Abstract
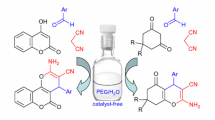
A simple and efficient protocol was established to synthesize γ-nitrocarbonyl compounds via three-component one-pot tandem condensation reaction of aldehydes, Meldrum’s acid and nitroalkanes under solvent-free conditions. The present method shows fascinating features such as using readily available starting materials, promoting the yields by inexpensive and mild organic base triethylamine, omiting the synthesis of Michael acceptor and avoiding the use of organic solvent. This strategy provides an easy alternative synthetic approach to the useful intermediate, γ-nitrocarbonyl compounds, with moderate to good yields.
Similar content being viewed by others
References
Mitchell C. E. T., Brenner S. E., Ley S. V., Chem. Commun., 2005, 41(42), 5346
Mei K., Jin M., Zhang S., Li P., Liu W., Chen X., Xue F., Duan W., Wang W., Org. Lett., 2009, 11(13), 2864
Roca-Lopez D., Sadaba D., Delso I., Herrera R. P., Tejero T., Merino P., Tetrahedron: Asymmetry, 2010, 21(21/22), 2561
Ballini R., Bosica G., Fiorini D., Palmieri A., Petrini M., Chem. Rev., 2005, 105(3), 933
Krishna P. R., Sreeshailam A., Srinivas R., Tetrahedron, 2009, 65(47), 9657
Lia J. H., Li Z. G., Chen Q. G., J. Chem. Res., 2004, 758
Bassas O., Huuskonen J., Rissanen K., KosKinen A. M. P., Eur. J. Org. Chem., 2009, 1340
Didiera D., Sergeyev S., Arkivoc, 2009, (xiv), 124
Kataja A. O., Koskinen A. M. P., Arkivoc, 2010, (ii), 205
Lou H. W., Zhang T., Li K., Zhang M., Li J. H., Chin. J. Org. Chem., 2011, 31(8), 1258
Leyva-Pérez A., García-García P., Corma A., Angew. Chem. Int. Ed., 2014, 53(33), 8687
Schreiber S. L., Science, 2000, 287(5460), 1964
Wen L. R., Li Z. R., Li M., Cao H., Green Chem., 2012, 14(3), 707
Jiang B., Rajale T., Wever W., Tu S. J., Li G. G., Chem. Asian J., 2010, 5(11), 2318
Lin X. F., Mao Z. J., Dai X. X., Lu P., Wang Y. G., Chem. Commun., 2011, 47(23), 6620
Hong D., Zhu Y. X., Li Y., Lin X. F., Lu P., Wang Y. G., Org. Lett., 2011, 13(17), 4668
Ghahremanzadeh R., Rashid Z., Zarnani A. H., Naeimi H., Ultrason. Sonochem., 2014, 21(4), 1451
Toure B. B., Hall D. G., Chem. Rev., 2009, 109(9), 4439
Candeias N. R., Montalbano F., Cal P. M., Gois P. M. P., Chem. Rev., 2010, 110(10), 6169
Ramachary D. B., Jain S., Org. Biomol. Chem., 2011, 9(5), 1277
Bonfield E. R., Li C. J., Adv. Synth. Catal., 2008, 350(3), 370
Elders N., vander Born D., Hendrickx L. J. D., Timmer B. J. J., Krause A., Janssen E., de Kanter F. J. J., Ruijter E., Orru R. V. A., Angew. Chem. Int. Ed., 2009, 48(32), 5856
Brahmachari G., Das S., Tetrahedron Lett., 2012, 53(12), 1479
Deng H., Li Z., Ke F., Zhou X., Chem. Eur. J., 2012, 18(16), 4840
Sunderhaus J. D., Dockendorff C., Martin S. F., Tetrahedron, 2009, 65(33), 6454
Tu X. C., Feng H., Tu M. S., Jiang B., Wang S. L., Tu S. J., Tetrahedron Lett., 2012, 53(25), 3169
Yu J., Shi F., Gong L. Z., Acc. Chem. Res., 2011, 44(11), 1156
Dömling A., Wang W., Wang K., Chem. Rev., 2012, 112(6), 3083
Hajipour A. R., Ghayeb Y., Sheikhan N., Tetrahedron Lett., 2009, 50(40), 5649
Jiang B., Xue L. Y., Wang X. H., Tu M. S., Liu Y. P., Tu S. J., Tetrahedron Lett., 2012, 53(10), 1261
Aditya K., Béla T., Green Chem., 2010, 12(5), 875
Ma N., Jiang B., Zhang G., Tu S. J., Wever W., Li G. G., Green Chem., 2010, 12(8), 1357
Cheng C., Jiang B., Tu S. J., Li G. G., Green Chem., 2011, 13(8), 2107
Martins M. A. P., Frizzo C. P., Moreira D. N., Buriol L., Machado P., Chem. Rev., 2009, 109(9), 4140
Kumar R., Raghuvanshi K., Verma R. K., Singh M. S., Tetrahedron Lett., 2010, 51(45), 5933
Kidwai M., Jahan A., Chauhan R., Mishra N. K., Tetrahedron Lett., 2012, 53(14), 1728
Wen L. R., Li Z. R., Li M., Cao H., Green Chem., 2012, 14(3), 707
Zong C. L., Zeng R. S., Zou J. P., Chem. Res. Chinese Universities, 2014, 30(4), 632
Poe S. L., Kobašlija M., McQuade D. T., J. Am. Chem. Soc., 2006, 128(49), 15586
Poe S. L., Kobašlija M., McQuade D. T., J. Am. Chem. Soc., 2007, 129(29), 9216
Li Y. L., Zhao B., Du B. X., Jiang Q. S., Wang X. S., Cao C. S., Tetrahedron Lett., 2013, 54(3), 227
Gusarova N. K., Volkov P. A., Ivanova N. I., Sukhov B. G., Larina L. I., Kazheva O. N., Alexandrov G. G., D’yachenko O. A., Trofimov B. A., Tetrahedron Lett., 2013, 54(49), 6772
Yavari I., Beheshti S., Hossaini Z., Souri S., Synth. Commun., 2011, 41(6), 907
Wassima G., Raouf B., Abdelmadjid D., Chin. J. Chem., 2012, 30(3), 733
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(No.21403100) and the Doctoral Scientific Research Foundation of Liaoning Province, China(No.20141100).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lü, C., Liu, Y., Zhou, X. et al. One-pot three-component reaction for efficient and facile synthesis of γ-nitrocarbonyl compounds under solvent-free conditions. Chem. Res. Chin. Univ. 31, 208–211 (2015). https://doi.org/10.1007/s40242-015-4410-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-015-4410-1