Abstract
Background
Obese human and mice were reported to have higher circularity endotoxin (LPS) levels as compared to their lean counter parts. The current study was aimed to reveal the molecular mechanisms underlying the LPS mediated induction of CCL2 in human monocytes/macrophages.
Methods
Human monocytic cell line THP-1, THP-1 cells derived macrophages and primary macrophages were treated with LPS and TNF-α (positive control). CCL2 expression was determined with real-time RT-PCR and ELISA. THP-1-XBlue™ cells, THP-1-XBlue™-defMyD cells, TLR4 neutralization antibody, TLR4 siRNA and inhibitors for NF-kB and MAPK were used to study the signaling pathways. Phosphorylation of NF-kB and c-Jun was analyzed by ELISA.
Results
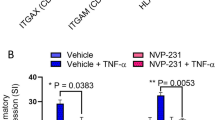
LPS upregulates CCL2 expression at both mRNA (THP-1: 23.40 ± .071 Fold, P < 0.0001; THP-1-derived macrophages: 103 ± 0.56 Fold, < 0.0001; Primary macrophages: 48 ± 1.41 Fold, P < 0.0005) and protein (THP1 monocytes:1048 ± 5.67 pg/ml, P < 0.0001; THP-1-derived macrophages; 2014 ± 2.12, P = 0.0001; Primary macrophages: 859.5 ± 3.54, P < 0.0001) levels in human monocytic cells/macrophages. Neutralization of TLR4 blocked LPS-induced CCL-2 secretion (P < 0.0001). Silencing of TLR4 by siRNA also significantly reduced LPS-induced CCL-2 production. Furthermore, MyD88-Knockout cells treated with LPS did not produce CCL-2. NF-kB and c-Jun phosphorylation was noted in LPS treated cells.
Conclusion
Overall, our data reveal that LPS induces CCL-2 in monocytes/macrophages via TLR4/MyD88 signaling which leads to the activation of NF-kB/AP-1 transcription factors.




Similar content being viewed by others
Abbreviations
- AP-1:
-
Activating protein-1
- ATCC:
-
American Type Culture Collection
- CCL-2:
-
Chemokine (C-C motif) ligand −2
- ELISA:
-
Enzyme-linked immunosorbent assay
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- LPS:
-
Lipopolysaccharide
- MAPK:
-
Mitogen-activated protein kinase
- MyD88:
-
Myeloid differentiation factor 88
- NF-κB:
-
Nuclear factor-kappaB
- PBMC:
-
Peripheral blood mononuclear cells
- PCR:
-
Polymerase chain reaction
- SEAP:
-
Secreted embryonic alkaline phosphatase
- THP-1:
-
A human monocytic cell line
- TLRs:
-
Toll-like receptors
- TNF-α:
-
Tumor necrosis factor-alpha
References
Ferrier L, Berard F, Debrauwer L, Chabo C, Langella P, Bueno L, et al. Impairment of the intestinal barrier by ethanol involves enteric microflora and mast cell activation in rodents. Am J Pathol. 2006;168:1148–54.
Bannerman DD, Goldblum SE. Direct effects of endotoxin on the endothelium: barrier function and injury. Lab Investig. 1999;79:1181–99.
Cani PD, Amar J, Iglesias MA, Poggi M, Knauf C, Bastelica D, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761–72.
Lu YC, Yeh WC, Ohashi PS. LPS/TLR4 signal transduction pathway. Cytokine. 2008;42:145–51.
Frost RA, Nystrom GJ, Lang CH. Lipopolysaccharide regulates proinflammatory cytokine expression in mouse myoblasts and skeletal muscle. Am J Phys Regul Integr Comp Phys. 2002;283:R698–709.
Gustot T, Lemmers A, Moreno C, Nagy N, Quertinmont E, Nicaise C, et al. Differential liver sensitization to toll-like receptor pathways in mice with alcoholic fatty liver. Hepatology. 2006;43:989–1000.
Song MJ, Kim KH, Yoon JM, Kim JB. Activation of toll-like receptor 4 is associated with insulin resistance in adipocytes. Biochem Biophys Res Commun. 2006Mori M;346:739–45.
Mita Y, Dobashi K, Endou K, Kawata T, Shimizu Y, Nakazawa T. Toll-like receptor 4 surface expression on human monocytes and B cells is modulated by IL-2 and IL-4. Immunol Lett. 2002;81:71–5.
Ahmad R, Al-Mass A, Atizado V, Al-Hubail A, Al-Ghimlas F, Al-Arouj M, et al. Elevated expression of the toll like receptors 2 and 4 in obese individuals: its significance for obesity-induced inflammation. J Inflamm (Lond). 2012;9:48.
Al-Rashed F, Kochumon S, Usmani S, Sindhu S, Ahmad R. Pam3CSK4 induces MMP-9 expression in human Monocytic THP-1 cells. Cell Physiol Biochem. 2017;41:1993–2003.
Sindhu S, Al-Roub A, Koshy M, Thomas R, Ahmad R. Palmitate-induced MMP-9 expression in the human Monocytic cells is mediated through the TLR4-MyD88 dependent mechanism. Cell Physiol Biochem. 2016;39:889–900.
Shihab PK, Al-Roub A, Al-Ghanim M, Al-Mass A, Behbehani K, Ahmad R. TLR2 and AP-1/NF-kappaB are involved in the regulation of MMP-9 elicited by heat killed listeria monocytogenes in human monocytic THP-1 cells. J Inflamm (Lond). 2015;12:32.
Ahmad R, Shihab PK, Jasem S, Behbehani K. FSL-1 induces MMP-9 production through TLR-2 and NF-kappaB /AP-1 signaling pathways in monocytic THP-1 cells. Cell Physiol Biochem. 2014;34:929–42.
Ahmad R, El Bassam S, Cordeiro P, Menezes J. Requirement of TLR2-mediated signaling for the induction of IL-15 gene expression in human monocytic cells by HSV-1. Blood. 2008;112:2360–8.
Matsushima K, Larsen CG, DuBois GC, Oppenheim JJ. Purification and characterization of a novel monocyte chemotactic and activating factor produced by a human myelomonocytic cell line. J Exp Med. 1989;169:1485–90.
Dordevic AL, Konstantopoulos N, Cameron-Smith D. 3T3-L1 preadipocytes exhibit heightened monocyte-chemoattractant protein-1 response to acute fatty acid exposure. PLoS One. 2014;9:e99382.
Kanda H, Tateya S, Tamori Y, Kotani K, Hiasa K, Kitazawa R, et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J Clin Invest. 2006;116:1494–505.
Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes. 2008;57:1470–81.
Kamei N, Tobe K, Suzuki R, Ohsugi M, Watanabe T, Kubota N, et al. Overexpression of monocyte chemoattractant protein-1 in adipose tissues causes macrophage recruitment and insulin resistance. J Biol Chem. 2006;281:26602–14.
Lassenius MI, Pietilainen KH, Kaartinen K, Pussinen PJ, Syrjanen J, Forsblom C, et al. Bacterial endotoxin activity in human serum is associated with dyslipidemia, insulin resistance, obesity, and chronic inflammation. Diabetes Care. 2011;34:1809–15.
Cho JS, Kang JH, Um JY, Han IH, Park IH, Lee HM. Lipopolysaccharide induces pro-inflammatory cytokines and MMP production via TLR4 in nasal polyp-derived fibroblast and organ culture. PLoS One. 2014;9:e90683.
Harris JF, Aden J, Lyons CR, Tesfaigzi Y. Resolution of LPS-induced airway inflammation and goblet cell hyperplasia is independent of IL-18. Respir Res. 2007;8:24.
Acknowledgments
This study was financially supported by Kuwait Foundation for the Advancement of Sciences (KFAS). Grant # RC14016001 (RA-AM-2017-007).
Funding
This study was supported by funds from Kuwait Foundation for Advancement of Sciences (KFAS), Grant # RC14016001 (RA-AM-2017-007).
Author information
Authors and Affiliations
Contributions
NA performed experiments, analyzed the data and participated in writing the paper. AW, SS, and SK participated in performing experiments. AH contributed in scientific discussions and critically reviewed/edited the manuscript. SA participated in arranging data and preparing graphs. JT critically reviewed and commented the manuscript. SS participated in designing experiments. RA conceived the idea, designed the experiments, analyzed the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Akhter, N., Hasan, A., Shenouda, S. et al. TLR4/MyD88 -mediated CCL2 production by lipopolysaccharide (endotoxin): Implications for metabolic inflammation. J Diabetes Metab Disord 17, 77–84 (2018). https://doi.org/10.1007/s40200-018-0341-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-018-0341-y