Abstract
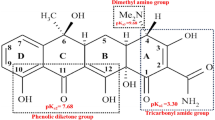
Microcystin-Leucine Arginine (MC-LR) is one of the most studied cyanotoxins due to its toxicity and abundant that cause health hazards for humans through of the drinking water. In this study, BiVO4/TiO2 nanocomposite was synthesized by hydrothermal method and employed for the removal of MC-LR. The characteristics of the catalysts were determined by FESEM, XRD and FTIR spectra. Response surface methodology (RSM) was applied to assess the effects of operating variables (pH, contact time, and catalyst dose) on the MC-LR removal. The coefficient of determination (R2) was calculated 98.7% for the response. The residual concentration of MC-LR was measured by high-performance liquid chromatography (HPLC). The results show that the highest removal efficiency of MC-LR was 98% under the optimum conditions (pH = 5, contact time = 90 min, and catalyst dose = 0.5 g/l). MC-LR decomposition efficiency by BiVO4/TiO2 nanocomposite was enhanced by pH reduction and increasing of contact time and catalyst dose. The prepared BiVO4/TiO2 nanocomposite with technological potential can be used directly in environmental preservation, specifically in the decontamination of MC-LR from aqueous solutions.






Similar content being viewed by others
References
Liu L, Huang Q, Qin B. Characteristics and roles of Microcystis extracellular polymeric substances (EPS) in cyanobacterial blooms: a short review. J Freshw Ecol. 2018;33(1):183–93.
Shamsollahi HR, Alimohammadi M, Nabizadeh R, Nazmara S, Mahvi AH. Monitoring of microcystin-LR concentration in water reservoir. Desalin Water Treat. 2018;126:345–9.
Sharma VK, Chen L, Marsalek B, Zboril R, O'Shea KE, Dionysiou DD. Iron based sustainable greener technologies to treat cyanobacteria and microcystin-LR in water. Water Sci Tech-W Sup. 2016;17(1):107–14.
Sorlini S, Gialdini F, Collivignarelli C. Removal of cyanobacterial cells and microcystin-LR from drinking water using a hollow fiber microfiltration pilot plant. Desalination. 2013;309:106–12.
Jaša L, Sadílek J, Kohoutek J, Straková L, Maršálek B, Babica P. Application of passive sampling for sensitive time-integrative monitoring of cyanobacterial toxins microcystins in drinking water treatment plants. Water Res. 2019;153:108–20.
Campos A, Vasconcelos V. Molecular mechanisms of microcystin toxicity in animal cells. Int J Mol Sci. 2010;11(1):268–87.
Shamsollahi HR, Alimohammadi M, Nabizadeh R, Nazmara S, Mahvi AH. Measurement of microcystin-LR in water samples using improved HPLC method. Glob J Health Sci. 2015;7(2):66.
Janssen EM-L. Cyanobacterial peptides beyond microcystins–a review on co-occurrence, toxicity, and challenges for risk assessment. Water Res. 2019;151:488–99.
Antoniou MG, De La Cruz AA, Dionysiou DD. Cyanotoxins: New generation of water contaminants. J Environ Eng-ASCE. 2005;131:9.
Yan H, Gong A, He H, Zhou J, Wei Y, Lv L. Adsorption of microcystins by carbon nanotubes. Chemosphere. 2006;62(1):142–148.11.
Liu B, Qu F, Chen W, Liang H, Wang T, Cheng X, et al. Microcystis aeruginosa-laden water treatment using enhanced coagulation by persulfate/Fe (II), ozone and permanganate: comparison of the simultaneous and successive oxidant dosing strategy. Water Res. 2017;125:72–80.
Mokoena M, Mukhola M, Okonkwo O. Hazard assessment of microcystins from the household’s drinking water. Appl Ecol Env Res. 2016;14(3):695–710.13.
Pham T-L, Dang TN. Microcystins in freshwater ecosystems: occurrence, distribution, and current treatment approaches. Water and Wastewater Treatment Technologies: Springer; 2019: 15–36.
Roegner AF, Brena B, González-Sapienza G, Puschner B. Microcystins in potable surface waters: toxic effects and removal strategies. J Appl Toxicol. 2014;34(5):441–57.
Fan J, Ho L, Hobson P, Brookes J. Evaluating the effectiveness of copper sulphate, chlorine, potassium permanganate, hydrogen peroxide and ozone on cyanobacterial cell integrity. Water Res. 2013;47(14):5153–64.
Fan W, Zhang Q, Wang Y. Semiconductor-based nanocomposites for photocatalytic H2 production and CO2 conversion. Phys Chem Chem Phys. 2013;15(8):2632–49.
Pantelić D, Svirčev Z, Simeunović J, Vidović M, Trajković I. Cyanotoxins: characteristics, production and degradation routes in drinking water treatment with reference to the situation in Serbia. Chemosphere. 2013;91(4):421–41.
Liu Y, Ren J, Wang X, Fan Z. Mechanism and reaction pathways for microcystin-LR degradation through UV/H2O2 treatment. PLoS One. 2016;11(6):e0156236.
Karci A, Wurtzler EM, Armah A, Wendell D, Dionysiou DD. Solar photo-Fenton treatment of microcystin-LR in aqueous environment: transformation products and toxicity in different water matrices. J Hazard Mater. 2018;349:282–92.
Park J-A, Yang B, Park C, Choi J-W, van Genuchten CM, Lee S-H. Oxidation of microcystin-LR by the Fenton process: kinetics, degradation intermediates, water quality and toxicity assessment. Chem Eng. 2017;309:339–48.
Pinho LX, Azevedo J, Brito Â, Santos A, Tamagnini P, Vilar VJ, et al. Effect of TiO2 photocatalysis on the destruction of Microcystis aeruginosa cells and degradation of cyanotoxins microcystin-LR and cylindrospermopsin. Chem Eng. 2015;268:144–52.
Miao H-F, Qin F, Tao G-J, Tao W-Y, Ruan W-Q. Detoxification and degradation of microcystin-LR and-RR by ozonation. Chemosphere. 2010;79(4):355–61.
Song W, De La Cruz AA, Rein K, O'Shea KE. Ultrasonically induced degradation of microcystin-LR and-RR: identification of products, effect of pH, formation and destruction of peroxides. Environ. Sci. Technol. 2006;40(12):3941–6.
Zong W, Sun F, Sun X. Oxidation by-products formation of microcystin-LR exposed to UV/H2O2: toward the generative mechanism and biological toxicity. Water Res. 2013;47(9):3211–9.
Kabra K, Chaudhary R, Sawhney RL. Treatment of hazardous organic and inorganic compounds through aqueous-phase photocatalysis: a review. Ind Eng Chem Res. 2004;43(24):7683–96.
Baiju K, Shukla S, Sandhya K, James J, Warrier K. Photocatalytic activity of sol− gel-derived nanocrystalline titania. J Phys Chem C. 2007;111(21):7612–22.
Choi H, Sofranko AC, Dionysiou DD. Nanocrystalline TiO2 photocatalytic membranes with a hierarchical mesoporous multilayer structure: synthesis, characterization, and multifunction. Adv Funct Mater. 2006;16(8):1067–74.
Hirakawa T, Sato K, Komano A, Kishi S, Nishimoto CK, Mera N, et al. Experimental study on adsorption and photocatalytic decomposition of isopropyl methylphosphonofluoridate at surface of TiO2 photocatalyst. J Phys Chem C. 2010;114(5):2305–14.
Pal S, Laera AM, Licciulli A, Catalano M, Taurino A. Biphase TiO2 microspheres with enhanced photocatalytic activity. Ind Eng Chem Res. 2014;53(19):7931–8.
Golmojdeh H, Zanjanchi M. A facile approach for synthesis of BiVO4 nanoparticles possessing high surface area and various morphologies. Cryst Res Technol. 2012;47(9):1014–25.
Kumar SG, Devi LG. Review on modified TiO2 photocatalysis under UV/visible light: selected results and related mechanisms on interfacial charge carrier transfer dynamics. J Phys Chem A. 2011;115(46):13211–41.
Sun J, Li X, Zhao Q, Ke J, Zhang D. Novel V2O5/BiVO4/TiO2 nanocomposites with high visible-light-induced photocatalytic activity for the degradation of toluene. J Phys Chem C. 2014;118(19):10113–21.
Seabold JA, Choi K-S. Efficient and stable photo-oxidation of water by a bismuth vanadate photoanode coupled with an iron oxyhydroxide oxygen evolution catalyst. J Am Chem Soc. 2012;134(4):2186–92.
Zhou D, Pang L-X, Guo J, Wang H, Yao X, Randall C. Phase evolution, phase transition, Raman spectra, infrared spectra, and microwave dielectric properties of low temperature firing (K0.5xBi1–0.5x)(MoxV1–x)O4 ceramics with Scheelite related structure. Inorg Chem. 2011;50(24):12733–8.
Wetchakun N, Chainet S, Phanichphant S, Wetchakun K. Efficient photocatalytic degradation of methylene blue over BiVO4/TiO2 nanocomposites. Ceram Int. 2015;41(4):5999–6004.
Aslani H, Nabizadeh R, Nasseri S, Mesdaghinia A, Alimohammadi M, Mahvi AH, et al. Application of response surface methodology for modeling and optimization of trichloroacetic acid and turbidity removal using potassium ferrate (VI). Desalin Water Treat. 2016;57(52):25317–28.
Bussi J, Ohanian M, Vázquez M, Dalchiele EA. Photocatalytic removal of hg from solid wastes of chlor-alkali plant. J Environ Eng. 2002;128(8):733–739.38.
Huang H, Xiao K, He Y, Zhang T, Dong F, Du X, et al. In situ assembly of BiOI@ Bi12O17Cl2 pn junction: charge induced unique front-lateral surfaces coupling heterostructure with high exposure of BiOI {001} active facets for robust and nonselective photocatalysis. Appl Catal B-Environ. 2016;199:75–86.
Huang H, He Y, Li X, Li M, Zeng C, Dong F, et al. Bi2O2(OH)(NO3) as a desirable [Bi2O2]2+ layered photocatalyst: strong intrinsic polarity, rational band structure and {001} active facets co-beneficial for robust photooxidation capability. J Mater Chem A. 2015;3(48):24547–56.
Huang H, Li X, Wang J, Dong F, Chu PK, Zhang T, et al. Anionic group self-doping as a promising strategy: band-gap engineering and multi-functional applications of high-performance CO32–doped Bi2O2CO3. ACS Catal. 2015;5(7):4094–103.
Rajalingam V. Synthesis and characterization of BiVO4 nanostructured materials: application to photocatalysis: Université du Maine; 2014.
Matthiensen A, Beattie KA, Yunes JS, Kaya K, Codd GA. [D-Leu1] microcystin-LR, from the cyanobacterium Microcystis RST 9501 and from a Microcystis bloom in the Patos lagoon estuary, Brazil. Phytochemistry. 2000;55(5):383–7.
Hu Y, Chen W, Fu J, Ba M, Sun F, Zhang P, et al. Hydrothermal synthesis of BiVO4/TiO2 composites and their application for degradation of gaseous benzene under visible light irradiation. Appl Surf Sci. 2018;436:319–26.
Rahimi B, Ebrahimi A. Photocatalytic process for total arsenic removal using an innovative BiVO4/TiO2/LED system from aqueous solution: optimization by response surface methodology (RSM). J Taiwan Inst Chem E. 2019;101:64–79.
Mohammadi A, Nemati S, Mosaferi M, Abdollahnejhad A, Almasian M, Sheikhmohammadi A. Predicting the capability of carboxymethyl cellulose-stabilized iron nanoparticles for the remediation of arsenite from water using the response surface methodology (RSM) model: modeling and optimization. J Contam Hydrol. 2017;203:85–92.
Venkatachalam N, Palanichamy M, Murugesan V. Sol–gel preparation and characterization of alkaline earth metal doped nano TiO2: efficient photocatalytic degradation of 4-chlorophenol. J Mol Catal A-Chem. 2007;273(1–2):177–85.
Nasseri S, Borna MO, Esrafili A, Kalantary RR, Kakavandi B, Sillanpää M, et al. Photocatalytic degradation of malathion using Zn2+−doped TiO2 nanoparticles: statistical analysis and optimization of operating parameters. Appl Phys A Mater Sci Process. 2018;124(2):175.
Bhatkhande DS, Pangarkar VG, Beenackers AA. Photocatalytic degradation for environmental applications–a review. J ChemTechnol Biotechnol. 2002;77(1):102–16.
He X, Pelaez M, Westrick JA, O’Shea KE, Hiskia A, Triantis T, et al. Efficient removal of microcystin-LR by UV-C/H2O2 in synthetic and natural water samples. Water Res. 2012;46(5):1501–10.
Mortazavian S, Saber A, James DE. Optimization of Photocatalytic degradation of acid blue 113 and acid red 88 textile dyes in a UV-C/TiO2 suspension system: application of response surface methodology (RSM). Catalysts. 2019;9(4):360.51.
Rahimi B, Jafari N, Abdolahnejad A, Farrokhzadeh H, Ebrahimi A. Application of efficient photocatalytic process using a novel BiVO/TiO2-NaY zeolite composite for removal of acid orange 10 dye in aqueous solutions: modeling by response surface methodology (RSM). J Environ Chem Eng. 2019;7(4):103253.
Huang W-J, Cheng B-L, Cheng Y-L. Adsorption of microcystin-LR by three types of activated carbon. J Hazard Mater. 2007;141(1):115–22.
Lawton LA, Robertson PK, Cornish BJ, Marr IL, Jaspars M. Processes influencing surface interaction and photocatalytic destruction of microcystins on titanium dioxide photocatalysts. J Catal. 2003;213(1):109–13.
Chen P, Zhu L, Fang S, Wang C, Shan G. Photocatalytic degradation efficiency and mechanism of microcystin-RR by mesoporous Bi2WO6 under near ultraviolet light. Environ Sci Technol. 2012;46(4):2345–51.
Zhang H, Zhu G, Jia X, Ding Y, Zhang M, Gao Q, et al. Removal of microcystin-LR from drinking water using a bamboo-based charcoal adsorbent modified with chitosan. J Environ Sci. 2011;23(12):1983–8.
Liu S, Hu X, Jiang W, Ma L, Cai M, Xu H, et al. Degradation of microcystins from Microcystis aeruginosa by 185-nm UV irradiation. Water Air Soil Pollut. 2016;227(4):129.
Pavithra KG, Kumar PS, Christopher FC, Saravanan A. Removal of toxic Cr (VI) ions from tannery industrial wastewater using a newly designed three-phase three-dimensional electrode reactor. J Phys Chem Solids. 2017;110:379–85.
Huang H, He Y, Du X, Chu PK, Zhang Y. A general and facile approach to heterostructured core/shell BiVO4/BiOI p–n junction: room-temperature in situ assembly and highly boosted visible-light photocatalysis. ACS Sustain Chem Eng. 2015;3(12):3262–73.
Huang H, Xiao K, Zhang T, Dong F, Zhang Y. Rational design on 3D hierarchical bismuth oxyiodides via in situ self-template phase transformation and phase-junction construction for optimizing photocatalysis against diverse contaminants. Appl Catal B-Environ. 2017;203:879–88.
Acknowledgements
This study is a part of a Ph.D. approved research Thesis (No. 395847 and Code of ethics. IR.MUI.REC.1395.3.847) performed at Isfahan University of Medical Sciences, Iran. The authors are thankful for the funding provided by the Department of Environmental Health Engineering and Environment Research Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare in this study has no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jafari, N., Ebrahimpour, K., Abdolahnejad, A. et al. Efficient degradation of microcystin-LR by BiVO4/TiO2 photocatalytic nanocomposite under visible light. J Environ Health Sci Engineer 17, 1171–1183 (2019). https://doi.org/10.1007/s40201-019-00432-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40201-019-00432-4