Abstract
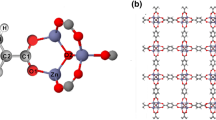
Periodic density functional theory(DFT) calculations are presented to describe the adsorption and decomposition of CH3OH on Ru(0001) surfaces with different coverages, including p(3×2), p(2×2), and p(2×1) unit cells, corresponding to monolayer(ML) coverages of 1/6, 1/4, and 1/2, respectively. The geometries and energies of all species involved in methanol dissociation were analyzed, and the initial decomposition reactions of methanol and the subsequent dehydrogenations reactions of CH3O and CH2OH were all computed at 1/2, 1/4, and 1/6 ML coverage on the Ru(0001) surface. The results show that coverage exerts some effects on the stable adsorption of CH3O, CH2OH, and CH3, that is, the lower the coverage, the stronger the adsorption. Coverage also exerts effects on the initial decomposition of methanol. C—H bond breakage is favored at 1/2 ML, whereas C—H and O—H bond cleavages are preferred at 1/4 and 1/6 ML on the Ru(0001) surface, respectively. At 1/4 ML coverage on the Ru(0001) surface, the overall reaction mechanism can be written as 9CH3OH→3CH3O+6CH2OH+9H→6CH2O+3CHOH+18H→ 7CHO+COH+CH+OH+26H→8CO+C+O+36H.
Similar content being viewed by others
References
Nislsen M., Alberico E., Baunann W., Drexler H. J., Junge H., Gladiali S., Beller M., Nature, 2013, 495, 85
Rdddington E., Sapienza A., Gurau B., Viswanathan R., Sarangapani S., Smotkin E. S., Mallouk T. E., Science, 1998, 280, 1735
Guo Q., Li L. R., Lu L., Ji Y., Lu T. H., Chem. J. Chinese Universities, 2012, 33(5), 1007
Hamnett A., Catal. Today., 1997, 38, 445
Oetjen H. F., Schmidt V. M., Stimming U., Trila F., J. Electrochem. Soc., 1996, 143, 3838
Gazdzicki, P., Jakob P., J. Phys. Chem. C, 2010, 114, 2655
Loveless B. T., Buda C., Neurock M., Iglesia E., J. Am. Chem. Soc., 2013, 135, 6107
Barros R. B., Garcia A. R., Ilharco L. M., J. Phys. Chem. B, 2001, 105, 11186
Barros R. B., Garcia A. R., Ilharco L. M., Surf. Sci., 2003, 532-535, 185
Barros R. B., Garcia A. R., Ilharco L. M., Surf. Sci., 2004, 572, 277
Hrbek J., DePaola R. A., Hoffmann F. M., J. Chem. Phys., 1984, 81, 2818
Gazdzicki P., Uvdal P., Jakob P., J. Chem. Phys., 2009, 130, 224703
García-Muelas R., Li Q., López N., ACS Catal., 2015, 5, 1027
Moura A. S., Fajín J. L. C., Pinto A. S. S., Mandado M., Cordeiro M. N. D. S., 2015, 119, 27382
Kresse G., Hafner J., Phys. Rev. B, 1994, 49, 14251
Kresse G, Furthmüller J., Comput. Mater. Sci., 1996, 6, 15
Perdew J. P., Chevary J. A., Vosko S. H., Jackson K. A., Pederson M. R., Singh D. J., Fiolhais C., Phys. Rev. B, 1992, 46, 6671
Blöchl P. E., Phys. Rev. B, 1994, 50, 17953
Kresse G, Joubert D., Phys. Rev. B, 1999, 59, 1758
Monkhorst H. J., Pack J. D., Phys. Rev. B, 1976, 13, 5188
Henkelman G., Uberuaga B. P., Jonsson H., J. Chem. Phys., 2000, 113, 9901
Huberty J. S., Madix R. J., Surf. Sci., 1996, 360, 144
Desai S. K., Neurock M., Kourtakis K., J. Phys. Chem. B, 2002, 106, 2559
Gazdzicki P., Uvdal P., Jakob P., J. Chem. Phys., 2009, 130, 224703
Gomes J. R. B., Gomes J. A. N. F., J. Mol. Struc.(Theochem.), 2000, 503, 189
Niu C. Y., Jiao J., Xing B., Wang G. C., Bu X. H., J. Comput. Chem., 2010, 31, 2023
Wang G. C., Li J., Xu X. F., J. Comput. Chem., 2005, 26, 871
Wang H. F., Liu Z. P., J. Am. Chem. Soc., 2008, 130, 10996
Lv C. Q., Liu J. H., Guo Y., Wang G. C., Phys. Chem. Chem. Phys., 2012, 14, 6869
Lv C. Q., Liu J. H., Song X. F., Guo Y., Wang G. C., J. Mol. Model., 2014, 20, 2137
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the Key Program of Natural Science of Tianjin, China(No.13JCZDJC26800), the National Natural Science Foundation of China(Nos.21503122, 21346002), the Shanxi Province Science Foundation for Youths, China(No.2014021016-2), the Scientific and Technological Programs in Shanxi Province, China(No.2015031017) and the Foundation of Key Laboratory of Advanced Energy Materials Chemistry of Ministry of Education of China.
Rights and permissions
About this article
Cite this article
Liu, J., LÜ, C., Jin, C. et al. Density functional theoretical studies on the methanol adsorption and decomposition on Ru(0001) surfaces. Chem. Res. Chin. Univ. 32, 234–241 (2016). https://doi.org/10.1007/s40242-016-5416-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-016-5416-z