Abstract
Objective
This study aimed to establish and validate a nomogram prognostic model for predicting short-term efficacy of acetylcholine receptor antibody-positive (AChR-Ab+) generalized myasthenia gravis (GMG).
Methods
A retrospective observational study was conducted at the First Hospital of Shanxi Medical University, enrolling patients diagnosed with AChR-Ab+ GMG from May 2020 to September 2022. The primary outcome was the change in the Myasthenia Gravis Foundation of America (MGFA) post-intervention status after 6 months of standard treatment. Predictive factors were identified through univariate and multivariate logistic regression analyses, with significant factors incorporated into the nomogram. The bootstrap test was used for internal validation of the nomogram model. Model performance was assessed using calibration curves, receiver-operating characteristic curve analysis, and decision curve analysis (DCA).
Results
A total of 90 patients were enrolled, of whom 30 achieved unchanged or worse status after 6 months of standard therapy. Univariate logistic regression analysis showed that quantitative myasthenia gravis score, gender, body mass index, course of disease, hemoglobin levels, and white blood cell counts were six potential predictors. These factors were used for multivariate logistic regression analysis, and a nomogram was constructed. The calibration curve showed that the predicted value was in good agreement with the actual value (p = 0.707), and the area under the curve value (0.792, 95% CI 0.686–0.899) indicated good discrimination ability. DCA suggests that this model has potential clinical application value.
Conclusion
The constructed nomogram, based on key patient indicators, shows promise as a clinically useful tool for predicting the short-term efficacy of treatment of AChR-Ab+ GMG. Validation in larger, multicenter cohorts is needed to further substantiate its applicability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Myasthenia gravis (MG) is a rare but impactful autoimmune neuromuscular disorder. Its variable treatment response and a significant number of patients experiencing suboptimal therapy outcomes highlight the need for effective predictive tools |
The study focuses on developing and validating a predictive nomogram for short-term (6 months) treatment efficacy in patients with acetylcholine receptor antibody-positive (AChR-Ab+) generalized myasthenia gravis (GMG), addressing the need for personalized treatment strategies |
What did the study ask?/What was the hypothesis of the study? |
The research aimed to construct a nomogram model that could accurately predict the short-term treatment response in AChR-Ab + GMG patients |
What was learned from the study? |
The developed nomogram model, incorporating factors like gender, body mass index (BMI), disease duration, hemoglobin levels, and white blood cell count, proved highly accurate in predicting suboptimal responses to 6-month standard treatment in patients with AChR-Ab+ GMG |
This model signifies a step towards personalized treatment strategies in MG, emphasizing the importance of individual patient characteristics in predicting treatment efficacy |
The study underscores the need for further validation of this nomogram in larger multicenter cohorts to confirm its effectiveness and applicability in diverse patient populations |
Introduction
Myasthenia gravis (MG) is an autoimmune neuromuscular disorder characterized by fluctuating muscle weakness and fatigability [1, 2]. It occurs when antibodies attack and disrupt the communication between nerves and muscles at the neuromuscular junction. Despite its rarity with a prevalence ranging from 15 to 179 per million people [3], the impact of MG on patients’ quality of life and health care systems is substantial [4].
Treatment response in MG is variable and can range from complete symptom relief to minimal improvement or even disease progression [5]. Unfortunately, a significant proportion of patients have a suboptimal response to therapy. Studies report that up to 10–20% of patients do not achieve satisfactory control of their symptoms despite treatment [6], highlighting the need for better therapeutic strategies and prognostic tools.
The ability to predict which patients are more likely to have a poor response to treatment would have significant clinical implications. Early prediction of treatment response can guide personalized treatment plans, potentially enabling more aggressive or alternative therapies for patients identified as high-risk. This can improve the therapeutic success rate, mitigate disease progression, decrease hospitalization rates, and improve quality of life [7]. Furthermore, it can facilitate patient counseling regarding the prognosis and expected course of the disease, which is important in disease acceptance and treatment adherence [7].
Current prediction of treatment response primarily relies on clinical experience and certain indicators such as disease duration and severity [8,9,10]. However, these measures are not always reliable or precise. Therefore, there is a pressing need to develop reliable, data-driven prediction tools to guide therapeutic decision-making in MG. Zhao et al. developed a model for the 12-month outcome of AChR-Ab+ (acetylcholine receptor antibody-positive) GMG (generalized myasthenia gravis) patients, which could predict short-term prognosis effectively [11]. However, the treatment responses of a shorter term are also of great importance. Physicians and patients are more interested in whether a balance between efficacy and side effects can be achieved within a short period.
In the realm of clinical decision-making, particularly in neurology, the nomogram stands as a pivotal tool, bridging the gap between statistical predictions and practical patient care. A nomogram is a graphical representation tool that simplifies the statistical prediction model into a user-friendly interface, allowing for the visualization and calculation of the probability of a clinical event based on multiple predictors. In the context of AChR-Ab+ GMG, the complexity of the disease necessitates an approach that can encompass multiple variables to predict patient outcomes accurately. Here, the nomogram’s ability to integrate various clinical parameters into a single predictive model is invaluable. It aids neurologists in making informed, personalized treatment decisions, aligning with the evolving paradigm of precision medicine in neurology. The present study aims to develop and validate a predictive nomogram for poor shorter-term (6 months) outcomes in AChR-Ab+ GMG patients.
Methods
Study Design and Setting
This research is a single-center, retrospective, observational study that involved a consecutive case series design. It adhered to the ethical guidelines set forth in the Declaration of Helsinki and was approved by the ethical committee at The First Hospital of Shanxi Medical University (no. KYLL-2023-107). Due to the retrospective nature of the study, the requirement for written informed consent for the utilization of clinical data was waived.
Study Population
Between September 2020 and November 2022, a retrospective cohort study was conducted at The First Hospital of Shanxi Medical University to analyze patients diagnosed with GMG. The inclusion criteria encompassed those who tested positive for the acetylcholine receptor antibody (serum AChR-Ab was detected by enzyme linked immunosorbent assay, and ≥ 0.450 nmol/l was positive; the detection limit is 20 nmol/l), demonstrated adherence to their therapeutic interventions, maintained a minimum outpatient follow-up of 6 months post-diagnosis, and underwent all essential diagnostic procedures, including electromyography and blood biochemistry (detailed in Supplemental Table 1). Power analysis using G*Power (version 3.1.9.7) showed that the sample size was 44 when the effect size was 80%. To analyze the effect of AChR-Ab level on the prognosis of the disease, according to the level of AChR-Ab, the patients were divided into three groups: < 10 nmol/l, 10-20 nmol/l and > 20 nmol/l. An additional criterion was the absence of other infectious diseases at the time of initial diagnosis. Therapeutically, 49 patients initiated treatment with a regimen starting at 20 mg prednisone acetate, increased by 10 mg weekly until the desired dose, and then gradually reduced. Additionally, 64 patients were prescribed tacrolimus, targeting a blood concentration of 3 to 10 mmol/l by the end of the first week, with 23 of them concurrently treated with both prednisone acetate tablets and tacrolimus capsules. All patients received pyridostigmine 60 mg three times daily.
Outcome Measures
The primary outcome of the present study was the Myasthenia Gravis Foundation of America (MGFA) post-intervention status after 6-month treatment. Poor and good prognosis was defined as “unchanged or worse” and “improved or better,” respectively. This outcome measure leverages the MGFA post-intervention status, a metric frequently utilized in both clinical practice and research environments, reflecting its robustness and widely recognized utility in assessing treatment effectiveness in the context of MG [12, 13].
For a thorough evaluation of treatment efficacy predictors in AChR-Ab+ GMG patients, we analyzed a spectrum of demographic and clinical indicators. Demographic insights were derived from age of onset and gender, with lifestyle elements captured via smoking and alcohol consumption status. Vital parameters included body mass index (BMI), duration, hematological measures [red blood cell (RBC), hemoglobin (Hb), white blood cell (WBC), lymphocyte (LYM), albumin (Alb)], and arterial blood gas readings [power of hydrogen (PH), oxygen partial pressure (PO2), partial pressure of carbon dioxide (PCO2)]. Autoantibody statuses of AChR-Ab and Titin-Ab, along with the RNS, informed on the disease’s neurophysiological aspects. Clinical manifestations such as extraocular, palpebralis, bulbar, vegetative nerve, thymoma, and thymic hyperplasia, as well as MGFA scale for disease severity and hypertension as a comorbidity, were also integrated. These indicators aimed to provide a comprehensive evaluation of potential prognostic markers.
Statistical Analyses
Data analyses were performed using R statistical software (version 4.1.2). Categorical variables are reported as counts and percentages, whereas continuous variables are expressed as means ± standard deviations for normally distributed data and medians (interquartile ranges, IQR) for skewed data. The Shapiro-Wilk test was used to assess data for normality. To evaluate the differences in clinical efficacy among patients receiving various treatment methods at a 6-month follow-up, a chi-square (χ2) test was performed, with a significance threshold set at p < 0.050. Predictive factors were initially screened using univariate logistic regression with a cut-off of p < 0.050. Subsequently, significant predictors identified from the univariate analysis were included in a multivariate logistic regression model and to construct a nomogram predictive model. The model’s predictive performance was evaluated using receiver-operating characteristic (ROC) curve analysis, calibration curves, and decision curve analysis (DCA). Note that to estimate the performance of the model, we used a multiple bootstrap procedure (n = 500 bootstraps).
Results
Patient Characteristics:
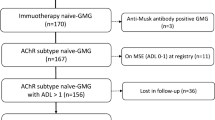
Patient recruitment for the study is presented in the flow diagram (Fig. 1). Ninety of 248 patients were enrolled. The median age at disease onset was 55 (IQR: 40.500–64) years, and 46.700% were women. A total of 30 patients were ‘unchanged’ or ‘worse’ after 6-month standard treatment (Table 1).
Development of the Nomogram
ROC analysis was utilized to establish the cut-off value for all count data, which were subsequently transformed into categorical variables to facilitate further analysis. Logistic regression analysis showed that there was no statistical significance in the effect of different treatment schemes on the condition of patients at 6 months (P: prednisone acetate 0.257, tacrolimus 0.150, drug combination 0.163), indicating the potential influence of other factors on this outcome. The correlation between AChR-Ab level and disease severity has been controversial. Logistic regression analysis showed that there was no significant difference between AChR-Ab level and disease severity at 6 months (P < 10 nmol/l 0.401, 10–20 nmol/l 0.352, > 20 nmol/l 0.505). Univariate logistic regression analysis revealed that factors such as gender, BMI, duration of illness, Hb, WBC, and LYM met the significance criterion of p < 0.050, as detailed in Table 2. In several studies concerning prognostic factors of MG, the quantitative myasthenia gravis (QMG) score has been identified as a critical determinant of poor prognosis [11, 14, 15]. Consequently, despite not satisfying the initial P-value criterion, QMG was included in further analyses. Considering the study’s limited sample size, we emphasized the robustness of the model. This entailed a restriction on the number of risk factors evaluated, giving precedence to those with higher odds ratios (OR), especially when similar types of factors were competing. Detailed outcomes from the multivariate analysis are presented in Table 3. Building on these insights, a nomogram model was developed, as illustrated in Fig. 2A.
A Nomogram prediction model obtained in this study. Hb hemoglobin, WBC white blood cell, BMI body mass index, QMG quantitative myasthenia gravis. B Calibration curve, which show high accuracy; x-axis represents the predictive potential for MG exacerbation, and y-axis represents the actual incidence of it; perfect prediction based on an idealized model is represented by the diagonal dotted line; the ideal performance of the nomogram is represented by the red solid line, and the closer the red solid line fits the dotted line, the better the prediction will be. C ROC analysis of the nomogram (QMG included), the nomogram (Non-QMG), and the bootstrap test model. D and E are decision analysis curves, and the use of the model in the blue range has high clinical benefit and a wide range of application
Model Evaluation
The calibration curve is a pivotal tool for evaluating the accuracy of a nomogram model. In our study, this curve demonstrated a negligible difference between the predicted outcomes and actual observations, indicating a well-calibrated model (Fig. 2B; p = 0.707). Furthermore, ROC analysis, a benchmark technique for assessing predictive accuracy, was employed. The AUC of our model was 0.792 (95% CI 0.686–0.899), suggesting substantial discriminative ability in identifying patients likely to exhibit poor efficacy following a 6-month standard treatment regimen (Fig. 2C).
To rigorously test the model’s validity, we conducted an analysis comparing its accuracy with and without the inclusion of QMG. Removal of QMG led to a decrease in accuracy (AUC: 0.749; 95% CI 0.634–0.863); however, DeLong’s test revealed no statistically significant difference between the two models (p = 0.079). Consequently, we opted for the model inclusive of QMG because of its superior accuracy.
We further assessed the clinical utility of our prognostic model using a DCA. The blue section of Fig. 2D delineates the model’s performance threshold area, underscoring its practical applicability.
In summary, the constructed nomogram exhibits high predictive accuracy for suboptimal response to a 6-month standard treatment in patients with AChR-Ab+ GMG. The validation of the predictive model is a crucial step in establishing its reliability and applicability in a clinical setting. For this purpose, we employed a bootstrapping technique with n = 500 iterations. Bootstrapping is a resampling method used extensively in statistics to estimate the distribution of a sample statistic by randomly selecting samples with replacement from the original dataset. This approach is particularly beneficial in assessing the stability and accuracy of a predictive model. In our study, the bootstrapping method was used to validate the nomogram developed for predicting the short-term prognosis of patients with AChR-Ab+ GMG. The choice of 500 bootstrap resamples was made to balance computational efficiency with the robustness of the validation process. This conclusion is bolstered by a bootstrap test with 500 repetitions on our dataset, which confirmed the consistency of the derived equations with those of the initial nomogram model (Fig. 2D, E). Such congruence not only reaffirms the robustness of our original model but also solidifies its reliability.
Discussion
In our study, we have constructed a predictive nomogram to estimate the short-term response to treatment in AChR-Ab+ GMG patients based on six significant factors, namely gender, BMI, QMG score, disease duration, Hb, and WBC count. This nomogram represents a step towards a more personalized, data-driven approach to the management of MG.
The finding that female gender is predictive of poorer response aligns with the recognized gender disparities in autoimmune diseases, including MG. Females have been shown to have a higher susceptibility and more severe disease course [16]. Hormonal and genetic factors have been proposed to underlie these disparities, and a deeper exploration of these aspects might refine the predictive power of gender in our model and contribute to a better understanding of sex-related differences in MG outcomes.
Prior literature has consistently indicated a linkage between elevated BMI and heightened clinical symptomatology in MG patients [16, 17]. However, there is a conspicuous dearth of research elucidating its implications on short-term prognostic outcomes. Within our investigational purview, an augmented BMI emerged as a potential harbinger of improved short-term outcomes. This correlation may stem from the perturbation in the disease’s natural trajectory, likely attributable to the initiation of a more rigorous therapeutic regimen for these particular patients during the disease’s early stages. For instance, within our dataset, 33.33% of patients with a BMI > 25.610 were administered a combined regimen of steroids and tacrolimus compared to 19.610% of those with a BMI < 25.61 who received the dual therapy. Nevertheless, it remains imperative to underscore the significance of adept weight management in the holistic care paradigm for MG.
Our model’s emphasis on high QMG scores and longer disease duration as indicators of poorer response is congruent with the understanding that these factors are generally linked to disease severity and chronicity. This stresses the need for early, aggressive intervention in patients presenting with severe symptoms and those with long-standing disease to improve treatment outcomes. This result is consistent with the results of some previous studies [14], but some studies have shown that patients with higher QMG scores at initial diagnosis have better prognosis [11, 18]. These differences may be explained by the longer observation periods of these studies, during which more drug doses may be administered, and by greater attention from physicians and patients with more severe symptoms. Second, changes in the definition of outcomes according to QMG scores in these studies may have influenced the results of the statistical analyses.
The inclusion of Hb and WBC levels in our model underscores the importance of considering the overall health status of MG patients. Suboptimal Hb levels coupled with elevated WBC counts potentially indicate systemic health anomalies—encompassing inflammatory responses, infectious processes, or persistent pathological conditions—that could influence the therapeutic responsiveness in MG. The amplification in WBC count, along with derivative markers like the neutrophil-to-lymphocyte ratio (NLR), predominantly indicates an underlying systemic inflammatory condition. Notably, NLR has emerged as a prognostic marker for a plethora of pathologies, as evidenced by several research publications [19,20,21]. In a retrospective investigation by Hsu et al., leukocytosis was identified as a robust predictor for mortality in MG [22]. Complementing this, numerous studies have elucidated the association of an elevated NLR with an adverse prognosis in MG [23,24,25]. Anemia’s role as a harbinger of adverse outcomes spans various diseases including but not limited to ischemic stroke and chronic obstructive pulmonary disease [26, 27]. Notably, a plethora of clinical studies have documented the occurrence of anemia in MG cohorts [28, 29]. Certain academic contributions suggest a heightened severity of MG in anemic individuals, although the underlying mechanisms remain an area of ongoing inquiry [22, 30]. Extrapolating from ischemic stroke analyses, some scholars propose that anemia might inflict localized skeletal muscle hypoxia by compromising tissue oxygenation, subsequently diminishing muscular prowess [31]. Such a hypothesis might elucidate the observation where a decrement in Hb levels correlates with exacerbated MG presentations.
The direction our study has taken towards developing predictive models for patient outcomes mirrors broader trends in MG research and healthcare. Machine learning and other advanced analytics tools are being employed to construct robust predictive models for disease outcomes, as illustrated by the study of Huahua Zhong et al. [15]. This move towards personalized medicine allows clinicians to tailor treatment strategies based on a patient’s individual disease characteristics and potential response to therapy.
Our study is an addition to the suite of tools being developed to predict disease outcomes in MG. However, it is important to note that further validation of our model in larger, multicenter cohorts is needed to ensure its broad applicability and robustness. The aim is to develop a model that can be used across various patient populations, thereby contributing significantly to improving the quality of life for individuals living with MG.
Historical literature has posited that factors such as dysphagia, hypoproteinemia, presence of anti-titin antibodies, and an advanced age of onset might be indicative of a worsening patient prognosis [13, 32, 33]. In the current investigation, variables including bulbar symptoms, thymoma, serum albumin levels, and the presence of anti-titin antibodies were precluded because their P-values exceeded 0.2 in the univariate logistic regression analysis. It is pertinent to note that the limited sample size necessitated stringent inclusion criteria. Consequently, the ‘age of onset’ variable, failing to meet these stipulated criteria, was not incorporated into the analysis.
In our endeavor to establish a nomogram for predicting short-term efficacy in AChR-Ab+ GMG patients, several limitations merit consideration. First, the study was conducted at a single institution and, with a cohort of only 90 patients, might reflect regional-specific biases and limitation of small sample size, thereby limiting its generalizability. Additionally, the retrospective design inherently introduces potential biases, such as selection bias. Furthermore, while our statistical models are conventional, they operate under certain assumptions, and these might not always mirror the nuances of complex clinical scenarios. Lastly, the model’s external validation across diverse settings remains imperative to affirm its universal applicability. In light of these considerations, we strongly advocate for future multicentric research with larger sample sizes to enhance the model’s robustness.
Conclusion
In conclusion, the predictive nomogram developed in our study provides an additional tool for the personalized management of MG. Incorporating key demographic, clinical, and laboratory parameters, our model reflects the multifaceted nature of MG prognosis. With further validation and potential refinement, this model could play a vital role in guiding therapeutic decisions, ultimately leading to improved patient outcomes.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol. 2015;14(10):1023–36.
Gilhus NE, Tzartos S, Evoli A, Palace J, Burns TM, Verschuuren J. Myasthenia gravis. Nat Rev Dis Primers. 2019;5(1):30.
Carr AS, Cardwell CR, McCarron PO, McConville J. A systematic review of population based epidemiological studies in myasthenia gravis. BMC Neurol. 2010;10:46.
Mahic M, Bozorg A, DeCourcy J, Golden K, Gibson G, Taylor C, et al. Physician- and patient-reported perspectives on myasthenia gravis in Europe: a real-world survey. Orphanet J Rare Dis. 2023;18(1):169.
Silvestri NJ, Wolfe GI. Treatment-refractory myasthenia gravis. J Clin Neuromuscul Dis. 2014;15(4):167–78.
Sanders DB, Wolfe GI, Benatar M, Evoli A, Gilhus NE, Illa I, et al. International consensus guidance for management of myasthenia gravis: executive summary. Neurology. 2016;87(4):419–25.
Offit K. Personalized medicine: new genomics, old lessons. Hum Genet. 2011;130(1):3–14.
Geng Y, Zhang H, Wang Y. Risk factors of myasthenia crisis after thymectomy among myasthenia gravis patients: a meta-analysis: erratum. Medicine. 2020;99(1): e18622.
Alqarni F, Almalki D, Aljohani Z, Ali A, AlSaleem A, Alotaibi N, et al. Prevalence and risk factors of myasthenia gravis recurrence post-thymectomy. Neurosciences (Riyadh, Saudi Arabia). 2021;26(1):4–14.
Chen K, Li Y, Yang H. Poor responses and adverse outcomes of myasthenia gravis after thymectomy: predicting factors and immunological implications. J Autoimmun. 2022;132: 102895.
Zhao R, Wang Y, Huan X, Zhong H, Zhou Z, Xi J, et al. Nomogram for short-term outcome assessment in AChR subtype generalized myasthenia gravis. J Transl Med. 2021;19(1):285.
Jaretzki A 3rd, Barohn RJ, Ernstoff RM, Kaminski HJ, Keesey JC, Penn AS, et al. Myasthenia gravis: recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Neurology. 2000;55(1):16–23.
Yoshimoto Y, Ishida S, Hosokawa T, Arawaka S. Assessment of clinical factors affecting outcome of myasthenia gravis. Muscle Nerve. 2021;64(1):90–4.
Nelke C, Stascheit F, Eckert C, Pawlitzki M, Schroeter CB, Huntemann N, et al. Independent risk factors for myasthenic crisis and disease exacerbation in a retrospective cohort of myasthenia gravis patients. J Neuroinflamm. 2022;19(1):89.
Zhong H, Ruan Z, Yan C, Lv Z, Zheng X, Goh LY, et al. Short-term outcome prediction for myasthenia gravis: an explainable machine learning model. Ther Adv Neurol Disord. 2023;16:17562864231154976.
Ortona E, Pierdominici M, Maselli A, Veroni C, Aloisi F, Shoenfeld Y. Sex-based differences in autoimmune diseases. Annali dell’Istituto superiore di sanita. 2016;52(2):205–12.
Wilcke H, Glaubitz S, Kück F, Anten C, Liebetanz D, Schmidt J, et al. Female sex and overweight are associated with a lower quality of life in patients with myasthenia gravis: a single center cohort study. BMC Neurol. 2023;23(1):366.
Katzberg HD, Barnett C, Bril V. Predictors of response to immunomodulation in patients with myasthenia gravis. Muscle Nerve. 2012;45(5):648–52.
Liberski PS, Szewczyk M, Krzych ŁJ. Haemogram-derived indices for screening and prognostication in critically ill septic shock patients: a case-control study. Diagnostics (Basel, Switzerland). 2020;10(9):638.
Gunaldi M, Goksu S, Erdem D, Gunduz S, Okuturlar Y, Tiken E, et al. Prognostic impact of platelet/lymphocyte and neutrophil/lymphocyte ratios in patients with gastric cancer: a multicenter study. Int J Clin Exp Med. 2015;8(4):5937–42.
Halazun HJ, Mergeche JL, Mallon KA, Connolly ES, Heyer EJ. Neutrophil-lymphocyte ratio as a predictor of cognitive dysfunction in carotid endarterectomy patients. J Vasc Surg. 2014;59(3):768–73.
Hsu CW, Chen NC, Huang WC, Lin HC, Tsai WC, Huang CC, et al. Hemogram parameters can predict in-hospital mortality of patients with myasthenic crisis. BMC Neurol. 2021;21(1):388.
Yang DH, Qian MZ, Wei MM, Li J, Yu MM, Lu XM, et al. The correlation of neutrophil-to-lymphocyte ratio with the presence and activity of myasthenia gravis. Oncotarget. 2017;8(44):76099–107.
Jiang Z, Ning Z, Yang L, Chen B, Tang J, Zhang J, et al. The correlation of neutrophil-to-lymphocyte ratio with the presence and short-time curative effect of myasthenia gravis in children: a retrospectively study. Int J Neurosci. 2021;131(9):894–901.
Duan Z, Jia A, Cui W, Feng J. Correlation between neutrophil-to-lymphocyte ratio and severity of myasthenia gravis in adults: a retrospective study. J Clin Neurosci. 2022;106:117–21.
Hao Z, Wu B, Wang D, Lin S, Tao W, Liu M. A cohort study of patients with anemia on admission and fatality after acute ischemic stroke. J Clin Neurosci. 2013;20(1):37–42.
Martinez-Rivera C, Portillo K, Muñoz-Ferrer A, Martínez-Ortiz ML, Molins E, Serra P, et al. Anemia is a mortality predictor in hospitalized patients for COPD exacerbation. COPD. 2012;9(3):243–50.
Sekiguchi K, Ishizuchi K, Takizawa T, Motegi H, Oyama M, Nakahara J, et al. Anemia in female patients with myasthenia gravis. PLoS ONE. 2022;17(9): e0273720.
Shi J, Huan X, Zhou L, Xi J, Song J, Wang Y, et al. Comorbid autoimmune diseases in patients with myasthenia gravis: a retrospective cross-sectional study of a Chinese cohort. Front Neurol. 2021;12: 790941.
Lv Z, Zhong H, Huan X, Song J, Yan C, Zhou L, et al. predictive score for in-hospital mortality of myasthenic crisis: a retrospective Chinese cohort study. Eur Neurol. 2019;81(5–6):287–93.
Yoshimura Y, Wakabayashi H, Nagano F, Bise T, Shimazu S, Shiraishi A. Low hemoglobin levels are associated with sarcopenia, dysphagia, and adverse rehabilitation outcomes after stroke. J Stroke Cerebrovasc Dis. 2020;29(12): 105405.
Kim KH, Kim SW, Cho J, Chung HY, Shin HY. Anti-titin antibody is associated with more frequent hospitalization to manage thymoma-associated myasthenia gravis. Front Neurol. 2022;13: 978997.
Tang YL, Ruan Z, Su Y, Guo RJ, Gao T, Liu Y, et al. Clinical characteristics and prognosis of very late-onset myasthenia gravis in China. Neuromusc Disord NMD. 2023;33(4):358–66.
Acknowledgements
We extend our profound gratitude to the experts and scholars in the realm of statistics; their seminal contributions have enabled the seamless application of sophisticated methods in our clinical research. Our appreciation also goes to the developers of the R software, whose innovations have greatly streamlined our analytical processes. We are deeply indebted to all the patients who generously provided detailed information, contributing significantly to this study. Furthermore, we acknowledge the invaluable assistance and support received from various quarters during this research. Special mention must be made of Dr. Jingfei Zhang, Dr. Fei Zhao, Dr. Jiaying Shi, Dr. Qi Wen, and Dr. Hong Wang for their pivotal roles in the initial data collection phase.
Medical Writing and Editorial Assistance
The authors did not use any medical writing or editorial assistance for this article.
Funding
No funding or sponsorship was received for this study or publication of this article. The Rapid Service Fee was funded by the authors.
Author information
Authors and Affiliations
Contributions
Feng Liang and Wei Zhang were responsible for drafting the manuscript. The study protocol was conceived and designed by Feng Liang, Wei Zhang, Junhong Guo, and Xueli Chang. Clinical data acquisition was meticulously undertaken by Feng Liang, Yaqian Li, Guanxi Li, Jing Ma, Huiqiu Zhang, Xiaoqian Xia, Xiaomin Pang, Juan Wang, Zhaoxu Yin and Make Yao. The data analysis was executed by Feng Liang, Wei Zhang and Xueli Chang. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors report no conflicts of interest.
Ethical Approval
This research adhered to the ethical guidelines set forth in the Declaration of Helsinki and was approved by the ethical committee at The First Hospital of Shanxi Medical University (No. KYLL-2023-107). Due to the retrospective nature of the study, the requirement for written informed consent for the utilization of clinical data was waived.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Liang, F., Yin, Z., Li, Y. et al. Constructing and Validating a Nomogram Model for Short-Term Prognosis of Patients with AChR-Ab+ GMG. Neurol Ther (2024). https://doi.org/10.1007/s40120-024-00590-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40120-024-00590-0