Abstract
Introduction
To determine any significant associations between abdominal aortic calcification (AAC) and clinical manifestations of pain symptoms following endoscopic lumbar discectomy.
Methods
Patients sequentially presenting with a history of unilateral radiculopathy with or without back pain, and with magnetic resonance imaging (MRI)-confirmed diagnosis of posterolateral disc herniation between August 2021 and December 2023 were eligible for inclusion in the study. All patients underwent endoscopic discectomy at our center during the study period. AAC stages were classified based on the Kauppila classification system using lateral lumbar radiographs.
Results
Between August 2021 and December 2023 a total of 120 patients were included in the study for analysis, of whom 82 (68.3%) exhibited mild AAC and 38 (31.7%) had moderate-severe AAC. Analyses using the multivariate linear regression model revealed a significant correlation between AAC comorbidity and postoperative clinical outcomes. At 1 year post-surgery, the mean change in leg pain following endoscopic lumbar discectomy was significantly less for patients with moderate-severe AAC (5.0 points) than for patients with mild ACC (p < 0.001). Even after adjusting for relevant confounders, this difference remained significant. Similar results were also observed in the postoperative improvement of back pain, the Oswestry Disability Index score, and the physical functioning, role physical, and bodily pain components of the 36-item Short Form Health Survey questionnaire. There was no significant difference in the rate of repeat surgery or post-surgical new-onset back pain between patients with different levels of severity of AAC at 1 year post-surgery.
Conclusions
There is a significant association between the severity of AAC and clinical outcomes among patients with lumbar disk herniation who underwent endoscopic lumbar discectomy. AAC may serve as a prognostic factor in predicting surgical outcomes and guiding management strategies for patients with lumbar disk herniation following endoscopic lumbar discectomy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Abdominal aortic calcification (AAC) is common in elderly patients, and emerging studies suggest that AAC can have broader implications for musculoskeletal disorders, especially in cases of degenerative spinal diseases |
Lumbar disc herniation is the most common cause of lower back and leg pain, and surgical interventions, such as intervertebral discectomy, to alleviate symptoms and restore nerve function is often needed |
We hypothesized that AAC will influence the clinical outcomes in patients with lumbar disk herniation who undergo endoscopic lumbar discectomy |
What was learned from the study? |
At 1 year post surgery, patients with moderate-severe AAC showed less improvement in leg pain, back pain, Oswestry Disability Index) score, and the physical functioning, role-physical), and bodily pain components of the 36-item Short Form Health Survey questionnaire than patients with mild AAC |
There is a significant association between the severity of AAC and clinical outcomes after endoscopic lumbar discectomy |
Introduction
Abdominal aortic calcification (AAC) is a common manifestation of arterial hardening and a strong indicator of fatal systemic vascular disorders and mortality. In those individuals with AAC, the aortic media along the elastic membrane gradually becomes calcified, leading to atherosclerosis and vascular aging symptoms [1]. Studies have shown that the onset and severity of AAC are the leading cause of higher rates of cardiovascular mortality and morbidity [2, 3]. Interestingly, emerging studies suggest that AAC can have broader implications for musculoskeletal disorders, especially in cases of degenerative spinal diseases and vertebral fracture, in addition to its pathogenic contribution to cardiovascular malfunction [4,5,6]. It has been speculated that prolonged deposition of calcium salts on the aortic walls causes arterial hardening, resulting in abnormally high systolic and low diastolic pressure. This altered heart pressure damages arterial blood vessels during excessive systolic contraction, with consequent failure to adequately supply blood to various organs and tissues due to over-relaxation during diastole. Moreover, the presence of calcified plaques in the aorta notably narrow down the lumbar arteries, causing reduced blood flow to the spinal vasculature [3, 7, 8], which potentially compromises spinal blood circulation and induces degenerative mechanisms in the central nervous system [4, 9].
Among the myriad of spinal disorders that AAC may promote, lumbar disc herniation (LDH) predominantly increases morbidity and long-term movement disabilities, often necessitating surgical interventions, such as intervertebral discectomy, to alleviate symptoms and restore nerve function [10, 11]. The association between AAC occurrence and clinical outcomes of intervertebral discectomy has garnered increasing interest due to its potential implications for risk stratification, treatment optimization, and prognosis of this subset of patients with spinal cord disorders.
While existing literature suggests the clinical implication of AAC in inducing spinal deformities, such as degenerative disc and spinal stenosis, the influence of AAC on patients with comorbid LDH who undergo intervertebral discectomy remains widely unexplored [7, 8, 12,13,14]. Understanding this clinical association is crucial because both AAC and LDH are prevalent conditions, especially in the elderly population [1, 15, 16]; in addition, emerging evidence suggests that AAC may contribute to the pathophysiology of spinal disorders through perturbed microcirculation, induction of inflammatory responses, and neural compression, as well as potentially influence the postoperative recovery and treatment outcomes [4].
Consequently, there is an urgent need for well-designed case-controlled large cohort studies to establish the clinical association between AAC onset and outcomes of LDH intervention.
We report here the results of our single-center prospective cohort study to determine any significant associations between AAC and clinical manifestations of pain symptoms following endoscopic lumbar discectomy.
Methods
Patient Population
This prospective, sequential cohort study enrolled patients who presented with a history of unilateral radiculopathy with or without back pain and with magnetic resonance imaging (MRI)-confirmed diagnosis of posterolateral disc herniation between August 2021 and December 2023. All patients underwent endoscopic lumbar discectomy via the transforaminal approach at our center during the study period. All surgeries were performed by the same two independent surgeons who are specialists in endoscopic lumbar spine surgery. Before surgery, all patients were examined by lumbar, lateral, and anterior-lumbar radiography, lumbar MRI, and three-dimensional computed tomography (3D-CT) imaging of the lumbar spine. Patients with previous spine surgeries, a history of cancer, or missing follow-up data were excluded from the study.
The Institutional Review Board (IRB) approved the study protocol (reference no. 2024–192). Patient consent was obtained in accordance with the Declaration of Helsinki. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines were strictly followed for reporting and documentation of the study results [17].
Outcome Parameters
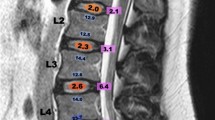
Pre- and postoperative back and leg pain symptoms were evaluated using the numeric rating scale (NRS) [18]. The Oswestry Disability Index (ODI) and the physical functioning (PF), role-physical (RP), and bodily pain (BP) components of the 36-item Short Form Health Survey questionnaire (SF-36) were also assessed. Preoperative and postoperative data were collected at the time of admission and at 1 year post-surgery, respectively. Any changes in the pain intensity were determined by subtracting the postoperative NRS score from the preoperative one. Likewise, changes in ODI, SF-36-PF, SF-36-RP, and SF-36-BP were calculated by subtracting the respective preoperative scores from the postoperative scores. AAC stages were classified based on the Kauppila classification system using lateral lumbar radiographs [19]. This system scores the degree of calcification in the anterior and posterior abdominal aorta in front of the first four lumbar vertebrae. Each segment is scored from 0 to 3, with 0 indicating no calcification, 1 indicating the presence of small, scattered calcific deposits filling less than one-third of the longitudinal wall, 2 indicating that one-third or more but less than two-thirds of the wall is calcified; and 3 indicating that two-thirds or more of the wall is calcified, totaling a maximum score of 24. Patients with scores < 4 were categorized as having mild AAC; those with scores ranging from 5 to 15 were classified as having moderate AAC; and those with scores > 15 were classified with severe AAC (Fig. 1) [20]. To evaluate the severity of lumbar degeneration, we utilized the Pfirrmann grading system at the index lumbar level [21]. Additionally, preoperative degenerative spondylolisthesis, characterized by a vertebra slipping forward at least 3 mm to the vertebra below it, was evaluated using conventional lateral radiography.
The abdominal aortic calcification (AAC) score assessed using a preoperative lateral radiograph of the lumbar (L) spine. The AAC score in this patient was 12 points (0 points at the L1 segment, 2 points at the L2 segment, 5 points at the L3 segment and 5 points at the L4 segment), leading to a classification of moderately severe AAC
Statistical Analysis
Categorical variables were presented as numbers (n) and percentages (%), and continuous variables were reported as medians with interquartile ranges or means with standard deviations (SD), depending on the distribution patterns. Differences in demographic parameters in patients with varying degrees of AAC severity were assessed using Welch’s two-sample t-test or Fisher's exact test. Linear regression analysis was used to test the linear relationship between AAC and post-surgical clinical outcomes. Potential covariates were incorporated into the regression model to conduct multivariate linear regression analysis.
A linear mixed model was used to compare differences in the improvement of clinical outcomes across patients with varying severity levels of AAC. The model included fixed effects for the severity of AAC. Random intercepts at the patient level were used, including baseline value of pain intensity score; ODI, SF-36-PF, SF-36-RP, and SF-36-BP scores; age; the degree of lumbar degeneration; and spondylolisthesis.
All statistical analyses were performed using R-Studio version 2023.03 (Posit Software, Boston, MA, USA), with statistical significance defined as p < 0.05.
Results
Demographic Characteristics
Between August 2021 and December 2023, a cohort of 134 patients presented at our clinic for endoscopic discectomy to treat LDH-associated unilateral radiculopathy. Of these, six patients were lost to follow-up at 1 year post-surgery, and eight patients were identified with various comorbidities and unique clinical conditions, resulting in 120 patients considered to be eligible for inclusion in the final analysis (Fig. 2). The baseline characteristics of these patients are detailed in Table 1. The average age of the 120 participants was 56 years, 48.3% (58/120) were female, and 71.7% (86/120) were non-smokers. In terms of herniation location, the majority (54.2%) of patients had herniation with the left-side onset, with the lumbar position L4/L5 being the most common site (51.7%). The mean (± SD) scores for back pain, leg pain, and ODI were 3.9 ± 2.2, 6.1 ± 1.7, and 31.1 ± 5.1, respectively. Also, 21 patients (17.5%) had preoperative degenerative spondylolisthesis, with most (74.2%) having Pfirrmann grades 2 and 3.
AAC and Clinical Outcomes
Of the 120 patients included in the analysis, 82 (68.3%) exhibited mild AAC and 38 (31.7%) showed moderate-severe AAC symptoms. There were no notable variations in terms of preoperative leg pain, back pain, ODI score, SF36-PF, SF36-RP, and SF36-BP between the mild and moderate-severe AAC patient groups. Notably, patients with moderate-severe AAC were older and had an average higher Pfirrmann grade than those with mild AAC (Table 2).
We found a linear relationship between the Kauppila classification score and postoperative improvement of leg pain, back pain, ODI score, SF-36-PF, SF-36-RP, or SF-36-BP score (Fig. 3). In addition, the multivariate linear regression model revealed a significant correlation between AAC comorbidity and postoperative clinical outcomes. Notably, the removal of relevant confounding factors such as age, body mass index, sex, spondylolisthesis, and degree of lumbar degeneration did not alter the significance of this association (Table 3).
At 1-year post-surgery, the mean change in leg pain was 3.5 points in patients with moderate-severe AAC, which was significantly lower than the mean change in patients with mild AAC (5.0 points) (p < 0.001). Even after adjusting for relevant confounders, this difference remained significant. Similar results were also observed in the postoperative improvement of back pain, ODI score, and SF36-PF, SF36-RP, and SF36-BP scores (Table 4; Fig. 4).
Comparison of clinical outcomes after endoscopic lumbar discectomy between patients with mild AAC and those with moderate to severe AAC revealed significant differences. A Difference in the improvement of leg pain from baseline (p < 0.001). B Difference in the improvement of back pain from baseline (p < 0.001). C Difference in the improvement of ODI (p < 0.001). D Difference in improvement of SF36-BP from baseline (p < 0.001). E Difference in the improvement of SF36-RP from baseline (p = 0.012). F Difference in improvement of SF36-PF from baseline (p < 0.001). AAC Abdominal aortic calcification, ODI Oswestry Disability Index, SF-36 Short Form 36 health survey questionnaire, BP bodily pain component of SF-36, RP role physical component of SF-36, PF physical functioning component of SF-36
In patients without preoperative back pain, the severity of AAC was not significantly correlated with an increased risk of developing post-surgical new-onset back pain (Odds ratio {OR] 3.0, 95% confidence interval [CI] 0.2–52.6, p = 0.447). Additionally, there was no significant difference in the rate of repeat surgery 1 year after the first surgery (OR 0.8, 95% CI 0.2–4.8, p = 0.835).
Discussion
The results of this study revealed a compelling association between AAC severity and postoperative pain outcomes in patients with LDH treated for unilateral radiculopathy. One year after endoscopic discectomy, patients with mild AAC experienced significantly greater mean changes in their back and leg pain scores compared to those with moderate-severe AAC. Beyond pain outcomes, the degree of AAC severity also impacted the postoperative functional outcomes and quality of life in patients with LDH. Importantly, patients with mild AAC demonstrated greater improvements in their functional scores, as indicated by corresponding ODI scores and SF-36 domain scores, than those with moderate-severe AAC symptoms. We did not observe any significant between-group differences regarding the rate of repeat surgery and new onset back pain. These findings suggest that AAC severity may play a crucial role in determining the efficacy of endoscopic lumbar discectomy and alleviating pain symptoms as well as overall functional recovery.
Although previous studies demonstrated an association between AAC severity and treatment outcomes in various musculoskeletal disorders, the specific impact of AAC comorbidity on endoscopic lumbar discectomy in patients with LDH has not been explored in detail [22,23,24]. The results of some studies indicate a positive correlation between AAC severity and the extent of lumbar disc degeneration, suggesting a link to the occurrence of back pain [4, 7, 25]. Several studies have investigated the impact of AAC on surgical outcomes after spine surgery. For example, Sakaura et al. reported that the AAC score has significantly negative correlations with both the Japanese Orthopaedic Association (JOA) score at 2-year post-decompressive laminotomy for lumbar spinal canal stenosis and the improvement of the JOA score [26]. Another study focusing on the association between AAC and lower back pain onset in patients with degenerative spondylolisthesis revealed that AAC can hinder recovery from back pain after lumbar spine fusion surgery [9]. Also, Guven et al. showed that AAC is a significant predictor of perioperative blood loss in posterior spinal fusion surgery [27].
The mechanistic underpinnings of the observed associations between AAC severity and surgical outcomes merit further exploration. The main potential pathophysiology can be explained as AAC being an indicator of the risk of systemic vascular problems, including atherosclerosis [1]. Vascular calcification is not limited to large vessels such as the aorta, but also affects smaller arteries like the coronary and lumbar arteries that supply the lumbar spine [28]. Severe AAC, particularly atherosclerosis in the ostia of segment arteries and stenotic changes in the lumbar artery, can result in reduced spinal blood flow, reducing the delivery of essential nutrients and oxygen to spinal tissues. This, in turn, can lead to conditions like degenerative disc disease, facet arthritis, and stenosis, ultimately impacting post-operative outcomes [29, 30]. Additionally, AAC is associated with elevated levels of pro-inflammatory cytokines [31]. Heightened inflammatory response can impair tissue healing, increase postoperative pain, and contribute to the recurrence of symptoms. Thirdly, AAC can alter the biomechanical properties of the aorta and surrounding structures, potentially affecting spinal mechanics. Changes in spinal biomechanics due to AAC may contribute to altered load distribution on the lumbar spine, potentially affecting the stability and success of the discectomy.
Compromised vascular status may also contribute to an increased risk of infections. AAC is also associated with endothelial dysfunction, characterized by impaired vasodilation and increased vascular stiffness [32]. Endothelial dysfunction can cause suboptimal tissue oxygenation, compromise wound healing, and increase susceptibility to postoperative complications such as thrombosis and ischemia [33]. Severe AAC alters spinal blood flow dynamics and increases the risk of vascular injury during surgical procedures [7].
The findings of this study are likely to have practical implications for clinical decision-making and patient counseling. Surgeons and healthcare providers should consider AAC severity as a potential prognostic factor when discussing surgical risks, expected outcomes, and postoperative care plans with patients and their caregivers. Tailoring intervention strategies and follow-up protocols based on the AAC severity may improve patient satisfaction, functional recovery, and long-term outcomes.
The novelty of this study lies in its focused investigation of the impact of AAC severity on the recovery of postoperative pain symptoms and functional outcomes in lumbar discectomy-treated patients. It is the first study to evaluate the impact of AAC on clinical outcomes after endoscopic lumbar discectomy. Here, we strictly adjusted all possible confounding factors, including baseline clinical indicators, age, spondylolisthesis, and degree of lumbar degeneration, to test the rigor of the study results, and found that the observed differences in pain and functional outcomes between the mild and moderate-severe AAC patient groups remained statistically significant, underscoring a high significance of the association between AAC severity and postoperative clinical outcomes.
Limitations and Future Directions
Several limitations of this study warrant acknowledgment. First, the small sample size and reliance on a single-center cohort might limit the generalizability of our study findings. Second, long-term follow-up investigations, which we could not accommodate in this report, assessing the durability of outcomes and the impact of interventions targeting AAC are also warranted. Third, a significant association was observed between the severity of AAC symptoms and the clinical outcomes of endoscopic lumbar discectomy. However, establishing causation is challenging due to potential confounding factors that were not addressed in the current study, such as lifestyle factors, inflammatory markers, and socioeconomic status. Future randomized controlled trials is still needed. Furthermore, exploring the molecular and cellular mechanisms linking AAC severity to surgical outcomes could pave the way for targeted therapies and personalized treatment strategies.
Conclusion
The findings of this study highlight a significant association between the severity of AAC symptoms and clinical outcomes among the comorbid LDH patients who underwent endoscopic lumbar discectomy. Notably, patients with mild AAC demonstrated superior postoperative improvements in terms of back and leg pain symptoms, functional scores, and postoperative quality of life compared to those with moderate-severe AAC, suggesting that AAC severity may serve as a prognostic factor in predicting surgical outcomes and guiding LDH patient management strategies following endoscopic lumbar discectomy. Finally, assessment of AAC symptoms is recommended as part of the preoperative evaluation of patients with LDH, allowing better risk stratification and effective personalized care planning.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Szulc P. Abdominal aortic calcification: a reappraisal of epidemiological and pathophysiological data. Bone. 2016;84:25–37.
Lehto S, Niskanen L, Suhonen M, Rönnemaa T, Laakso M. Medial artery calcification. A neglected harbinger of cardiovascular complications in non-insulin-dependent diabetes mellitus. Arterioscler Thromb Vasc Biol. 1996;16(8):978–83.
Jayalath RW, Mangan SH, Golledge J. Aortic calcification. Eur J Vasc Endovasc Surg. 2005;30(5):476–88.
Schönnagel L, Muellner M, Suwalski P, et al. Abdominal aortic calcification is independently associated with lumbar endplate degeneration. Eur Spine J. 2023;32(10):3387–93.
El Maghraoui A, Rezqi A, Mounach A, Achemlal L, Bezza A, Ghozlani I. Relationship between vertebral fracture prevalence and abdominal aortic calcification in men. Rheumatology (Oxford). 2012;51(9):1714–20.
Majjad A, Ghassem MA, Toufik H, et al. Relationship between vertebral fracture prevalence and abdominal aortic calcification in women with rheumatoid arthritis. Bone. 2020;141:115599.
Kauppila LI. Atherosclerosis and disc degeneration/low-back pain–a systematic review. Eur J Vasc Endovasc Surg. 2009;37(6):661–70.
Sfyri P, Matsakas A. Crossroads between peripheral atherosclerosis, western-type diet and skeletal muscle pathophysiology: emphasis on apolipoprotein E deficiency and peripheral arterial disease. J Biomed Sci. 2017;24(1):42.
Schönnagel L, Muellner M, Suwalski P, et al. Association of abdominal aortic calcification and lower back pain in patients with degenerative spondylolisthesis. Pain. 2024;165(2):376–82.
Deyo RA, Mirza SK. CLINICAL PRACTICE. Herniated lumbar intervertebral disk. N Engl J Med. 2016;374(18):1763–72.
Bailey CS, Rasoulinejad P, Taylor D, et al. Surgery versus conservative care for persistent sciatica lasting 4 to 12 months. N Engl J Med. 2020;382(12):1093–102.
Sakaura H, Ikegami D, Fujimori T, Sugiura T, Mukai Y, Hosono N. Does atherosclerosis have negative impacts on early adjacent segment degeneration after posterior lumbar interbody fusion? Global Spine J. 2021;11(5):674–8.
Mohammad A, Lohan D, Bergin D, et al. The prevalence of aortic calcification on vertebral fracture assessment imaging among patients with rheumatoid arthritis. J Clin Densitom. 2014;17(1):72–7.
Li W, Djuric N, Cobbaert C, Vleggeert-Lankamp CLA. Modic changes in the lumbar spine: exploring their association with abdominal aortic calcification as a potential indicator of systemic atherosclerosis. World Neurosurg. 2024;184:e503–10.
Teh R, Prince RL, Sim M, et al. Abdominal aortic calcification, cardiac troponin I and atherosclerotic vascular disease mortality in older women. Heart. 2022;108(16):1274–80.
Ramaswami R, Ghogawala Z, Weinstein JN. Management of sciatica. N Engl J Med. 2017;376(12):1175–7.
Cuschieri S. The STROBE guidelines. Saudi J Anaesth. 2019;13(Suppl 1):S31-s34.
Bielewicz J, Daniluk B, Kamieniak P. VAS and NRS, same or different? Are visual analog scale values and numerical rating scale equally viable tools for assessing patients after microdiscectomy? Pain Res Manag. 2022;2022:5337483.
Kauppila LI, Polak JF, Cupples LA, Hannan MT, Kiel DP, Wilson PW. New indices to classify location, severity and progression of calcific lesions in the abdominal aorta: a 25-year follow-up study. Atherosclerosis. 1997;132(2):245–50.
Verbeke F, Van Biesen W, Honkanen E, et al. Prognostic value of aortic stiffness and calcification for cardiovascular events and mortality in dialysis patients: outcome of the calcification outcome in renal disease (CORD) study. Clin J Am Soc Nephrol. 2011;6(1):153–9.
Urrutia J, Besa P, Campos M, et al. The Pfirrmann classification of lumbar intervertebral disc degeneration: an independent inter- and intra-observer agreement assessment. Eur Spine J. 2016;25(9):2728–33.
Simon SP, Fodor D, Muntean L, Poanta L, Cristea P, Rednic S. Bone mineral density, vertebral fractures and body mass index in postmenopausal women with abdominal aortic calcification. Endocr Res. 2014;39(1):1–6.
Szulc P, Kiel DP, Delmas PD. Calcifications in the abdominal aorta predict fractures in men: MINOS study. J Bone Miner Res. 2008;23(1):95–102.
Golestani R, Tio R, Zeebregts CJ, et al. Abdominal aortic calcification detected by dual X-ray absorptiometry: a strong predictor for cardiovascular events. Ann Med. 2010;42(7):539–45.
Kauppila LI, McAlindon T, Evans S, Wilson PW, Kiel D, Felson DT. Disc degeneration/back pain and calcification of the abdominal aorta. A 25-year follow-up study in Framingham. Spine (Phila Pa 1976). 1997;22(14):1642–7.
Sakaura H, Ikegami D, Fujimori T, Sugiura T, Owaki H, Fuji T. Abdominal aortic calcification is a significant poor prognostic factor for clinical outcomes after decompressive laminotomy for lumbar spinal canal stenosis. Global Spine J. 2019;9(7):724–8.
Guven AE, Evangelisti G, Schönnagel L, et al. Abdominal aortic calcification is an independent predictor of perioperative blood loss in posterior spinal fusion surgery. Eur Spine J. 2024;33(5):2049–55.
Kauppila LI. Prevalence of stenotic changes in arteries supplying the lumbar spine. A postmortem angiographic study on 140 subjects. Ann Rheum Dis. 1997;56(10):591–5.
Sethi A, Taylor DL, Ruby JG, et al. Calcification of the abdominal aorta is an under-appreciated cardiovascular disease risk factor in the general population. Front Cardiovasc Med. 2022;9:1003246.
Beckworth WJ, Holbrook JF, Foster LG, Ward LA, Welle JR. Atherosclerotic disease and its relationship to lumbar degenerative disk disease, facet arthritis, and stenosis with computed tomography angiography. PM R. 2018;10(4):331–7.
Engelen SE, Robinson AJB, Zurke YX, Monaco C. Therapeutic strategies targeting inflammation and immunity in atherosclerosis: how to proceed? Nat Rev Cardiol. 2022;19(8):522–42.
Karwowski W, Naumnik B, Szczepański M, Myśliwiec M. The mechanism of vascular calcification—a systematic review. Med Sci Monit. 2012;18(1):Ra1-11.
Deanfield JE, Halcox JP, Rabelink TJ. Endothelial function and dysfunction: testing and clinical relevance. Circulation. 2007;115(10):1285–95.
Acknowledgements
We thank the participants of the study
Medical Writing
The authors wish to thank MedEditing LLC company for providing medical writing support, which was funded by the authors, in accordance with Good Publication Practice Guidelines (https://www.ismpp.org/gpp-2022).
Funding
The Rapid Service Fee was partly funded by the The First Affiliated Hospital of Soochow Unversity and partly funded by the authors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conceptualization, methodology, writing-review, and editing of the manuscript. Ying Zhang conducted the clinical investigation. Peng Huang developed the study design, performed the statistical analysis, and drafted the manuscript. ZhaoJi Zhu and XiaoHong Jin contributed to validation of the data and revision of the manuscript. All named authors meet the International Committee of Medical Journal Editors(ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Corresponding authors
Ethics declarations
Conflict of Interest
All authors (Ying Zhang, Zhaoji Zhu, Xiaohong Jin, Peng Huang) have nothing to disclose.
Ethical Approval
The Institutional Review Board (IRB) approved the study protocol (Reference No. 2024–192). Patient consent was obtained in accordance with the Declaration of Helsinki. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines were strictly followed for reporting and documentation of the study results [17]. The use of Oswestry Disability Index (ODI) and Pfirrmann grading system in the manuscript was permitted by the respective developers.
Additional information
Prior Publication We confirm that the content of this manuscript has not been previously published and is not under consideration for publication elsewhere. Portions of this work have not been presented at conferences or included in preprint repositories.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Zhang, Y., Zhu, Z., Jin, X. et al. Association of Abdominal Aortic Calcification and Clinical Outcomes in LDH Patients Treated with Endoscopic Lumbar Discectomy. Pain Ther (2024). https://doi.org/10.1007/s40122-024-00633-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40122-024-00633-2