Abstract
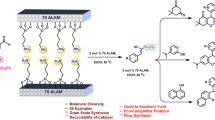
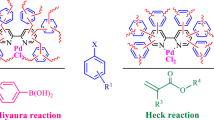
An ordered cyclopallated thiophene imine self-assembly monolayer(Si@Pd3TI) was designed and fabricated. It was characterized by water contact angle(WCA), ultraviolet-visible spectroscopy(UV), cyclic voltammetry(CV), infrared(IR) spectrum, atomic force microscopy(AFM) and X-ray photoelectron spectroscopy(XPS). Its catalytic performance for Suzuki coupling reaction and catalytic mechanism were systematic investigated. Si@Pd3TI was validated as a heterogeneous catalyst identified by poisoning tests, hot filtration test and three-phase test. The heterogeneous catalytic mechanism was investigated by WCA, UV, Raman spectrum(RS), AFM, XPS and density functional theory(DFT). The catalytic mechanism proceeded via surface-catalysis process, on which the Pd(II)/Pd(I)/Pd(0) synergistic active center acted likely as “multimetallic cluster”. It played a great role for catalyzing coupling reaction, in which the real active species was Pd(I).
Similar content being viewed by others
References
Li W. W., Hendriks K. H., Furlan A., Christian R. W. S., Wienk M. M., René A., Janssen J., J. Am. Chem. Soc., 2013, 135(50), 18942
Mishra A., Ma C. Q., Chem. Rev., 2009, 109, 1141
Vercelli B., Zotti G., Berlin A., Pasini M., Botta C., Gerbasi R., Nelson T. L., McCullough R. D., J. Phys. Chem. C, 2012, 116(2), 2033
Otley M. T., Alamer F. A., Zhu Y. M., Singhaviranon A., Zhang X. Z., Li M. F., Kumar A., Sotzing G. A., ACS Appl. Mater. Interfaces, 2014, 6(3), 1734
Maynor B. W., Filocamo S. F., Grinstaff M. W., Liu J., J. Am. Chem. Soc., 2002, 124(4), 522
Suzuki A., Chem. Commun., 2005, 4759
Albano V. G., Bandini M., Barbarella G., Melucci M., Monari M., Piccinelli F., Tommasi S., Umani-Ronchi A., Chem. Eur. J., 2006, 12, 667
Richard K. P., Swager T. M., J. Am. Chem. Soc., 1999, 121(38), 8825
Bhunia S., Koner A., Indian J. Chem., 2011, 50A, 1380
Huang P. P., Xue Z. Q., Li T. S., Liu Z. Y., Wei D. H., Liu M. H., Wu Y. J., ChemCatChem, 2018, 10, 5141
Liu H., Xue X. X., Li T. S, Wang J., Xu W. J., Liu M. H., Chen P. L., Wu Y. J., RSC Adv., 2016, 6, 84815
Wang L. H., Huang P. P., Yang J., Li T. S., Mao L. Y., Liu M. H., Wu Y. J., RSC Adv., 2018, 8, 31860
Albano V. G., Bandini M., Moorlag C., Piccinelli F., Pietrangelo A., Tommasi S., Umani-Ronchi A., Wolf M. O., Organometallics, 2007, 26, 4373
Zhang L., Colella N. S., Liu F., Trahan S., Baral J. K., Winter H. H., Mannsfeld S. C. B., Briseno A. L., J. Am. Chem. Soc., 2013, 135, 844
Nogues C., Lang P., Desbat B., Buffeteau T., Leiserowitz L., Langmuir, 2008, 24(16), 8458
Yang F., Zhang G. D., Wu Y. J., Green Chemistry, 2013, 15(4), 1055
Mu B., Li T. S., Li J. Y., Wu Y. J., J. Organomet. Chem., 2008, 693, 1243
Mu B., Li T. S., Fu Z. H., Wu Y. J., Catal. Commun., 2009, 10, 1497
Li Y., Fu Z. H., Rao L., Wang L., Li R., Li T.S., Wu Y. J., J. Chin. Chem. Soc., 2014, 61(3), 397
Mi X., Huang M. M., Zhang J., Wang C., Wu Y. J., Org. Lett., 2013, 15(24), 6266
Li X. J., Zou D. P., Leng F. Q., Sun C. X., Li J. Y., Wu Y. J., Wu Y. S., Chem. Commun., 2013, 49, 312
Wang J., Mu B., Fu Z. H., Wang L., Li T. S., Wu Y. J., Chin. J. Catal., 2014, 35, 1059
Fu Z. H., Li T. S., Mu B., Mao L. Y., Li G. Q., Xu W. J., Wu Y. J., J. Mol. Catal. A: Chem., 2012, 363/364, 200
Mu B., Li T. S., Li C., Liu P. P., Shang W., Wu Y. J., Tetrahedron, 2009, 65, 2599
Zhao N., Li T. S., Zhai Z., Qiu J. J., Xu W. J., Liu H., Wu Y. J., ChemCatChem, 2013, 5, 1481
Fu Z. H., Zhang N., Li T. S., Xu W. J., Wu Y. J., J. Colloid. &Intef. Sci., 2013, 394, 409
Fu Z. H., Liu J., He X. H., Li T. S., Wu Y. J., RSC Adv., 2014, 4(50), 26413
Zhao N., Wang F., Zhou M. L., Li T. S., Liu H., Xu W. J., Wu Y. J., Chin. J. Catal., 2013, 34(8), 1583
Zou K. K., Liu H., Li T. S., Chen P. L., Liu M. H., Wu Y. J., RSC Adv., 2015, 5(2), 16654
Reddinger J. L., Reynolds J. R., Chem. Mater., 1998, 10, 1236
Liu H., Li T. S., Xue X. X., Xu W. J., Wu Y. J., Catal. Sci.&Technol., 2016, 6(6), 1667
Aaronson B. D. B., Garoz-Ruiz J., Byers J. C., Colina A., Unwin P. R., Langmuir, 2015, 31(46), 12814
Gogoll A., Pettersson L. G. M., Bohman O., Siegbahn H. O. G., Organometallics, 1992, 11(5), 1784
Fu Z. H., Li T. S., He X. H., Liu J., Xu W. J., Wu Y. J., J. Mol. Catal. A: Chem., 2014, 395, 293
Xue Z. Q., Huang P. P., Li T. S., Qin P. X., Xiao D., Liu M. H., Chen P. L., Wu Y. J., Nanoscale, 2017, 9, 781
Huang P. P., Song E. R., Sun Y. M., Li T. S., Wei D. H., Liu M. H., Wu Y. J., Molecular Catalysis, 2019, 469, 75
Rebek J., Gavina F., J. Am. Chem. Soc., 1974, 96(22), 7112
Crudden C. M., Sateesh M., Lewis R., J. Am. Chem. Soc., 2005, 127, 10045
Richardson J. M., Jones C. W., J. Catal., 2007, 251, 80
Wei D. H., Huang X. Q., Qiao Y., Rao J. J., Wang L., Liao F., Zhan C. G., ACS Catalysis, 2017, 7, 4623
Wang Y., Wei D. H., Tang M. S., J. Org. Chem., 2017, 82, 13043
Wang Y., Du C., Wang Y. Y., Guo X. K., Fang L., Song M. P., Niu J. L., Wei D. H., Adv. Synth. Catal., 2018, 360(14), 2668
Li X., Wang Y. Y., Wang Y., Tang M. S., Qu L. B., Li Z. J., Wei D. H., J. Org. Chem., 2018, 83(15), 8543
Liu N., Xie Y. F., Wang C., Li S. J., Wei D. H., Dai B., ACS Catalysis, 2018, 8, 9945
Acknowledgements
The authors gratefully thank Prof. MAO Luyuan(College of Materials Science and Engineering, Zhengzhou University, China) for AFM measurement.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(No.21861132002) and the Natural Science Foundation of Henan Province, China(No.192102210046).
Supporting Information
40242_2020_9087_MOESM1_ESM.pdf
Self-assembly Palladacycle Thiophene Imine Monolayer—Investigating on Catalytic Activity and Mechanism for Coupling Reaction
Rights and permissions
About this article
Cite this article
Li, L., Xue, X., Sun, Y. et al. Self-assembly Palladacycle Thiophene Imine Monolayer—Investigating on Catalytic Activity and Mechanism for Coupling Reaction. Chem. Res. Chin. Univ. 36, 821–828 (2020). https://doi.org/10.1007/s40242-020-9087-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-020-9087-4