https://doi.org/10.1002/clc.4960290406.
Saberi S, Cardim N, Yamani MH, et al. Abstract 18654: Mavacamten favorably impacts cardiac structure in obstructive hypertrophic cardiomyopathy: EXPLORER-HCM CMR substudy analysis. Circulation. 2020;142(24):e470–500.
Satpathy C, Mishra TK, Satpathy R, et al. Diagnosis and management of diastolic dysfunction and heart failure. Am Fam Physician. 2006;73(5):841–6.
Kalra A, Harris KM, Maron BA, et al. Relation of doppler tissue imaging parameters with heart failure progression in hypertrophic cardiomyopathy. Am J Cardiol. 2016;117(11):1808–14. https://doi.org/10.1016/j.amjcard.2016.03.018.
Saccheri MC, Cianciulli TF, Lax JA, et al. Impaired myocardial function in hypertrophic cardiomyopathy. Echocardiography. 2009;26(6):657–64. https://doi.org/10.1111/j.1540-8175.2008.00871.x.
De Marchi SF, Allemann Y, Seiler C. Relaxation in hypertrophic cardiomyopathy and hypertensive heart disease: relations between hypertrophy and diastolic function. Heart. 2000;83(6):678–84. https://doi.org/10.1136/heart.83.6.678.
Yelamarty RV, Moore RL, Yu FT, et al. Relaxation abnormalities in single cardiac myocytes from renovascular hypertensive rats. Am J Physiol. 1992;262(4 Pt 1):C980–90. https://doi.org/10.1152/ajpcell.1992.262.4.C980.
Douglas PS, Tallant B. Hypertrophy, fibrosis and diastolic dysfunction in early canine experimental hypertension. J Am Coll Cardiol. 1991;17(2):530–6. https://doi.org/10.1016/s0735-1097(10)80127-5.
Ciampi Q, Betocchi S, Losi MA, et al. Effect of hypertrophy on left ventricular diastolic function in patients with hypertrophic cardiomyopathy. Heart Int. 2006;2(2):106. https://doi.org/10.4081/hi.2006.106.
Avegliano G, Costabel JP, Huguet M, et al. Influence of dynamic obstruction and hypertrophy location on diastolic function in hypertrophic cardiomyopathy. J Cardiovasc Med (Hagerstown). 2014;15(3):207–13. https://doi.org/10.2459/JCM.0b013e3283638093.
Wigle ED, Sasson Z, Henderson MA, et al. Hypertrophic cardiomyopathy. The importance of the site and the extent of hypertrophy. A review. Prog Cardiovasc Dis. 1985;28(1):1–83. https://doi.org/10.1016/0033-0620(85)90024-6.
Chacko BR, Karur GR, Connelly KA, et al. Left ventricular structure and diastolic function by cardiac magnetic resonance imaging in hypertrophic cardiomyopathy. Indian Heart J. 2018;70(1):75–81. https://doi.org/10.1016/j.ihj.2016.12.021.
Finocchiaro G, Haddad F, Pavlovic A, et al. How does morphology impact on diastolic function in hypertrophic cardiomyopathy? A single centre experience. BMJ Open. 2014;4(6): e004814. https://doi.org/10.1136/bmjopen-2014-004814.
Hayashida W, Kumada T, Kohno F, et al. Left ventricular regional relaxation and its nonuniformity in hypertrophic nonobstructive cardiomyopathy. Circulation. 1991;84(4):1496–504. https://doi.org/10.1161/01.cir.84.4.1496.
Hayashida W, Kumada T, Kambayashi M, et al. Early diastolic regional function of the hypertrophied left ventricle. Int J Cardiol. 1996;53(2):153–62. https://doi.org/10.1016/0167-5273(95)02534-0.
Mitter SS, Shah SJ, Thomas JD. A test in context: E/A and E/e’ to assess diastolic dysfunction and LV filling pressure. J Am Coll Cardiol. 2017;69(11):1451–64. https://doi.org/10.1016/j.jacc.2016.12.037.
Park JH, Marwick TH. Use and limitations of E/e’ to assess left ventricular filling pressure by echocardiography. J Cardiovasc Ultrasound. 2011;19(4):169–73. https://doi.org/10.4250/jcu.2011.19.4.169.
Okura H, Takada Y, Kubo T, et al. Tissue Doppler-derived index of left ventricular filling pressure, E/E’, predicts survival of patients with non-valvular atrial fibrillation. Heart. 2006;92(9):1248–52. https://doi.org/10.1136/hrt.2005.082594.
Nagueh SF, Smiseth OA, Appleton CP, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2016;17(12):1321–60. https://doi.org/10.1093/ehjci/jew082.
Badran HM, Soltan G, Almeleigi R, et al. Prognostic significance of left ventricular end diastolic pressure using E/E’ in patients with hypertrophic cardiomyopathy. Echocardiography. 2019;36(12):2167–75. https://doi.org/10.1111/echo.14539.
Appleton CP, Galloway JM, Gonzalez MS, et al. Estimation of left ventricular filling pressures using two-dimensional and Doppler echocardiography in adult patients with cardiac disease. Additional value of analyzing left atrial size, left atrial ejection fraction and the difference in duration of pulmonary venous and mitral flow velocity at atrial contraction. J Am Coll Cardiol. 1993;22(7):1972–82. https://doi.org/10.1016/0735-1097(93)90787-2.
Limongelli G, Fioretti V, Di Maio M, et al. Left atrial volume during stress is associated with increased risk of arrhythmias in patients with hypertrophic cardiomyopathy. J Cardiovasc Echogr. 2019;29(1):1–6. https://doi.org/10.4103/jcecho.jcecho_45_18.
Guttmann OP, Rahman MS, O’Mahony C, et al. Atrial fibrillation and thromboembolism in patients with hypertrophic cardiomyopathy: systematic review. Heart. 2014;100(6):465–72. https://doi.org/10.1136/heartjnl-2013-304276.
Tani T, Tanabe K, Ono M, et al. Left atrial volume and the risk of paroxysmal atrial fibrillation in patients with hypertrophic cardiomyopathy. J Am Soc Echocardiogr. 2004;17(6):644–8. https://doi.org/10.1016/j.echo.2004.02.010.
Debonnaire P, Joyce E, Hiemstra Y, et al. Left atrial size and function in hypertrophic cardiomyopathy patients and risk of new-onset atrial fibrillation. Circ Arrhythm Electrophysiol. 2017;10(2): e004052. https://doi.org/10.1161/circep.116.004052.
Costabel JP, Galve E, Terricabras M, et al. E/e’ ratio and left atrial area are predictors of atrial fibrillation in patients with hypertrophic cardiomyopathy. Echocardiography. 2018;35(7):935–40. https://doi.org/10.1111/echo.13857.
Owens A, Sherrid M, Wong TC, et al. Long-term efficacy and safety of mavacamten in patients with non-obstructive hypertrophic cardiomyopathy: interim results from the MAVERICK-LTE cohort of the MAVA-LTE study (abstract). Accepted to the AHA Scientific Sessions 2021 (not published yet). 2021. https
Weatherald J, Sattler C, Garcia G, et al. Ventilatory response to exercise in cardiopulmonary disease: the role of chemosensitivity and dead space. Eur Respir J. 2018;51(2):1700860. https://doi.org/10.1183/13993003.00860-2017.
Maron MS, Olivotto I, Maron BJ, et al. The case for myocardial ischemia in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2009;54(9):866–75. https://doi.org/10.1016/j.jacc.2009.04.072.
Cambronero F, Marín F, Roldán V, et al. Biomarkers of pathophysiology in hypertrophic cardiomyopathy: implications for clinical management and prognosis. Eur Heart J. 2009;30(2):139–51. https://doi.org/10.1093/eurheartj/ehn538.
Sato Y, Taniguchi R, Nagai K, et al. Measurements of cardiac troponin T in patients with hypertrophic cardiomyopathy. Heart. 2003;89(6):659–60. https://doi.org/10.1136/heart.89.6.659.
Aoyama R, Takano H, Kobayashi Y, et al. Evaluation of myocardial glucose metabolism in hypertrophic cardiomyopathy using 18F-fluorodeoxyglucose positron emission tomography. PLoS ONE. 2017;12(11): e0188479. https://doi.org/10.1371/journal.pone.0188479.
Raman B, Ariga R, Spartera M, et al. Progression of myocardial fibrosis in hypertrophic cardiomyopathy: mechanisms and clinical implications. Eur Heart J Cardiovasc Imaging. 2019;20(2):157–67. https://doi.org/10.1093/ehjci/jey135.
Ho JE, Shi L, Day SM, et al. Biomarkers of cardiovascular stress and fibrosis in preclinical hypertrophic cardiomyopathy. Open Heart. 2017;4(2): e000615. https://doi.org/10.1136/openhrt-2017-000615.
Ho CY, Abbasi SA, Neilan TG, et al. T1 measurements identify extracellular volume expansion in hypertrophic cardiomyopathy sarcomere mutation carriers with and without left ventricular hypertrophy. Circ Cardiovasc Imaging. 2013;6(3):415–22. https://doi.org/10.1161/circimaging.112.000333.
Yang EY, Ghosn MG, Khan MA, et al. Myocardial extracellular volume fraction adds prognostic information beyond myocardial replacement fibrosis. Circ Cardiovasc Imaging. 2019;12(12): e009535. https://doi.org/10.1161/circimaging.119.009535.
Sartipy U, Dahlström U, Fu M, et al. Atrial fibrillation in heart failure with preserved, mid-range, and reduced ejection fraction. JACC Heart Fail. 2017;5(8):565–74. https://doi.org/10.1016/j.jchf.2017.05.001.
Kotecha D, Lam CS, Van Veldhuisen DJ, et al. Heart failure with preserved ejection fraction and atrial fibrillation: vicious twins. J Am Coll Cardiol. 2016;68(20):2217–28. https://doi.org/10.1016/j.jacc.2016.08.048.
Obokata M, Reddy YNV, Melenovsky V, et al. Myocardial injury and cardiac reserve in patients with heart failure and preserved ejection fraction. J Am Coll Cardiol. 2018;72(1):29–40. https://doi.org/10.1016/j.jacc.2018.04.039.
Olivotto I, Camici PG, Merlini PA, et al. Efficacy of ranolazine in patients with symptomatic hypertrophic cardiomyopathy: the RESTYLE-HCM randomized, double-blind, placebo-controlled study. Circ Heart Fail. 2018;11(1): e004124. https://doi.org/10.1161/circheartfailure.117.004124.
Stern JA, Markova S, Ueda Y, et al. A small molecule inhibitor of sarcomere contractility acutely relieves left ventricular outflow tract obstruction in feline hypertrophic cardiomyopathy. PLoS ONE. 2016;11(12): e0168407. https://doi.org/10.1371/journal.pone.0168407.
Cytokinetics. Phase II clinical trial of CK-274 demonstrated consistent and clinically meaningful reductions in left ventricular outflow tract gradients within 2 weeks in patients with obstructive hypertrophic cardiomyopathy. 2021 [cited 2022 February]. https://ir.cytokinetics.com/news-releases/news-release-details/cytokinetics-announces-positive-topline-results-redwood-hcm.
Acknowledgements
The authors would like to thank all the patients and participants who were included in these studies, and the study investigators and personnel who contributed to the collection of the data. Medical writing and editorial support were provided by Nicolas Bertheleme, PhD, of Oxford PharmaGenesis, Oxford, UK, with financial support from MyoKardia, Inc., a wholly owned subsidiary of Bristol Myers Squibb.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by MyoKardia, Inc., a wholly owned subsidiary of Bristol Myers Squibb.
Conflict of interest
AJS and RM are employees of MyoKardia, Inc., a wholly owned subsidiary of Bristol Myers Squibb. JME, MEM, and CLdR were employees of MyoKardia, Inc., a wholly owned subsidiary of Bristol Myers Squibb.
Ethical approval
This article does not contain any previously unpublished studies with human participants or animals performed by any of the authors.
Availability of data
The data underlying this article will be shared on reasonable request to the corresponding author.
Code availability
Not applicable.
Consent for publication
The authors, jointly and severally, give the publisher permission to publish the work.
Consent to participate
Not applicable.
Author contributions
All the authors contributed to the conception and design of the work. All the authors participated in the drafting of this manuscript and provided final approval of the submitted version of the article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Edelberg, J.M., Sehnert, A.J., Mealiffe, M.E. et al. The Impact of Mavacamten on the Pathophysiology of Hypertrophic Cardiomyopathy: A Narrative Review. Am J Cardiovasc Drugs 22, 497–510 (2022). https://doi.org/10.1007/s40256-022-00532-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40256-022-00532-x