Abstract
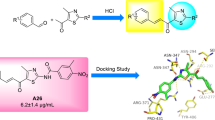
Twenty-seven novel chalcone derivatives were designed and synthesized as neuraminidase(NA) inhibitors. A concise suitable synthetic strategy was employed in the target compounds’ synthesis with relatively high yields. The synthesized compounds were evaluated for their inhibitory activities against the NA of influenza A virus in vitro. The results show that compound 9b possesses the most potent NA inhibitory activity. Structure-activity relationship studies indicate that the chalcone system and hydrogen bond donor substituent are significant for the NA inhibitory activity. And the chalcone derivatives containing pyran ring have better NA inhibitory activity than those without the pyran ring. In addition, molecular docking studies reveal that compounds 9b and 9u are in the good binding mode with Zanamivir binding sites. This study indicates that compound 9b could be selected as a potent compound for further structural optimization and development of novel NA inhibitors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhu Q., Bang T. H., Ohnuki K., Sawai T., Sawai K., Shimizua K., Sci. Rep., 2015, 5, 13194
Mills C. E., Robins J. M., Lipsitch M., Nature, 2004, 432(7019), 904
Wang Y., Lei F., Li X. L., He Y., Li J., Qiu R., Wu X. Y., Hai L., Wu Y., Chem. Res. Chinese Universities, 2015, 31(6), 9421
Subbarao K., Joseph T., Nat. Rev. Immunol., 2007, 7, 267
De Clercq E., Nat. Rev. Drug Discov., 2006, 5(12), 1015
Moscona A., N. Engl. J. Med., 2009, 360, 953
Liu D., Fan Z., Jiang J., Wei J., Xin J., Chem. Res. Chinese Universities, 2013, 29(4), 706
Sidwell R. W., Smee D. F., Expert Opin. Investig. Drugs, 2002, 11, 859
Palese P., Nat. Med., 2004, 10(12), 82
Shi F. Y., Fang H., Xu W. F., Chem. Res. Chinese Universities, 2016, 32(1), 28
Ghedin E., Sengamalay N. A., Shumway M., Zaborsky J., Taylor J., Lipman D. J., Fraser C. M., Taubenberger J. K., Salzberg S. L., Nature, 2005, 437, 1162
Chen B., Wang Y., Guo H., Zeng G., Eur. J. Med. Chem., 2016, 109, 199
Newman D. J., Cragg G. M., J. Nat. Prod., 2016, 79(3), 629
Cragg G. M., Newman D. J., BBA-Gen. Subjects, 2013, 1830(6), 3670
Newman D. J., Cragg G. M., J. Nat. Prod., 2012, 75(3), 311
Liu A., Wang H., Lee S. M., Wang Y., Du G., Bioorg. Med. Chem., 2008, 16, 7141
Park J. Y., Jeong H. J., Kim Y. M., Park S. J., Rho M. C., Park K. H., Ryu Y. B., Lee W. S., Bioorg. Med. Chem. Lett., 2011, 21, 5602
Ryu Y. B., Kim J. H., Park S. J., Chang J. S., Rho M. C., Bae K. H., Park K. H., Lee W. S., Bioorg. Med. Chem. Lett., 2010, 20(3), 971
Zu M., Yang F., Zhou W., Liu A., Du G., Zheng L., Antivir. Res., 2012, 94(3), 217
Zhang J., Wang Q., Fang H., Xu W., Liu A., Du G., J. Med. Chem., 2008, 16(7), 3839
Zhao H., Huang D., Caflisch A., Chem. Med. Chem., 2012, 7, 1983
DeLano W. L., PyMOL, 2002, http://www.pymol.org
Wallace A. C., Laskowski R. A., Thornton J. M., Protein Eng., 1995, 8, 127
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the Hunan Provincial Natural Science Foundation, China(No.2019JJ40030) and the National Natural Science Foundation of China(No.81673480).
Rights and permissions
About this article
Cite this article
Chen, A., Liang, Y., Ye, J. et al. Synthesis of Chalcone Derivatives Containing Furan or/and Pyran Ring as Neuraminidase Inhibitors. Chem. Res. Chin. Univ. 35, 395–402 (2019). https://doi.org/10.1007/s40242-019-8346-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-019-8346-8