Abstract
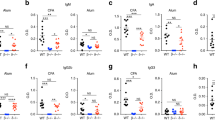
CD4+ T cell subsets including regulatory T cells (Tregs), Th1 and Th17 are critical for control and development of inflammation and autoimmunity. We investigated the in vitro and in vivo effects of silymarin, a well-known herbal medicine on differentiation and function of Tregs and Th1 and Th17 responses. For in vitro study, mice splenocytes treated with 20–30 μg/ml silymarin were evaluated for gene expressions of specific transcription factors and cytokines of CD4+ T cell subsets using real-time PCR. Induction of Treg cell development in the presence of silymarin was performed on isolated naïve CD4+ T cells. Effect of silymarin-induced Tregs on T cell suppression was determined by CFSE labeling method. Results of this part showed that silymarin significantly decreased IFNγ, RORγt and IL-17 gene expressions and upregulated Foxp3, TGF-β and IL-10 mRNA. More silymarin-enhanced naïve CD4+ T cells differentiated to Tregs (67%) than the control (47%). Silymarin-induced Tregs reduced proliferation of naïve activated T cells (<50%). For in vivo study, mice were immunized with ovalbumin (Ova) on days 1 and 14. Silymarin (100 mg/Kg) was intraperitoneally administered two days before the first Ova challenge followed by on every day for two weeks. Splenocytes were then isolated for assessment of CD4+ T cell subsets and ex vivo analysis using flow cytometry. Treatment of Ova-immunized mice with silymarin increased Tregs (11.24 ± 1.2%, p < 0.01(but decreased Th1 (1.72 ± 0.4%, p < 0.001) and Th17 (1.07 ± 0.04%, p < 0.001) cells. Ex vivo Ova challenge of splenocytes from Ova-immunized mice treated with silymarin decreased proliferation of splenocytes, IFNγ (2.76% of control) and IL-17 (<8%) along with increased TGF-β (59.7%) expressions in CD4+T-bet+, CD4+RORγt+ and CD4+Foxp3+ T cells, respectively. In conclusion, silymarin promoted Treg differentiation and function and decreased Th1 and Th17 cells. Silymarin may differentially regulate CD4+ T cell responses which can provide potential benefits for its use as treatment of immune-related diseases.

ᅟ







Similar content being viewed by others
Data availability
Datasets analyzed during the current study available from the corresponding author on reasonable request.
References
Raphael I, Nalawade S, Eagar TN, Forsthuber TG. T cell subsets and their signature cytokines in autoimmune and inflammatory diseases. Cytokine. 2015;74(1):5–17.
Huber S, Gagliani N, O'Connor W Jr, Geginat J, Caprioli F. CD4(+) T Helper Cell Plasticity in Infection, Inflammation, and Autoimmunity. Mediat Inflamm. 2017;2017:7083153. https://doi.org/10.1155/2017/7083153.
Rezaei N, Amirghofran Z, Nikseresht A, Ashjazade N, Zoghi S, Tahvili S, et al. In Vitro effects of sodium benzoate on Th1/Th2 deviation in patients with multiple sclerosis. Immunol Investig. 2016;45(7):679–91. https://doi.org/10.1080/08820139.2016.1208216.
Namdari H, Izad M, Amirghofran Z. Modulation of CD4+ T cell subsets by Euphorbia microciadia and Euphorbia osyridea plant extracts.IJI . 2017;14(2):134–50.
Krebs CF, Schmidt T, Riedel JH, Panzer U. T helper type 17 cells in immune-mediated glomerular disease. Nat Rev Nephrol. 2017;13(10):647–59. https://doi.org/10.1038/nrneph.2017.112.
Miossec P, Korn T, Kuchroo VK. Interleukin-17 and type 17 helper T cells. N Engl J Med. 2009;361(9):888–98.
McGovern JL, Wright GP, Stauss HJ. Engineering specificity and function of therapeutic regulatory T cells. Front Immunol. 2017;8:1517. https://doi.org/10.3389/fimmu.2017.01517.
Li J, Dong X, Zhao L, Wang X, Wang Y, Yang X, et al. Natural killer cells regulate Th1/Treg and Th17/Treg balance in chlamydial lung infection. J Cell Mol Med. 2016;20(7):1339–51. https://doi.org/10.1111/jcmm.12821.
Lee GR. The balance of Th17 versus Treg cells in autoimmunity. Int J Mol Sci. 2018;19(3):730.
Lee W, Lee GR. Transcriptional regulation and development of regulatory T cells. Exp Mol Med. 2018;50(3):e456.
Schmidt A, Eriksson M, Shang MM, Weyd H, Tegner J. Comparative analysis of protocols to induce human CD4+Foxp3+ regulatory T cells by combinations of IL-2, TGF-beta, retinoic acid, rapamycin and butyrate. PLoS One. 2016;11(2):e0148474. https://doi.org/10.1371/journal.pone.0148474.
Mohr A, Malhotra R, Mayer G, Gorochov G, Miyara M. Human FOXP 3+ T regulatory cell heterogeneity. Clin Transl Immunology. 2018;7(1):e1005.
Karimi G, Vahabzadeh M, Lari P, Rashedinia M, Moshiri M. "Silymarin", a promising pharmacological agent for treatment of diseases.IJBMS . 2011;14(4):308–17.
Hellerbrand C, Schattenberg JM, Peterburs P, Lechner A, Brignoli R. The potential of silymarin for the treatment of hepatic disorders. Clin Phytosci. 2017;2(1):7.
Federico A, Dallio M, Loguercio C. Silymarin/silybin and chronic liver disease: a marriage of many years. Molecules. 2017;22(2):191.
Ferenci P. Silymarin in the treatment of liver diseases: what is the clinical evidence? CLD. 2016;7(1):8–10.
Gharagozloo M, Amirghofran Z. Effects of silymarin on the spontaneous proliferation and cell cycle of human peripheral blood leukemia T cells. J Cancer Res Clin Oncol. 2007;133(8):525–32. https://doi.org/10.1007/s00432-007-0197-x.
Tamayo C, Diamond S. Review of clinical trials evaluating safety and efficacy of milk thistle (Silybum marianum [L.] Gaertn.). Integrative Cancer Therapies. 2007;6(2):146–57.
Morishima C, Shuhart MC, Wang CC, Paschal DM, Apodaca MC, Liu Y et al. Silymarin inhibits in vitro T-cell proliferation and cytokine production in hepatitis C virus infection. Gastroenterology. 2010;138(2):671–81, 81 e1–2. https://doi.org/10.1053/j.gastro.2009.09.021.
Polyak SJ, Morishima C, Shuhart MC, Wang CC, Liu Y, Lee DYW. Inhibition of T-cell inflammatory cytokines, hepatocyte NF-κB signaling, and HCV infection by standardized silymarin. Gastroenterology. 2007;132(5):1925–36.
Schümann J, Prockl J, Kiemer AK, Vollmar AM, Bang R, Tiegs G. Silibinin protects mice from T cell-dependent liver injury☆. J Hepatol. 2003;39(3):333–40.
Almasi E, Gharagozloo M, Eskandari N, Almasi A, Sabzghabaee AM. Inhibition of Apoptosis and Proliferation in T Cells by Immunosuppressive Silymarine. Iranian Journal of Allergy, Asthma and Immunology. 2017;16(2):107.
Gharagozloo M, Javid EN, Rezaei A, Mousavizadeh K. Silymarin inhibits cell cycle progression and mTOR activity in activated human T cells: therapeutic implications for autoimmune diseases. Basic Clin Pharmacol Toxicol. 2013;112(4):251–6. https://doi.org/10.1111/bcpt.12032.
Gharagozloo M, Karimi M, Amirghofran Z. Immunomodulatory effects of silymarin in patients with beta-thalassemia major. Int Immunopharmacol. 2013;16(2):243–7. https://doi.org/10.1016/j.intimp.2013.04.016.
Riccardi C, Nicoletti I. Analysis of apoptosis by propidium iodide staining and flow cytometry. Nat Protoc. 2006;1(3):1458–61. https://doi.org/10.1038/nprot.2006.238.
Long AE, Tatum M, Mikacenic C, Buckner JH. A novel and rapid method to quantify Treg mediated suppression of CD4 T cells. J Immunol Methods. 2017;449:15–22.
Gharagozloo M, Velardi E, Bruscoli S, Agostini M, Di Sante M, Donato V, et al. Silymarin suppress CD4+ T cell activation and proliferation: effects on NF-κB activity and IL-2 production. Pharmacol Res. 2010;61(5):405–9.
Kuwabara T, Ishikawa F, Kondo M, Kakiuchi T. The role of IL-17 and related cytokines in inflammatory autoimmune diseases. Mediat Inflamm. 2017;2017:1–11. https://doi.org/10.1155/2017/3908061.
Adeyemo O, Doi H, Rajender Reddy K, Kaplan DE. Impact of oral silymarin on virus- and non-virus-specific T-cell responses in chronic hepatitis C infection. J Viral Hepat. 2013;20(7):453–62. https://doi.org/10.1111/jvh.12050.
Lovelace ES, Maurice NJ, Miller HW, Slichter CK, Harrington R, Magaret A, et al. Silymarin suppresses basal and stimulus-induced activation, exhaustion, differentiation, and inflammatory markers in primary human immune cells. PLoS One. 2017;12(2):e0171139. https://doi.org/10.1371/journal.pone.0171139.
Dardalhon V, Korn T, Kuchroo VK, Anderson AC. Role of Th1 and Th17 cells in organ-specific autoimmunity. J Autoimmun. 2008;31(3):252–6. https://doi.org/10.1016/j.jaut.2008.04.017.
Sonar SA, Lal G. Differentiation and transmigration of CD4 T cells in Neuroinflammation and autoimmunity. Front Immunol. 2017;8:1695. https://doi.org/10.3389/fimmu.2017.01695.
Arellano B, Graber DJ, Sentman CL. Regulatory T cell-based therapies for autoimmunity. Discov Med. 2016;22(119):73–80.
Acknowledgments
We would like to express our specific thanks to the Deputy of Research Affairs of Shiraz University of Medical Sciences (grant no. 7611) for financial support. This work was extracted from thesis written by one of the authors H. Namdari.
Author information
Authors and Affiliations
Contributions
HN and ZA wrote this manuscript, designed this study and analyzed the data; HN performed the experiments. MI and FR help in flow cytometry analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent for publication
All authors agree to publish our manuscript.
Competing interests
The authors declare that they have no conflict of interest in this work.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. All protocols for animal care and treatment were approved by the local ethics committee.
Electronic supplementary material
ESM 1
(DOCX 1091 kb)
Rights and permissions
About this article
Cite this article
Namdari, H., Izad, M., Rezaei, F. et al. Differential regulation of CD4+ T cell subsets by Silymarin in vitro and in ovalbumin immunized mice. DARU J Pharm Sci 26, 215–227 (2018). https://doi.org/10.1007/s40199-018-0229-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-018-0229-x