Abstract
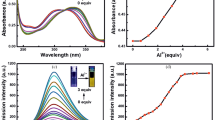
A novel molecular probe system(NapTpa-dnbs) was prepared based on the chromophore napthalimide fused triphenylamine unit. NapTpa-dnbs was tuned to absorb a large fraction of UV and near-infrared radiation. The turn-on fluorescent response of NapTpa-dnbs to thiophenol caused an increased fluorescent emission by a factor of 11.5 at 597 nm in a PBS buffer solution. The photoinduced electron transform(PET) process of the probe became less feasible with the addition of thiophenol than that of the free probe. NapTpa-dnbs demonstrated high selective and sensitive detection toward thiophenol with respect to other competitive amine acids. The detection limit was determined to be 2.03×10−7 mol/L thiophenol. The sensing process is rapid(3 min). NapTpa-dnbs can accurately detect the thiophenol with a qualified recovery in real water samples. NapTpa-dnbs on test strips can detect the thiophenol vapour in the atmosphere.
Similar content being viewed by others
References
Hong R., Han G., Joseph M. F., Kim B. G., Neil S. F., Vincent M. R., Journal of the American Chemical Society, 2006, 128(4), 1078
Verma R. P., Kapur S., Barberena O., Shusterman A., Hansch C. H., Selassie C. D., Chemical Research in Toxicology, 2003, 16(3), 276
Proctor N. H., Hughes J. P., Hathaway G. J., Proctor & Hughes Chemical Hazards of the Workplace, 2004
Heil T. P., Lindsay R. C. Journal of Environmental Science and Health, Part B, 1989, 24(4), 361
Wu H., Jiang J., Gu X., Tong C., Microchimica Acta, 2017, 184(7), 2291
Wu H., Tong C., ACS Sensors, 2018, 3(8), 1539
Wu J., Liu W., Ge J., Zhang H., Wang P., Chemical Society Reviews, 2011, 40(7), 3483
Yin J., Kwon Y., Kim D., Lee D., Kim G., Hu Y., Ryu J. H., Yoon J., Journal of the American Chemical Society, 2014, 136(14), 5351
Jung H. S., Chen X., Kim J. S., Yoon J., Cheminform, 2013, 42(14), 6019
Yu F., Li P., Song P., Wang B., Zhao J., Han K., Chemical Communications, 2012, 48(41), 4980
Kim G. J., Lee K., Kwon H., Kim H. J., Organic Letters, 2011, 13(11), 2799
Wang Z., Han D. M., Jia W. P., Zhou Q. Z., Deng W. P., Analytical Chemistry, 2012, 84(11), 4915
Sreejith S., Divya K. P., Ajayaghosh A., Angewandte Chemie International Edition, 2010, 47(41), 7883
Wu H., Tong C., Journal of Agricultural and Food Chemistry, 2019, 67(10), 2794
Kim K. R., Kim H. J., Hong J. I., Analytical Chemistry, 2019, 91(2), 1353
Yin C., Huo F., Zhang J., Martínez-Máñez R., Yang Y., Lv H., Li S., Chemical Society Reviews, 2013, 42(14), 6032
Hu J. J., Wong N. K., Ye S., Chen X., Lu M. Y., Zhao A. Q., Guo Y., Ma C. H., Leung Y. H., Shen J., Journal of the American Chemical Society, 2015, 137(21), 6837
Shiraishi Y., Yamamoto K., Sumiya S., Hirai T., Chemical Communications, 2013, 49(99), 11680
Kand D., Mishra P. K., Saha T., Lahiri M., Talukdar P., Analyst, 2012, 137(17), 3921
Kand D., Mandal P. S., Datar A., Talukdar P., Dyes and Pigments, 2014, 106(1), 25
Tang B., Xing Y., Li P., Zhang N., Yu F., Yang G., Journal of the American Chemical Society, 2007, 129(38), 11666
Yi L., Li H., Sun L., Liu L., Zhang C., Xi Z., Angewandte Chemie International Edition, 2010, 121(22), 4094
Tanaka F., Mase N., Barbas C., Chemical Communications, 2004, 15(15), 1762
Bouffard J., Kim Y., Swager T. M., Weissleder R., Hilderbrand S. A., Organic Letters, 2008, 10(1), 37
RosLis J., García B., Jiménez D., Martínez-Máñez R., Sancenón F., Soto J., Gonzalvo F., Valldecabres M. C., Journal of the American Chemical Society, 2004, 126(13), 4064
Hewage H. S., Anslyn E. V., Journal of the American Chemical Society, 2009, 131(36), 13099
Lin W., Long L., Tan W., Chemical Communications, 2010, 46(9), 1503
Yu D., Huang F., Ding S., Feng G., Analytical Chemistry, 2014, 86(17), 8835
Sun Q., Yang S., Wu L., Yang W., Yang G., Analytical Chemistry, 2016, 88(4), 2266
Xia T., Wang L., Qu Y., Rui Y., Cao J., Hu Y., Yang J., Wu J., Xu J., Journal of Materials Chemistry C, 2016, 4(24), 5695
Qian X., Xiao Y., Xu Y., Guo X., Qian J., Zhu W., Chemical Communications, 2010, 46(35), 6418
Boens N., Leen V., Dehaen W., Chemical Society Reviews, 2012, 41(24), 8212
Maeda H., Matsuno H., Ushida M., Katayama K., Saeki K., Itoh N., Angewandte Chemie International Edition, 2010, 44(19), 2922
Yang L., Su Y., Geng Y., Zhang Y., Ren X., He L., Song X. Z., ACS Sensors, 2018, 3(9), 1863
Jiang W., Cao Y., Liu Y., Wang W., Chemical Communications, 2010, 46(11), 1944
Watanabe Y., Imai K., Analytical Biochemistry, 1981, 116(2), 471
Qi Q., Wang X., Fan L., Zheng B., Zeng W., Luo J., Huang K., Wang Q., Wu J., Organic Letter, 2015, 17(3), 724
Li Z., Zhu Z., Chueh C., Jo S., Luo J., Jang S., Jen A. K. Y., Journal of the American Chemical Society, 2016, 138(36), 11833
Wang X. B., Zhou J., Zhang D., Wang B., Analytical Methods, 2016, 8(38), 6916
Wang X., Jian C., Zhao C., Organic Biomolecular Chemistry, 2012, 10(24), 4689
Khandare D. G., Banerjee M., Gupta R., Kumar N., Ganguly A., Singh D., Chatterjee A., RSC Advances, 2016, 6(58), 52790
Zhang W., Liu X., Zhang H., Feng C., Liu C., Yu M., Wei L., Li Z., Journal of Materials Chemistry C, 2015, 3(31), 8248
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Innovation Fund for Graduate Students of Shanghai University of Engineering Science, China (Nos. 17KY0408, 18KY0416), the Talent Program of Shanghai University of Engineering Science, China(No. 2017RC422017) and the Degree and Postgraduate Education Teaching Reform Research Project of Shanghai University of Engineering Science, China (No. 17XJC002).
Electronic Supplementary Information
40242_2019_9033_MOESM1_ESM.pdf
Fluorescent Turn-on Probe Based on Napthalimide Fused Triphenylamine Unit for Quickly Detecting Thiophenol in Aqueous Solution
Rights and permissions
About this article
Cite this article
Sun, J., Cao, J., Cheng, H. et al. Fluorescent Turn-on Probe Based on Napthalimide Fused Triphenylamine Unit for Quickly Detecting Thiophenol in Aqueous Solution. Chem. Res. Chin. Univ. 35, 990–996 (2019). https://doi.org/10.1007/s40242-019-9033-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-019-9033-5